
Mucopolysaccharidosis Treatment Market Report 2026
Global Outlook – By Treatment (Enzyme Replacement Therapy, Stem Cell Therapy), By Disease (Mucopolysaccharidosis- I, Mucopolysaccharidosis- II, Mucopolysaccharidosis-IV, Mucopolysaccharidosis-VI, Other Diseases), By Route Of Administration (Intravenous, Intracerebroventricular), By End-User (Hospitals, Specialty Clinics, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Mucopolysaccharidosis Treatment Market Overview
• Mucopolysaccharidosis Treatment market size has reached to $2.58 billion in 2025 • Expected to grow to $3.79 billion in 2030 at a compound annual growth rate (CAGR) of 8% • Growth Driver: Rising Prevalence Of Mucopolysaccharidosis Sparks Growth In The Mucopolysaccharidosis Treatment Market • Market Trend: Regulatory Progress Strengthens Future Options For Hunter Syndrome Care • North America was the largest region in 2025.What Is Covered Under Mucopolysaccharidosis Treatment Market?
Mucopolysaccharidosis (MPS) treatment is the therapy of hereditary illnesses in which the body cannot break down glycosaminoglycan (long sugar chains) due to the lack or failure of certain enzymes. A deficiency or malfunction of a specific lysosomal enzyme commonly causes mucopolysaccharidosis. The main types of mucopolysaccharidosis treatments are enzyme replacement therapy and stem cell therapy. Enzyme replacement therapy refers to a medical procedure in which patients who experience chronic diseases brought on by enzyme deficiency or dysfunction receive replacement enzymes. They are administered for various diseases such as mucopolysaccharidosis I, mucopolysaccharidosis II, mucopolysaccharidosis IV, mucopolysaccharidosis VI, and others. They are available via intravenous and intracerebroventricular routes of administration and are used by various end-users, such as hospitals, specialty clinics, and others.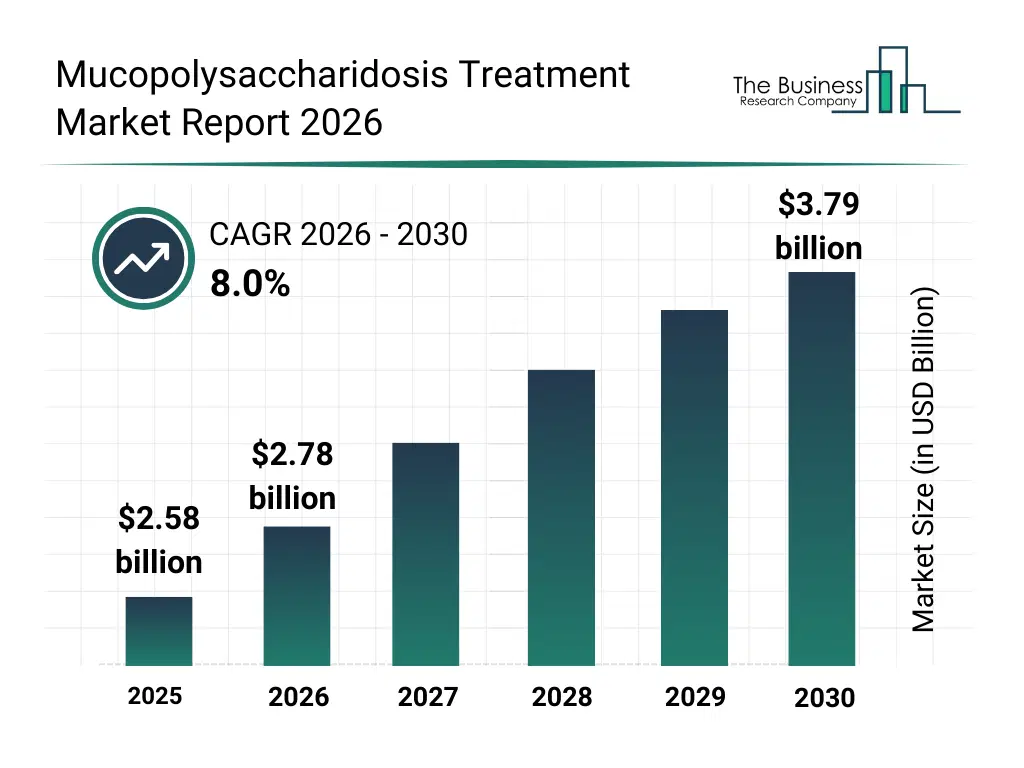
What Is The Mucopolysaccharidosis Treatment Market Size and Share 2026?
The mucopolysaccharidosis treatment market size has grown strongly in recent years. It will grow from $2.58 billion in 2025 to $2.78 billion in 2026 at a compound annual growth rate (CAGR) of 7.7%. The growth in the historic period can be attributed to limited awareness of mps disorders, high treatment costs, slow adoption of enzyme therapy, restricted clinical trial availability, reliance on international drug supply.What Is The Mucopolysaccharidosis Treatment Market Growth Forecast?
The mucopolysaccharidosis treatment market size is expected to see strong growth in the next few years. It will grow to $3.79 billion in 2030 at a compound annual growth rate (CAGR) of 8.0%. The growth in the forecast period can be attributed to expansion of gene therapy research, increasing healthcare investments, improved rare disease screening programs, broader reimbursement coverage, growth in precision medicine platforms. Major trends in the forecast period include advancements in enzyme replacement therapy, growing adoption of stem cell transplantation, increasing development of gene therapy approaches, rising focus on early diagnosis and newborn screening, expansion of specialized treatment centers.Global Mucopolysaccharidosis Treatment Market Segmentation
1) By Treatment: Enzyme Replacement Therapy, Stem Cell Therapy 2) By Disease: Mucopolysaccharidosis- I, Mucopolysaccharidosis- II, Mucopolysaccharidosis-IV, Mucopolysaccharidosis-VI, Other Diseases 3) By Route Of Administration: Intravenous, Intracerebroventricular 4) By End-User: Hospitals, Specialty Clinics, Other End-Users Subsegments: 1) By Enzyme Replacement Therapy: Idursulfase (Elaprase), Laronidase (Aldurazyme), Velaglucerase Alfa (Vpriv), Galsulfase (Naglazyme) 2) By Stem Cell Therapy: Hematopoietic Stem Cell Transplantation (HSCT), Umbilical Cord Blood Transplantation, Gene Therapy ApproachesWhat Is The Driver Of The Mucopolysaccharidosis Treatment Market?
The increasing prevalence of various forms of mucopolysaccharidosis is expected to propel the growth of the mucopolysaccharidosis treatment market going forward. Mucopolysaccharide is a form of complex carbohydrate that accumulates in bodily tissues because the individual affected lacks the particular enzyme required to digest it. Mucopolysaccharidosis (MPS) therapy can help prevent disease development by lowering glycosaminoglycan (GAG) formation in the body, controlling symptoms, and preventing complications through various therapeutic approaches. For Instance, in July 2024, according to the Orphanet Journal of Rare Diseases, a France-based peer-reviewed open-access medical journal, the incidence of mucopolysaccharidosis IVa (MPS IVa) is estimated at around 1 in 201,000 live births, though this figure is thought to vary considerably among different populations. Similarly, the birth prevalence of Mucopolysaccharidosis VI (MPS VI) is estimated to range between 1 in 43,261 and 1 in 1,505,160, depending on the population. Therefore, the increasing prevalence of various forms of mucopolysaccharidosis will drive the mucopolysaccharidosis treatment industry.Key Players In The Global Mucopolysaccharidosis Treatment Market
Major companies operating in the mucopolysaccharidosis treatment market are BioMarin Pharmaceuticals Inc., Sanofi S.A., Takeda Pharmaceutical Co. Ltd., JCR Pharmaceuticals Co. Ltd., Ultragenyx Pharmaceutical Inc., RegenxBio Inc., Sangamo Therapeutics Inc., Audentes Therapeutics Inc., Abeona Therapeutics Inc., Immusoft Corporation, Neurogene Inc., Mallinckrodt Pharmaceuticals, Green Cross Corporation, Denali Therapeutics Inc., Passage Bio Inc., Homology Medicines Inc., AVROBIO Inc., Rocket Pharmaceuticals Inc., Taysha Gene Therapies Inc., Freeline Therapeutics Holdings plc, 4D Molecular Therapeutics Inc., Sio Gene Therapies Inc., SpliceBio S.L., LogicBio Therapeutics Inc., Orchard Therapeutics plc, Asklepios BioPharmaceutical Inc.Global Mucopolysaccharidosis Treatment Market Trends and Insights
Major companies operating in the MPS II treatment market are focusing on developing innovative products such as AAV gene therapies to address the underlying genetic drivers of the disorder and pursue durable neurological correction. An AAV gene therapy is a targeted genetic intervention that uses adeno-associated viral vectors to deliver functional genes into central nervous system cells, enabling long-term production of missing or deficient proteins. For instance, in August 2025, REGENXBIO Inc., a US-based biotechnology company, reported that the United States Food and Drug Administration extended its review timeline for RGX-121. The extension follows the submission of longer-term clinical data from all pivotal-study participants, which remained consistent with earlier biomarker and neurodevelopmental outcomes. The clean pre-license inspections and absence of safety concerns indicate ongoing regulatory momentum, reinforcing the therapy’s potential to become the first one-time treatment targeting the neurological manifestations of MPS II.What Are Latest Mergers And Acquisitions In The Mucopolysaccharidosis Treatment Market?
In January 2024, Kyowa Kirin Co., Ltd., a Japan-based pharmaceutical company acquired Orchard Therapeutics for an undisclosed amount. This acquisition is significant as it combines Kyowa Kirin’s established expertise in biopharmaceuticals with Orchard’s cutting-edge gene therapy platform, aimed at delivering life-changing treatments for patients with severe genetic disorders. Orchard Therapeutics is a UK-based biotech company that provides mucopolysaccharidosis treatment.Regional Outlook
North America was the largest region in the mucopolysaccharidosis treatment market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Mucopolysaccharidosis Treatment Market?
The mucopolysaccharides treatment market includes revenues earned by entities by providing medication services such as enzyme replacement therapy, stem cell transplantation, gene therapy, substrate reduction therapy, and palliative care. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Mucopolysaccharidosis Treatment Market Report 2026?
The mucopolysaccharidosis treatment market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the mucopolysaccharidosis treatment industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Mucopolysaccharidosis Treatment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.78 billion |
| Revenue Forecast In 2035 | $3.79 billion |
| Growth Rate | CAGR of 7.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Treatment, Disease, Route Of Administration, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | BioMarin Pharmaceuticals Inc., Sanofi S.A., Takeda Pharmaceutical Co. Ltd., JCR Pharmaceuticals Co. Ltd., Ultragenyx Pharmaceutical Inc., RegenxBio Inc., Sangamo Therapeutics Inc., Audentes Therapeutics Inc., Abeona Therapeutics Inc., Immusoft Corporation, Neurogene Inc., Mallinckrodt Pharmaceuticals, Green Cross Corporation, Denali Therapeutics Inc., Passage Bio Inc., Homology Medicines Inc., AVROBIO Inc., Rocket Pharmaceuticals Inc., Taysha Gene Therapies Inc., Freeline Therapeutics Holdings plc, 4D Molecular Therapeutics Inc., Sio Gene Therapies Inc., SpliceBio S.L., LogicBio Therapeutics Inc., Orchard Therapeutics plc, Asklepios BioPharmaceutical Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
