
mRNA Therapeutics CDMO Market Report 2026
Global Outlook – By Type Of Service (Development Services, Manufacturing Services, Laboratory Services), By Indication (Infectious Diseases, Metabolic And Genetic Diseases, Cardiovascular And Cerebrovascular Diseases), By Application (Viral Vaccines, Protein Replacement Therapies, Cancer Immunotherapies), By End User (Pharmaceutical Companies, Biotechnology Companies, Academic Institutions, Research Organizations) – Market Size, Trends, Strategies, and Forecast to 2035
mRNA Therapeutics CDMO Market Overview
• mRNA Therapeutics CDMO market size has reached to $4.74 billion in 2025 • Expected to grow to $8.26 billion in 2030 at a compound annual growth rate (CAGR) of 11.7% • Growth Driver: The Growing Burden Of Chronic And Infectious Diseases Fueling The Market Due To Increasing Therapeutic Demand • Market Trend: Advancing mRNA Vaccines With In Situ Nanomedicine For Targeted Immunity • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under mRNA Therapeutics CDMO Market?
An mRNA therapeutics CDMO, or messenger ribonucleic acid therapeutics contract development and manufacturing organization, is a specialized third-party entity that provides end-to-end support for the research, development, and scalable production of mRNA-based therapeutic agents under contractual arrangements with pharmaceutical or biotechnology companies. These organizations play a critical role in accelerating time-to-market, ensuring regulatory compliance, and enabling flexible capacity for mRNA therapeutic pipelines. The main types of services in the mRNA therapeutics CDMO are development services, manufacturing services, and laboratory services. Development services of mRNA therapeutics CDMO refers to the end-to-end support provided for the design, optimization, and scale-up of mRNA-based therapeutics, including synthesis, formulation, analytical development, and process development tailored to client-specific product requirements. These therapeutics are used in the indications including infectious diseases, metabolic and genetic diseases, and cardiovascular and cerebrovascular diseases in applications such as viral vaccines, protein replacement therapies, and cancer immunotherapies used by the end users, including pharmaceutical companies, biotechnology companies, academic institutions, and research organizations.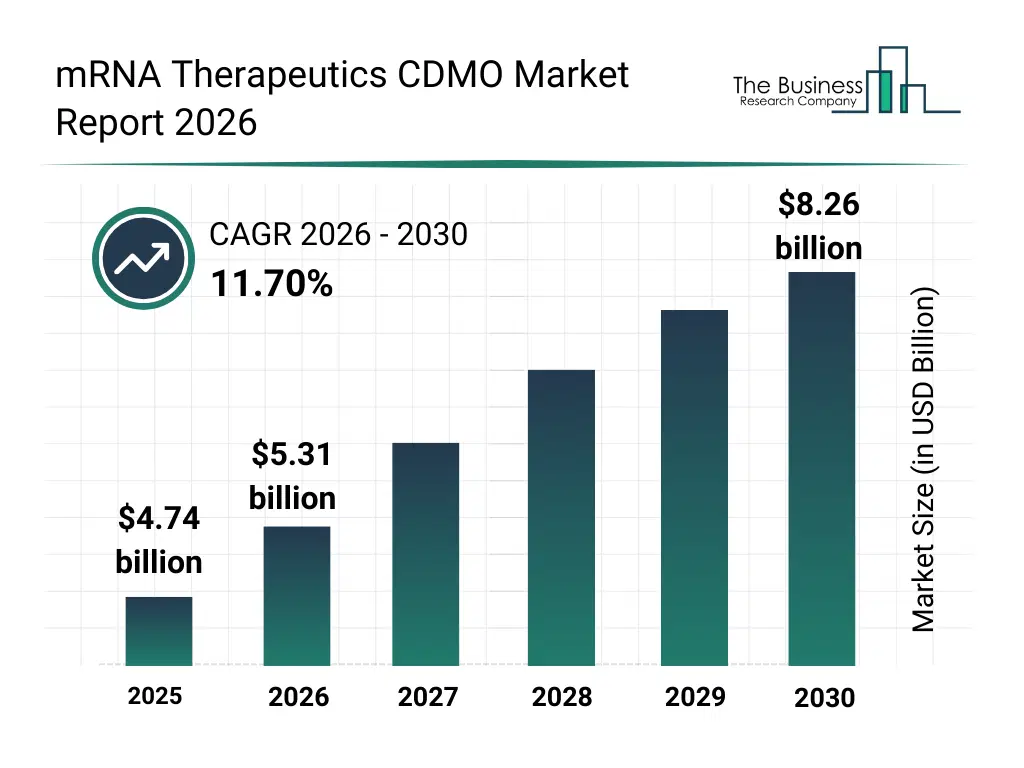
What Is The mRNA Therapeutics CDMO Market Size and Share 2026?
The mrna therapeutics cdmo market size has grown rapidly in recent years. It will grow from $4.74 billion in 2025 to $5.31 billion in 2026 at a compound annual growth rate (CAGR) of 12.0%. The growth in the historic period can be attributed to limited availability of specialized mrna cdmo services, reliance on in-house pharmaceutical manufacturing, growing infectious disease outbreaks, adoption of early-stage analytical testing, expansion of academic and research collaborations.What Is The mRNA Therapeutics CDMO Market Growth Forecast?
The mrna therapeutics cdmo market size is expected to see rapid growth in the next few years. It will grow to $8.26 billion in 2030 at a compound annual growth rate (CAGR) of 11.7%. The growth in the forecast period can be attributed to rising investment in mrna therapeutics pipelines, technological advancements in plasmid dna and mrna synthesis, growth of cancer immunotherapy and protein replacement therapies, increasing demand for fill-finish services, expansion of partnerships between biotechs and cdmos. Major trends in the forecast period include rising demand for outsourced mrna development and manufacturing, increasing focus on lipid nanoparticle (lnp) formulation technologies, expansion of viral vaccine and cancer immunotherapy pipelines, enhanced regulatory compliance and quality assurance measures, growing adoption of analytical and bioanalytical laboratory services.Global mRNA Therapeutics CDMO Market Segmentation
1) By Type Of Service: Development Services, Manufacturing Services, Laboratory Services 2) By Indication: Infectious Diseases, Metabolic And Genetic Diseases, Cardiovascular And Cerebrovascular Diseases 3) By Application: Viral Vaccines, Protein Replacement Therapies, Cancer Immunotherapies 4) By End User: Pharmaceutical Companies, Biotechnology Companies, Academic Institutions, Research Organizations Subsegments: 1) By Development Services: Process Development, Analytical Method Development, Formulation Development, Regulatory Support 2) By Manufacturing Services: Plasmid DNA Manufacturing, mRNA Synthesis, Lipid Nanoparticle (LNP) Formulation, Fill-Finish Services 3) By Laboratory Services: Quality Control Testing, Stability Testing, Bioanalytical Testing, Preclinical TestingWhat Is The Driver Of The mRNA Therapeutics CDMO Market?
The growing burden of chronic and infectious diseases is expected to propel the growth of the mRNA therapeutics CDMO market going forward. Chronic and infectious diseases encompass long-term health conditions such as diabetes, cardiovascular disorders, and asthma, as well as illnesses caused by pathogenic microorganisms, including viruses, bacteria, and parasites, which can spread directly or indirectly among individuals. Chronic and infectious diseases are increasing due to aging populations, which are more susceptible to both long-term illnesses and infections. mRNA therapeutics CDMO helps address chronic and infectious diseases by providing end-to-end development and manufacturing services that accelerate the delivery of targeted, personalized mRNA therapies to patients. For instance, in October 2024, according to the World Health Organization, a Switzerland-based international public health organization, approximately 8.2 million people were newly diagnosed with tuberculosis in 2023, up from about 7.5 million in 2022, highlighting the escalating burden of infectious diseases globally. Therefore, the growing burden of chronic and infectious diseases is driving the growth of the mRNA therapeutics CDMO industry.Key Players In The Global mRNA Therapeutics CDMO Market
Major companies operating in the mrna therapeutics cdmo market are Thermo Fisher Scientific Inc., Danaher Corporation, Merck KGaA, FUJIFILM Holdings Corporation, Evonik Industries AG, AGC Biologics GmbH, Eurofins Scientific SA, Samsung Biologics Co. Ltd., Wuxi Biologics, Recipharm AB, EMERGENT BioSolutions Inc., Genscript Biotech, Ginkgo Bioworks Holdings Inc., Corden Pharma Corp., TriLink BioTechnologies LLC, Biocina Pty Ltd., Aldevron LLC, Synbio Technologies LLC, etherna manufacturing, Bio-Synthesis Inc., Biomay AG.Global mRNA Therapeutics CDMO Market Trends and Insights
Major companies operating in the mRNA therapeutics CDMO market are focusing on developing innovative solutions, such as in situ nanomedicine vaccines, to enhance the effectiveness and precision of mRNA-based vaccines through targeted immune stimulation. An in situ nanomedicine vaccine is a next-generation mRNA-based therapeutic designed to generate nanomedicines within the patient’s body, enabling targeted delivery and customized immune activation through engineered transgene expression. For instance, in January 2025, Esphera SynBio Inc., a Canada-based biotechnology company, launched a new project supported by the CQDM Quantum Leap program. The initiative aims to enhance first-generation lipid nanoparticle or messenger RNA (LNP/mRNA) vaccines by incorporating proprietary transgene designs that induce in vivo production of immunostimulatory nanomedicines. This approach enables precise antigen delivery and tailored immune responses, positioning the technology as a potential breakthrough in vaccine innovation.What Are Latest Mergers And Acquisitions In The mRNA Therapeutics CDMO Market?
In September 2024, Agilent Technologies Inc., a US-based medical equipment company, acquired BIOVECTRA for $0.925 billion. This deal aims to expand Agilent’s CDMO capabilities into high-growth modalities, including mRNA therapeutics, by adding BIOVECTRA’s expertise in biologics and advanced pharmaceutical manufacturing. BIOVECTRA is a Canada-based contract development and manufacturing organization specializing in mRNA therapeutics and other modalities.Regional Insights
North America was the largest region in the mRNA therapeutics CDMO market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the mRNA Therapeutics CDMO Market?
The mRNA therapeutics CDMO market consists of revenues earned by entities by providing services such as supply chain management, cold chain logistics, quality assurance auditing, clinical trial material preparation, batch release testing, serialization, and packaging. The market value includes the value of related goods sold by the service provider or included within the service offering. The mRNA therapeutics CDMO market also includes sales of products such as lipid excipients, nucleotide reagents, capping analogs, custom RNA oligonucleotides, and modified nucleosides. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the mRNA Therapeutics CDMO Market Report 2026?
The mrna therapeutics cdmo market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the mrna therapeutics cdmo industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.mRNA Therapeutics CDMO Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $5.31 billion |
| Revenue Forecast In 2035 | $8.26 billion |
| Growth Rate | CAGR of 12.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type Of Service, Indication, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Danaher Corporation, Merck KGaA, FUJIFILM Holdings Corporation, Evonik Industries AG, AGC Biologics GmbH, Eurofins Scientific SA, Samsung Biologics Co. Ltd., Wuxi Biologics, Recipharm AB, EMERGENT BioSolutions Inc., Genscript Biotech, Ginkgo Bioworks Holdings Inc., Corden Pharma Corp., TriLink BioTechnologies LLC, Biocina Pty Ltd., Aldevron LLC, Synbio Technologies LLC, etherna manufacturing, Bio-Synthesis Inc., Biomay AG. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
