Molecular Diagnostics For Sexually Transmitted Diseases Market Report 2026

Molecular Diagnostics For Sexually Transmitted Diseases Market Report 2026
Global Outlook – By Product (Instruments And Services, Consumables, Software), By Technology (Laboratory Testing, Point-Of-Care Testing), By Application (Chlamydia Trachomatis (CT) Or Neisseria Gonorrhoeae (NG) Testing, Syphilis Testing, Gonorrhea Testing, Herpes Simplex Virus (HSV) Testing, Human Papillomavirus (HPV) Testing, Human Immunodeficiency Virus (HIV) Testing, Trichomonas, Ureaplasma And Mycoplasma, Other Applications) – Market Size, Trends, Strategies, and Forecast to 2035
Molecular Diagnostics For Sexually Transmitted Diseases Market Overview
• Molecular Diagnostics For Sexually Transmitted Diseases market size has reached to $3.67 billion in 2025 • Expected to grow to $6.05 billion in 2030 at a compound annual growth rate (CAGR) of 10.4% • Growth Driver: Rising Prevalence Of STIs Fueling The Growth Of The Market Due To Increasing Need For Early And Accurate Detection • Market Trend: Innovation In Rapid Point-Of-Care PCR Devices Enhancing STI Diagnostic Accessibility And Efficiency • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Molecular Diagnostics For Sexually Transmitted Diseases Market?
Molecular diagnostics for sexually transmitted diseases involve techniques that detect the genetic material of infectious organisms, such as DNA or RNA, to identify the presence of pathogens. These methods are known for their high accuracy and sensitivity, enabling the detection of infections even when symptoms are not present. By analyzing the molecular structure of the pathogens, these diagnostics provide reliable identification, contributing to faster diagnosis. The main product types of molecular diagnostics for sexually transmitted diseases include instruments and services, consumables, and software. Instruments refer to the diagnostic devices and platforms used for detecting pathogens, while services include laboratory testing, sample processing, and diagnostic result interpretation provided by healthcare or diagnostic companies. These diagnostics utilize various technologies such as laboratory testing and point-of-care testing and are applied in a range of applications, including chlamydia trachomatis (CT) or neisseria gonorrhoeae (NG) testing, syphilis testing, gonorrhea testing, herpes simplex virus (HSV) testing, human papillomavirus (HPV) testing, human immunodeficiency virus (HIV) testing, trichomonas, ureaplasma and mycoplasma, and others.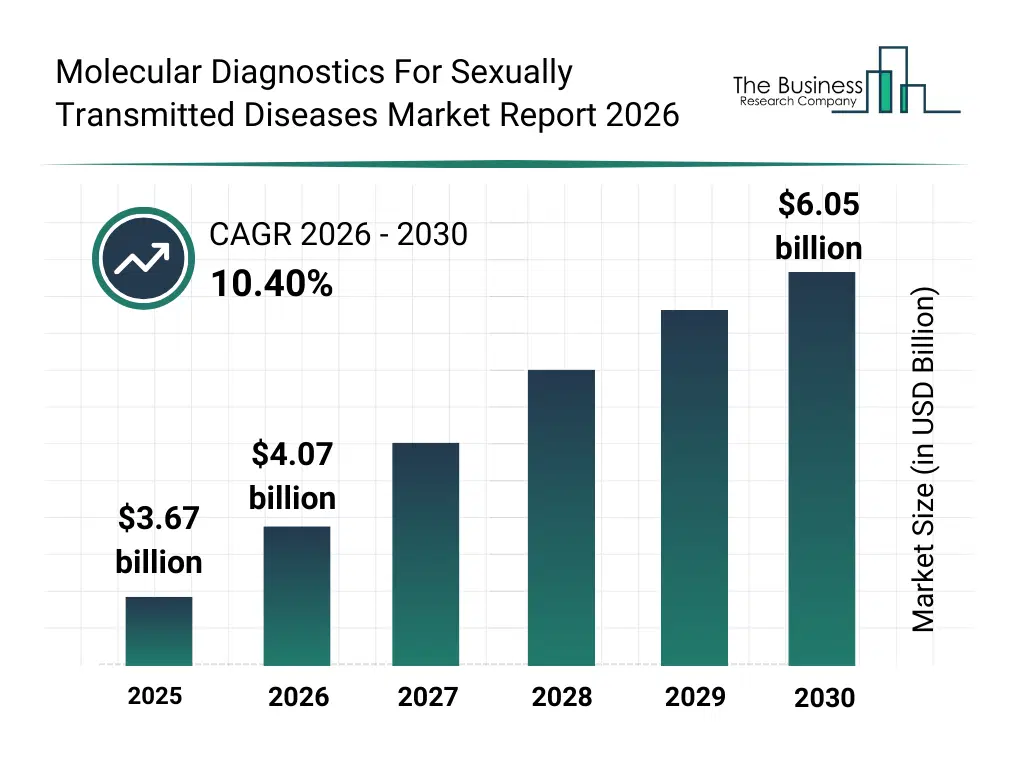
What Is The Molecular Diagnostics For Sexually Transmitted Diseases Market Size and Share 2026?
The molecular diagnostics for sexually transmitted diseases market size has grown rapidly in recent years. It will grow from $3.67 billion in 2025 to $4.07 billion in 2026 at a compound annual growth rate (CAGR) of 10.7%. The growth in the historic period can be attributed to reliance on conventional serological tests, limited availability of automated pcr systems, low awareness of molecular diagnostics in emerging markets, growing prevalence of sexually transmitted infections, manual workflow and labor-intensive testing.What Is The Molecular Diagnostics For Sexually Transmitted Diseases Market Growth Forecast?
The molecular diagnostics for sexually transmitted diseases market size is expected to see rapid growth in the next few years. It will grow to $6.05 billion in 2030 at a compound annual growth rate (CAGR) of 10.4%. The growth in the forecast period can be attributed to technological advancements in automated pcr and nucleic acid extraction, expansion of point-of-care molecular diagnostics, increasing healthcare infrastructure in developing regions, rising adoption of laboratory information management systems (lims), growing investment in r&d for rapid and multiplexed diagnostic assays. Major trends in the forecast period include increasing adoption of nucleic acid-based testing methods, expansion of point-of-care molecular diagnostic solutions, rising demand for rapid and accurate std detection, integration of molecular diagnostics with laboratory information systems, growth in automated pcr and high-throughput diagnostic platforms.Global Molecular Diagnostics For Sexually Transmitted Diseases Market Segmentation
1) By Product: Instruments And Services, Consumables, Software 2) By Technology: Laboratory Testing, Point-Of-Care Testing 3) By Application: Chlamydia Trachomatis (CT) Or Neisseria Gonorrhoeae (NG) Testing, Syphilis Testing, Gonorrhea Testing, Herpes Simplex Virus (HSV) Testing, Human Papillomavirus (HPV) Testing, Human Immunodeficiency Virus (HIV) Testing, Trichomonas, Ureaplasma And Mycoplasma, Other Applications Subsegments: 1) By Instruments And Services: Molecular Diagnostic Analyzers, Automated Polymerase Chain Reaction Systems, Nucleic Acid Extraction Instruments, Diagnostic Service Providers, Maintenance And Calibration Services, Installation And Training Services 2) By Consumables: Deoxyribonucleic Acid Extraction Kits, Ribonucleic Acid Extraction Kits, Polymerase Chain Reaction Reagents, Nucleic Acid Probes, Control And Reference Standards, Detection Enzymes And Buffers 3) By Software: Data Interpretation And Reporting Software, Polymerase Chain Reaction Workflow Management Software, Laboratory Information Management Systems, Diagnostic Decision Support Software, Quality Control Monitoring SoftwareWhat Is The Driver Of The Molecular Diagnostics For Sexually Transmitted Diseases Market?
The rising prevalence of sexually transmitted infections is expected to propel the growth of the molecular diagnostics for sexually transmitted diseases market going forward. Sexually transmitted infections (STIs) refer to infections caused by bacteria, viruses, or parasites that are transmitted through sexual contact, affecting the reproductive and urinary systems. The prevalence of sexually transmitted infections is increasing primarily due to the inconsistent use of protection methods. This lack of protection facilitates the easier transmission of infections among sexually active individuals. Molecular diagnostics for sexually transmitted infections (STIs) play a critical role in managing these infections by enabling early and accurate detection of pathogens, even in asymptomatic individuals, facilitating timely treatment, reducing disease transmission, and improving overall patient outcomes. For instance, in June 2024, according to the UK Health Security Agency (UKHSA), a UK-based government agency, in 2023, there were 401,800 new STI diagnoses in England, marking a 4.7% increase compared to the 383,789 cases reported in 2022. Therefore, a rising prevalence of sexually transmitted infections is driving the growth of the molecular diagnostics for sexually transmitted diseases industry.Key Players In The Global Molecular Diagnostics For Sexually Transmitted Diseases Market
Major companies operating in the molecular diagnostics for sexually transmitted diseases market are F. Hoffmann-La Roche Ltd, Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthineers AG, Illumina Inc., Hologic Inc., bioMérieux S.A., QuidelOrtho Corporation, QIAGEN N.V., Cepheid, Sansure Biotech Inc., OraSure Technologies Inc., Meridian Bioscience, Seegene Inc., Visby Medical Inc., Zeesan Biotech, Binx Health Inc., GCC Biotech, GeneProof, Solone Life Sciences India (P) Ltd.Global Molecular Diagnostics For Sexually Transmitted Diseases Market Trends and Insights
Major companies operating in the molecular diagnostics for sexually transmitted diseases market are focusing on developing innovative solutions, such as rapid point-of-care polymerase chain reaction (PCR) devices, to improve diagnostic speed, accuracy, and accessibility. Rapid point-of-care polymerase chain reaction (PCR) devices are portable molecular diagnostic tools that combine sample preparation, amplification, and detection into a single, compact unit, enabling timely and highly sensitive pathogen detection outside traditional laboratory settings. For instance, in March 2025, Visby Medical, a US-based diagnostic technology company, launched the Visby Medical Women’s Sexual Health Test. The product is a single-use, handheld PCR device designed to identify chlamydia, gonorrhea, and trichomoniasis from vaginal samples within 30 minutes. It operates without the need for external instrumentation or cold-chain logistics, features an integrated amplification system, and provides lab-quality results directly at the point of care to support immediate clinical decisions. The test is authorized for prescription home use, making it the first U.S. Food and Drug Administration (FDA)-cleared molecular diagnostic test for multiple sexually transmitted infections (STIs) that can be used outside clinical settings. This launch reflects a shift toward decentralized diagnostic solutions that increase accessibility, especially in underserved or remote populations.What Are Latest Mergers And Acquisitions In The Molecular Diagnostics For Sexually Transmitted Diseases Market?
In December 2024, OraSure Technologies, a US-based provider of point-of-care and home diagnostic solutions, acquired Sherlock Biosciences for an undisclosed amount. With this acquisition, OraSure aimed to expand its molecular diagnostics portfolio in sexual health by integrating Sherlock’s over-the-counter chlamydia trachomatis and neisseria gonorrhoeae self-test, which delivers laboratory-quality results in under 30 minutes using a clustered regularly interspaced short palindromic repeats-based and isothermal amplification-based platform. Sherlock Biosciences is a US-based biotechnology company specializing in molecular diagnostics for sexually transmitted diseases.Regional Outlook
North America was the largest region in the molecular diagnostics for sexually transmitted diseases market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Molecular Diagnostics For Sexually Transmitted Diseases Market?
The molecular diagnostics for sexually transmitted diseases market consists of revenues earned by entities by providing services such as test development, sample collection, nucleic acid amplification, and point of care diagnostics. The market value includes the value of related goods sold by the service provider or included within the service offering. The molecular diagnostics for sexually transmitted diseases market also includes sales of polymerase chain reaction kits, isothermal nucleic acid amplification kits, clustered regularly interspaced short palindromic repeats-based tests, and instrument-free molecular diagnostic systems. values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Molecular Diagnostics For Sexually Transmitted Diseases Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $4.07 billion |
| Revenue Forecast In 2035 | $6.05 billion |
| Growth Rate | CAGR of 10.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Technology, Application |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd, Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthineers AG, Illumina Inc., Hologic Inc., bioMérieux S.A., QuidelOrtho Corporation, QIAGEN N.V., Cepheid, Sansure Biotech Inc., OraSure Technologies Inc., Meridian Bioscience, Seegene Inc., Visby Medical Inc., Zeesan Biotech, Binx Health Inc., GCC Biotech, GeneProof, Solone Life Sciences India (P) Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Molecular Diagnostics For Sexually Transmitted Diseases Market Report 2026 market was valued at $3.67 billion in 2025, increased to $4.07 billion in 2026, and is projected to reach $6.05 billion by 2030.
request a sample hereThe expected CAGR for the Molecular Diagnostics For Sexually Transmitted Diseases Market Report 2026 market during the forecast period 2025–2030 is 10.4%.
request a sample hereMajor growth driver of the market includes: Rising Prevalence Of STIs Fueling The Growth Of The Market Due To Increasing Need For Early And Accurate Detection in the Molecular Diagnostics For Sexually Transmitted Diseases Market Report 2026 market. For further insights on this market,
request a sample hereThe molecular diagnostics for sexually transmitted diseases market covered in this report is segmented –
1) By Product: Instruments And Services, Consumables, Software
2) By Technology: Laboratory Testing, Point-Of-Care Testing
3) By Application: Chlamydia Trachomatis (CT) Or Neisseria Gonorrhoeae (NG) Testing, Syphilis Testing, Gonorrhea Testing, Herpes Simplex Virus (HSV) Testing, Human Papillomavirus (HPV) Testing, Human Immunodeficiency Virus (HIV) Testing, Trichomonas, Ureaplasma And Mycoplasma, Other Applications Subsegments:
1) By Instruments And Services: Molecular Diagnostic Analyzers, Automated Polymerase Chain Reaction Systems, Nucleic Acid Extraction Instruments, Diagnostic Service Providers, Maintenance And Calibration Services, Installation And Training Services
2) By Consumables: Deoxyribonucleic Acid Extraction Kits, Ribonucleic Acid Extraction Kits, Polymerase Chain Reaction Reagents, Nucleic Acid Probes, Control And Reference Standards, Detection Enzymes And Buffers
3) By Software: Data Interpretation And Reporting Software, Polymerase Chain Reaction Workflow Management Software, Laboratory Information Management Systems, Diagnostic Decision Support Software, Quality Control Monitoring Software
request a sample here1) By Product: Instruments And Services, Consumables, Software
2) By Technology: Laboratory Testing, Point-Of-Care Testing
3) By Application: Chlamydia Trachomatis (CT) Or Neisseria Gonorrhoeae (NG) Testing, Syphilis Testing, Gonorrhea Testing, Herpes Simplex Virus (HSV) Testing, Human Papillomavirus (HPV) Testing, Human Immunodeficiency Virus (HIV) Testing, Trichomonas, Ureaplasma And Mycoplasma, Other Applications Subsegments:
1) By Instruments And Services: Molecular Diagnostic Analyzers, Automated Polymerase Chain Reaction Systems, Nucleic Acid Extraction Instruments, Diagnostic Service Providers, Maintenance And Calibration Services, Installation And Training Services
2) By Consumables: Deoxyribonucleic Acid Extraction Kits, Ribonucleic Acid Extraction Kits, Polymerase Chain Reaction Reagents, Nucleic Acid Probes, Control And Reference Standards, Detection Enzymes And Buffers
3) By Software: Data Interpretation And Reporting Software, Polymerase Chain Reaction Workflow Management Software, Laboratory Information Management Systems, Diagnostic Decision Support Software, Quality Control Monitoring Software
Major trend in this market includes: Innovation In Rapid Point-Of-Care PCR Devices Enhancing STI Diagnostic Accessibility And Efficiency For further insights on this market,
request a sample hereMajor companies operating in the Molecular Diagnostics For Sexually Transmitted Diseases Market Report 2026 market are Major companies operating in the molecular diagnostics for sexually transmitted diseases market are F. Hoffmann-La Roche Ltd, Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthineers AG, Illumina Inc., Hologic Inc., bioMérieux S.A., QuidelOrtho Corporation, QIAGEN N.V., Cepheid, Sansure Biotech Inc., OraSure Technologies Inc., Meridian Bioscience, Seegene Inc., Visby Medical Inc., Zeesan Biotech, Binx Health Inc., GCC Biotech, GeneProof, Solone Life Sciences India (P) Ltd.
request a sample hereNorth America was the largest region in the molecular diagnostics for sexually transmitted diseases market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the molecular diagnostics for sexually transmitted diseases market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
request a sample here