
Minimally Invasive Urinary Incontinence Device Market Report 2026
Global Outlook – By Type (Tension Free Vaginal Tape (TVT), Tension Free Vaginal Tape (TVT) Obturator System, Other Types), By Age Group (Adults (18-65 years), Senior Population (65 Years And Above), Pediatric Population), By End-User (Hospitals, Clinics, Home Healthcare Settings, Research Institutions) – Market Size, Trends, Strategies, and Forecast to 2035
Minimally Invasive Urinary Incontinence Device Market Overview
• Minimally Invasive Urinary Incontinence Device market size has reached to $1.25 billion in 2025 • Expected to grow to $1.91 billion in 2030 at a compound annual growth rate (CAGR) of 8.8% • Growth Driver: Growing Incidence Of Prostate Cancer Driving The Market Growth Due To Rising Urinary Complications In Patients • Market Trend: Advancements In Minimally Invasive Neuromodulation Therapy For Urinary Incontinence • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Minimally Invasive Urinary Incontinence Device Market?
A minimally invasive urinary incontinence device is a medical device designed to manage or treat urinary incontinence using techniques that reduce surgical trauma, recovery time, and complication risks compared to traditional open procedures. These devices work by supporting the urethra, enhancing sphincter function, or modulating nerve activity to improve bladder control, and they are typically implanted or applied through small incisions, injections, or percutaneous approaches The main types of minimally invasive urinary incontinence devices are tension-free vaginal tape (TVT), tension-free vaginal tape obturator system (TVTO), and other types. Tension-free vaginal tape (TVT) is a minimally invasive surgical procedure that supports the urethra with a synthetic mesh to prevent stress urinary incontinence in women. These devices are used by various age groups, including adults (18–65 years), the senior population (65 years and above), and pediatric patients and are utilized across different end-users, such as hospitals, clinics, home healthcare settings, and research institutions.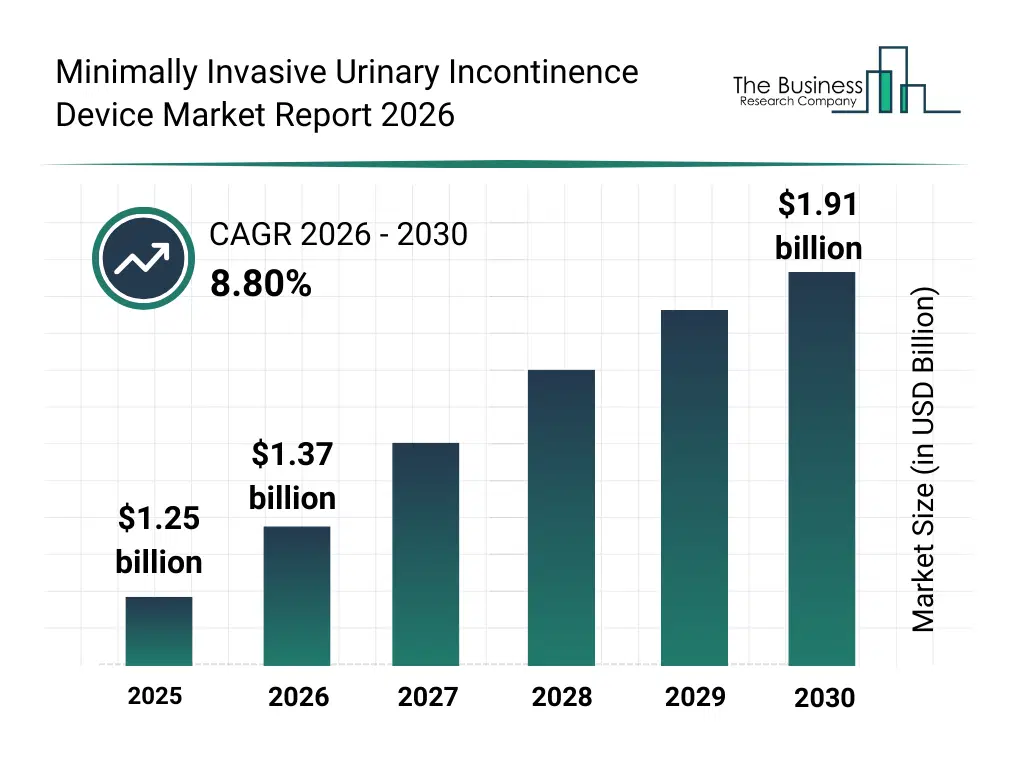
What Is The Minimally Invasive Urinary Incontinence Device Market Size and Share 2026?
The minimally invasive urinary incontinence device market size has grown strongly in recent years. It will grow from $1.25 billion in 2025 to $1.37 billion in 2026 at a compound annual growth rate (CAGR) of 9.1%. The growth in the historic period can be attributed to reliance on traditional open surgical procedures, limited device options for urinary incontinence, growing awareness of female pelvic health, increasing hospital and clinic-based procedures, adoption of tension-free vaginal tape (tvt) systems.What Is The Minimally Invasive Urinary Incontinence Device Market Growth Forecast?
The minimally invasive urinary incontinence device market size is expected to see strong growth in the next few years. It will grow to $1.91 billion in 2030 at a compound annual growth rate (CAGR) of 8.8%. The growth in the forecast period can be attributed to development of advanced mid-urethral and single-incision sling systems, rising investment in home healthcare devices, increasing geriatric population and incontinence prevalence, regulatory approvals for innovative bulking agents and artificial urinary sphincters, integration with smart and robotic-assisted surgical systems. Major trends in the forecast period include rising adoption of minimally invasive surgical procedures, growth in urinary incontinence cases due to aging population, increasing preference for home healthcare management, expansion of mid-urethral sling and sling systems, focus on reducing recovery time and surgical complications.Global Minimally Invasive Urinary Incontinence Device Market Segmentation
1) By Type: Tension Free Vaginal Tape (TVT), Tension Free Vaginal Tape (TVT) Obturator System, Other Types 2) By Age Group: Adults (18-65 years), Senior Population (65 Years And Above), Pediatric Population 3) By End-User: Hospitals, Clinics, Home Healthcare Settings, Research Institutions Subsegments: 1) By Tension Free Vaginal Tape (TVT): Mid Urethral Sling, Retropubic Sling, Single Incision Sling 2) By Tension Free Vaginal Tape Obturator System (TVTO): Inside Out Approach, Outside In Approach 3) By Other Types: Bulking Agents, Artificial Urinary Sphincter, Sling SystemsWhat Is The Driver Of The Minimally Invasive Urinary Incontinence Device Market?
The growing incidence of prostate cancer is expected to propel the growth of the minimally invasive urinary incontinence device market going forward. Prostate cancer is the uncontrolled growth of cells in the prostate gland that can impact urinary and reproductive health. The rising incidence of prostate cancer is largely driven by an aging male population, as older age is a significant risk factor for the disease. Minimally invasive urinary incontinence devices help manage prostate cancer-related urinary complications by providing targeted support to control bladder leakage, restore normal urinary function, and significantly improve the overall quality of life for patients undergoing treatment or recovery. For instance, in June 2025, according to the Centers for Disease Control and Prevention, a US-based government agency, in the United States, an estimated 255,395 new cases of prostate cancer were diagnosed in 2022, and in 2023, the disease was responsible for the deaths of approximately 33,881 men. Therefore, the growing incidence of prostate cancer is driving the growth of the minimally invasive urinary incontinence device industry.Key Players In The Global Minimally Invasive Urinary Incontinence Device Market
Major companies operating in the minimally invasive urinary incontinence device market are Johnson & Johnson Services Inc., Medtronic plc, Becton Dickinson and Company, Stryker Corporation, Boston Scientific Corporation, Olympus Corporation, Coloplast A/S, Teleflex Incorporated, Hollister Incorporated, CooperSurgical Inc., B. Braun Melsungen Aktiengesellschaft, LiNA Medical ApS, BlueWind Medical Ltd., Caldera Medical Inc., Neuspera Medical Inc., InControl Medical LLC, UroMems Inc., Rigicon Inc., Valencia Technologies Corporation, Zephyr Surgical Implants SAGlobal Minimally Invasive Urinary Incontinence Device Market Trends and Insights
Major companies operating in the minimally invasive urinary incontinence device market are focusing on developing advanced products such as neuromodulation therapy devices to offer effective long-term solutions with minimal surgical intervention. Neuromodulation therapy devices are implants that activate bladder-controlling nerves to regulate urinary function and help manage incontinence by enhancing bladder control and reducing accidental leakage. For instance, in December 2023, BlueWind Medical Ltd., a US-based company that manufactures neurostimulation devices, launched its Revi implantable tibial neuromodulation (iTNM) device. It is a battery-free, minimally invasive implant designed to treat urge urinary incontinence (UUI) by stimulating the posterior tibial nerve. The device is implanted near the ankle during a single outpatient procedure under local anesthesia and is activated using a lightweight wearable, allowing patients to manage symptoms conveniently at home. This launch reflects the ongoing trend of introducing innovative, patient-friendly neuromodulation solutions in the urinary incontinence device market.What Are Latest Mergers And Acquisitions In The Minimally Invasive Urinary Incontinence Device Market?
In November 2024, Boston Scientific Corporation, a US-based medical device company, acquired Axonics Inc. for approximately $3.7 billion. With this acquisition, Boston Scientific aims to expand its urology business into the high-growth sacral neuromodulation segment and offer a comprehensive portfolio of tailored treatment options for patients with urinary and bowel dysfunction, while leveraging Axonics’ innovative technologies to enhance patient outcomes, strengthen its global market presence, and drive long-term growth in the urology device sector. Axonics Inc. is a US-based medical technology company that develops minimally invasive urinary incontinence devices.Regional Outlook
North America was the largest region in the minimally invasive urinary incontinence device market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Minimally Invasive Urinary Incontinence Device Market?
The minimally invasive urinary incontinence device market consists of sales of wearable devices, catheter-based devices, sling systems, adjustable balloon systems and bulking agent delivery systems. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Minimally Invasive Urinary Incontinence Device Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.37 billion |
| Revenue Forecast In 2035 | $1.91 billion |
| Growth Rate | CAGR of 9.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Age Group, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Johnson & Johnson Services Inc., Medtronic plc, Becton Dickinson and Company, Stryker Corporation, Boston Scientific Corporation, Olympus Corporation, Coloplast A/S, Teleflex Incorporated, Hollister Incorporated, CooperSurgical Inc., B. Braun Melsungen Aktiengesellschaft, LiNA Medical ApS, BlueWind Medical Ltd., Caldera Medical Inc., Neuspera Medical Inc., InControl Medical LLC, UroMems Inc., Rigicon Inc., Valencia Technologies Corporation, Zephyr Surgical Implants SA |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
