
Nasal Spray Influenza Vaccine Market Report 2026
Global Outlook – By Type (Live Attenuated Influenza Vaccine (LAIV), Inactivated Influenza Vaccine), By Administration Method (Single-Dose Administration, Multi-Dose Administration), By Age Group (Pediatric, Adult, Geriatric), By Distribution Channel (Hospitals, Clinics, Pharmacies, Online Stores, Other Distribution Channels), By End User (Healthcare Providers, Individuals, Government Health Departments, Pharmaceutical Companies) - Market Size, Trends, And Global Forecast 2026-2035
Nasal Spray Influenza Vaccine Market Overview
• Nasal Spray Influenza Vaccine market size has reached to $1.48 billion in 2025 • Expected to grow to $2.68 billion in 2030 at a compound annual growth rate (CAGR) of 12.7% • Growth Driver: Increasing Prevalence Of Influenza Is Fueling The Market Growth Due To Rising Infection Rates And Virus Mutations • Market Trend: The Role Of Live Attenuated Influenza Vaccines In Immune Response • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Nasal Spray Influenza Vaccine Market?
Nasal spray influenza vaccine refers to a needle-free flu vaccine administered through the nose that uses live attenuated viruses to protect against seasonal influenza. Nasal spray influenza vaccine is utilized by spraying it into the nostrils, where it stimulates an immune response in the nasal passages to protect against seasonal flu. The main types of nasal spray influenza vaccines include the live attenuated influenza vaccine (LAIV) and the inactivated influenza vaccine. The live attenuated influenza vaccine (LAIV) is a needle-free flu vaccine that uses weakened live viruses to trigger strong systemic and mucosal immune responses. These vaccines are administered through various methods, such as single-dose and multi-dose administration. They are intended for different age groups, including pediatric, adult, and geriatric populations. Distribution channels include hospitals, clinics, pharmacies, online stores, and other outlets. The end users of nasal spray influenza vaccines include healthcare providers, individuals, government health departments, and pharmaceutical companies.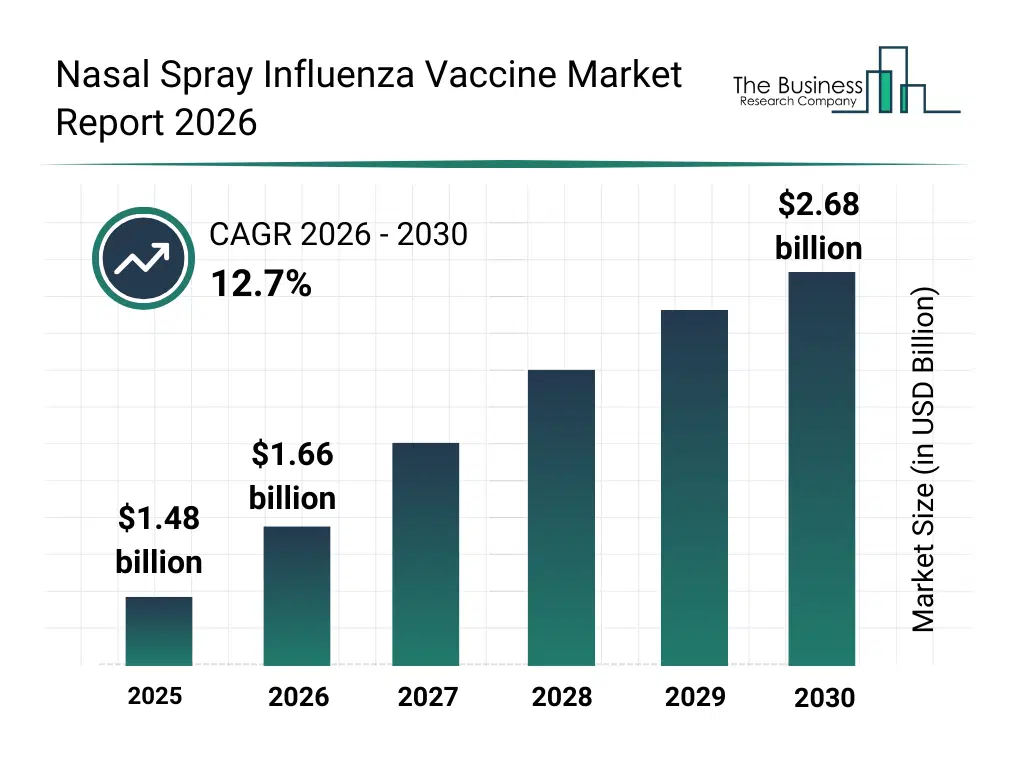
What Is The Nasal Spray Influenza Vaccine Market Size and Share 2026?
The nasal spray influenza vaccine market size has grown rapidly in recent years. It will grow from $1.48 billion in 2025 to $1.66 billion in 2026 at a compound annual growth rate (CAGR) of 12.7%. The growth in the historic period can be attributed to increasing flu vaccination awareness, reliance on traditional injection-based flu vaccines, government-led influenza immunization programs, seasonal flu outbreaks, limited availability of needle-free options.What Is The Nasal Spray Influenza Vaccine Market Growth Forecast?
The nasal spray influenza vaccine market size is expected to see rapid growth in the next few years. It will grow to $2.68 billion in 2030 at a compound annual growth rate (CAGR) of 12.7%. The growth in the forecast period can be attributed to growing demand for needle-free delivery systems, expansion of pediatric and geriatric immunization programs, rising investment in nasal vaccine r&d, development of advanced attenuated vaccine formulations, increasing global vaccination coverage initiatives. Major trends in the forecast period include growing adoption of needle-free vaccination methods, rising demand for pediatric-friendly flu vaccines, expanding use of quadrivalent live attenuated vaccines, increasing seasonal flu prevention campaigns, development of multi-dose and single-dose nasal formulations.Global Nasal Spray Influenza Vaccine Market Segmentation
1) By Type: Live Attenuated Influenza Vaccine (LAIV), Inactivated Influenza Vaccine 2) By Administration Method: Single-Dose Administration, Multi-Dose Administration 3) By Age Group: Pediatric, Adult, Geriatric 4) By Distribution Channel: Hospitals, Clinics, Pharmacies, Online Stores, Other Distribution Channels 5) By End User: Healthcare Providers, Individuals, Government Health Departments, Pharmaceutical Companies Subsegments: 1) By Live Attenuated Influenza Vaccine (LAIV): Trivalent Live Attenuated Influenza Vaccine (Trivalent LAIV), Quadrivalent Live Attenuated Influenza Vaccine (Quadrivalent LAIV) 2) By Inactivated Influenza Vaccine: Whole Virus Inactivated Vaccine, Split Virus Inactivated Vaccine, Subunit Inactivated Vaccine, Adjuvanted Inactivated Vaccine, High-Dose Inactivated VaccineWhat Is The Driver Of The Nasal Spray Influenza Vaccine Market?
The increasing prevalence of influenza is expected to propel the growth of nasal spray influenza vaccine going forward. Influenza refers to a highly contagious viral infection that primarily affects the respiratory system, causing symptoms such as fever, cough, sore throat, body aches, and fatigue. The increasing prevalence of influenza is primarily due to the frequent genetic mutations of the influenza virus, which enable it to evade existing immunity and spread rapidly among populations. Nasal spray influenza vaccine helps protect against influenza by delivering a weakened form of the virus directly into the nasal passages, stimulating an immune response where the virus typically enters the body. For instance, in September 2024, according to the data published by the Centers for Disease Control and Prevention (CDC), a US-based nation's health protection agency, preliminary estimates for the 2023–2024 U.S. influenza season suggest that between 34 million and 75 million symptomatic illnesses, 15 million to 33 million medical visits, 380,000 to 900,000 hospitalizations, and 17,000 to 100,000 deaths occurred, with older adults (≥65 years) accounting for 51% of hospitalizations and 68% of deaths. Therefore, the increasing prevalence of influenza is driving the growth of the nasal spray influenza vaccine industry.Key Players In The Global Nasal Spray Influenza Vaccine Market
Major companies operating in the nasal spray influenza vaccine market are Sanofi Pasteur SA, GSK plc, FluGen Inc., BioDiem Ltd, Codagenix Inc, Altimmune Inc, AstraZeneca PLC, MedImmune LLC, BCHT Biotechnology Co Ltd, Serum Institute of India Pvt Ltd, Vivaldi Biosciences, Vaccitech, Blue Water Vaccines, VaxInnate, INOVIO Pharmaceuticals, Cipla Ltd, Beijing Wantai Biological Pharmacy Enterprise Co Ltd, Bharat Biotech, Novavax Inc, BioNTech SE, Transgene SA, BiondVax Pharmaceuticals Ltd, Adimmune Corporation, Teva Pharmaceutical Industries LtdGlobal Nasal Spray Influenza Vaccine Market Trends and Insights
Major companies operating in the nasal spray influenza vaccine market are focusing on developing innovative formulations such as live attenuated influenza vaccine to enhance immune response and improve patient compliance. A live attenuated influenza vaccine (LAIV) is a flu vaccine made from live, weakened influenza viruses that helps prevent influenza by stimulating the immune system to produce antibodies, providing protection without causing the disease, and offering a more comprehensive immune response through both local and systemic immunity, which enhances the body's ability to defend against future flu infections. For instance, in September 2024, AstraZeneca Plc, a UK-based pharmaceutical industry company, received approval from the U.S. Food and Drug Administration (FDA) for FluMist, a live attenuated influenza vaccine (LAIV), to be self-administered by adults aged up to 49 years or administered by caregivers to children aged 2 to 17 years, making it the first and only influenza vaccine approved for self- or caregiver-administration in the United States. This approval was based on comprehensive usability studies that demonstrated the ability of individuals to correctly self-administer the vaccine, with 100% of participants delivering a full dose as instructed.What Are Latest Mergers And Acquisitions In The Nasal Spray Influenza Vaccine Market?
In October 2023, Mylab Discovery Solutions Private Limited, an India-based biotechnology company, partnered with Serum Institute of India Private Limited to launch Nasovac S4, India's first nasal influenza vaccine. With this partnership, Mylab Discovery Solutions Private Limited and Serum Institute of India Private Limited aim to offer comprehensive protection against a wide range of influenza strains through the introduction of Nasovac S4, India's first needle-free nasal influenza vaccine. Serum Institute of India Private Limited is an India-based biotechnology company that specializes in providing Nasovac-S and Nasovac-S4 nasal influenza vaccines.Regional Outlook
North America was the largest region in the nasal spray influenza vaccine market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Nasal Spray Influenza Vaccine Market?
The nasal spray influenza vaccine market consists of sales of flumist quadrivalent, fluenz tetra, vaxinate, centripetal flu nasal spray vaccine, nasovac-s, fluvac nasal spray, and live attenuated influenza vaccine. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Nasal Spray Influenza Vaccine Market Report 2026?
The nasal spray influenza vaccine market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the nasal spray influenza vaccine industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Nasal Spray Influenza Vaccine Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.66 billion |
| Revenue Forecast In 2035 | $2.68 billion |
| Growth Rate | CAGR of 12.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Administration Method, Age Group, Distribution Channel, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Sanofi Pasteur SA, GSK plc, FluGen Inc., BioDiem Ltd, Codagenix Inc, Altimmune Inc, AstraZeneca PLC, MedImmune LLC, BCHT Biotechnology Co Ltd, Serum Institute of India Pvt Ltd, Vivaldi Biosciences, Vaccitech, Blue Water Vaccines, VaxInnate, INOVIO Pharmaceuticals, Cipla Ltd, Beijing Wantai Biological Pharmacy Enterprise Co Ltd, Bharat Biotech, Novavax Inc, BioNTech SE, Transgene SA, BiondVax Pharmaceuticals Ltd, Adimmune Corporation, Teva Pharmaceutical Industries Ltd |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
