Neonatal Point Of Care Testing Market Report 2026

Neonatal Point Of Care Testing Market Report 2026
Global Outlook – By Product (Instruments, Assay), By Test Type (Neonatal Bilirubin Testing, Blood Glucose Testing, Infectious Disease Screening, Electrolyte Testing, Other Test Types), By End-Users (Hospitals, Maternity And Specialty Clinics, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Neonatal Point Of Care Testing Market Overview
• Neonatal Point Of Care Testing market size has reached to $1.32 billion in 2025 • Expected to grow to $2.59 billion in 2030 at a compound annual growth rate (CAGR) of 14.3% • Growth Driver: Rising Demand For Remote Patient Monitoring Driving The Growth Of The Market Due To Increasing Access To Timely Newborn Care • Market Trend: Strategic Collaborations Driving Expanded Access And Faster Diagnostics In Newborn Health • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Neonatal Point Of Care Testing Market?
Neonatal point of care testing is the process of performing diagnostic evaluations close to the location where newborns receive care. It provides immediate clinical information, supporting quick and informed decision-making. This method focuses on delivering timely results with accuracy, reducing the wait times and logistical challenges associated with traditional laboratory testing. The main products of neonatal point of care testing include instruments and assays. Instruments include portable analyzers, handheld readers, and bedside monitors used to test newborn health directly in care settings. It offers different types of tests, such as neonatal bilirubin testing, newborn metabolic screening, blood glucose testing, infectious disease screening, electrolyte testing, and others. It is used by several end-users, including hospitals, diagnostic centers, maternity and specialty clinics, and others.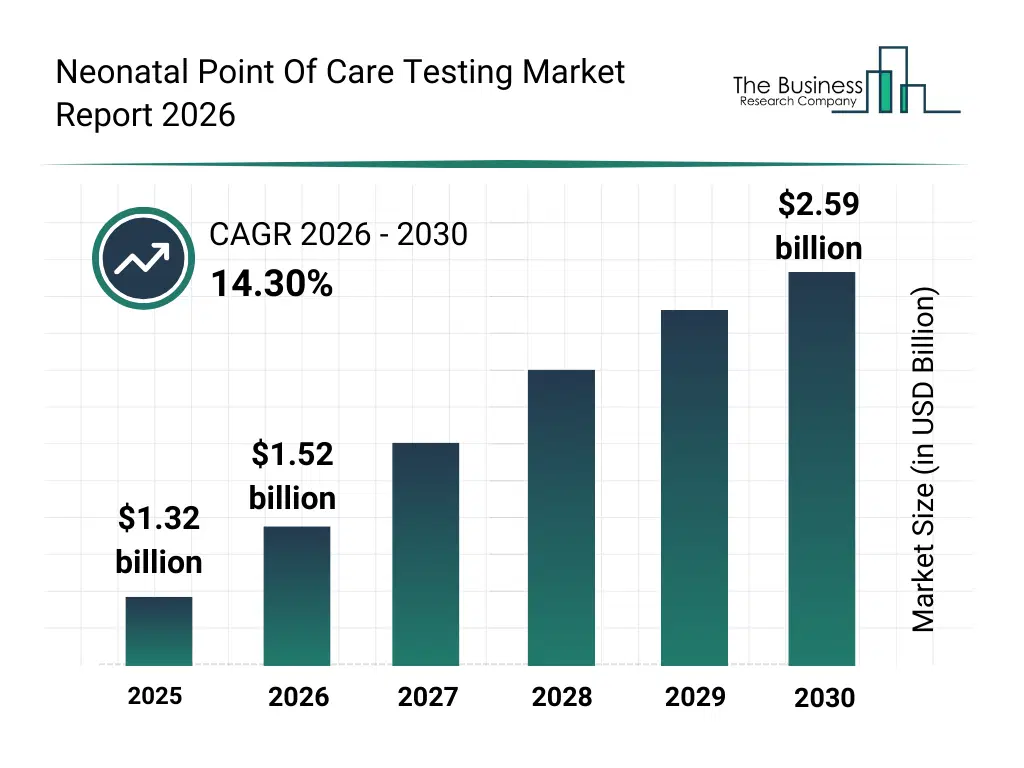
What Is The Neonatal Point Of Care Testing Market Size and Share 2026?
The neonatal point of care testing market size has grown rapidly in recent years. It will grow from $1.32 billion in 2025 to $1.52 billion in 2026 at a compound annual growth rate (CAGR) of 14.5%. The growth in the historic period can be attributed to increasing neonatal mortality awareness, adoption of rapid testing devices in hospitals, government support for neonatal care, expansion of maternity clinics, technological improvements in bedside testing.What Is The Neonatal Point Of Care Testing Market Growth Forecast?
The neonatal point of care testing market size is expected to see rapid growth in the next few years. It will grow to $2.59 billion in 2030 at a compound annual growth rate (CAGR) of 14.3%. The growth in the forecast period can be attributed to rise of AI-based diagnostic solutions, cloud integration for neonatal data management, portable and compact point-of-care devices, increasing private-public partnerships in newborn health, growth in emerging economies’ neonatal healthcare infrastructure. Major trends in the forecast period include AI-enabled neonatal diagnostics, cloud-based point-of-care platforms, portable testing devices, real-time data analysis, integration of IoT for newborn monitoring.Global Neonatal Point Of Care Testing Market Segmentation
1) By Product: Instruments, Assay 2) By Test Type: Neonatal Bilirubin Testing, Blood Glucose Testing, Infectious Disease Screening, Electrolyte Testing, Other Test Types 3) By End-Users: Hospitals, Maternity And Specialty Clinics, Other End-Users Subsegments: 1) By Instruments: Blood Gas Analyzers, Bilirubinometers, Glucose Meters 2) By Assay: Bilirubin Assays, Glucose Assays, Infectious Disease Assays, Electrolyte Assays, Blood Gas AssaysWhat Is The Driver Of The Neonatal Point Of Care Testing Market?
The increasing demand for remote patient monitoring is expected to propel the growth of the neonatal point of care testing market going forward. Remote patient monitoring involves the provision of healthcare services using technology to connect patients with healthcare professionals from a distance. The demand for remote patient monitoring is rising due to the increasing need for accessible and convenient medical care, especially for patients in remote or underserved areas. Neonatal point-of-care testing benefits remote patient monitoring by providing immediate bedside diagnostic results, allowing healthcare providers to quickly identify and manage critical conditions in newborns without relying on a central laboratory. For instance, in August 2023, according to Vivalink, a US-based healthcare technology company, a recent survey revealed that 84% of current remote patient monitoring (RPM) users intend to expand their use in 2024, 45% of healthcare providers are utilizing RPM for acute monitoring in hospital-at-home programs, and 77% believe that RPM-based care will overtake traditional inpatient hospital care within the next five years. Therefore, the increasing demand for remote patient monitoring is expected to drive the growth of the neonatal point of care testing industry.Key Players In The Global Neonatal Point Of Care Testing Market
Major companies operating in the neonatal point of care testing market are Abbott Laboratories, Siemens Healthineers AG, GE Healthcare Technologies Inc., Koninklijke Philips N.V., Roche Diagnostics International AG, Danaher Corporation, Dräger AG & Co. KGaA, Masimo Corporation, Radiometer Medical ApS, Nova Biomedical Corporation, bioMérieux SA, Sysmex Corporation, Nihon Kohden Corporation, Natus Medical Incorporated, Contec Medical Systems Co. Ltd., Edan Instruments Inc., Trivitron Healthcare, Bistos Co. Ltd., Bilimetrix S.r.l., GINEVRI SrlGlobal Neonatal Point Of Care Testing Market Trends and Insights
Major companies operating in the neonatal point of care testing market are focusing on developing strategic collaborations to allow companies to access new markets and customer bases more efficiently. Strategic collaborations are alliances between organizations that pool resources and expertise to achieve shared business goals. For instance, in January 2025, QIAGEN, a Netherlands-based provider of sample and assay technologies, partnered with Genomics England, a UK-based genomic medicine company. The partnership has enabled the launch of the Generation Study, which will sequence the genomes of 100,000 newborns in England to screen for more than 200 actionable genetic conditions. QIAGEN will supply unique clinically relevant variant information for the genes included in the point-of-care sequencing test. This data will facilitate faster interpretation and reporting of sequencing results, supporting earlier detection and treatment of rare conditions. Officially starting in October 2024, the study targets conditions that may impact around 3,000 newborns annually in the UK.What Are Latest Mergers And Acquisitions In The Neonatal Point Of Care Testing Market?
In March 2025, LaCAR MDx Technologies, a Belgium-based biotechnology company, acquired Baebies Inc.'s newborn screening division for an undisclosed amount. The acquisition enables LaCAR to enhance its offerings by expanding the variety of solutions available. It also accelerates the company’s expansion into the U.S. market, supporting increased presence and business growth. Baebies Inc. is a US-based company specializing in neonatal point of care testing.Regional Outlook
North America was the largest region in the neonatal point of care testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Neonatal Point Of Care Testing Market?
The neonatal point of care testing market consists of revenues earned by entities by providing services such as rapid diagnostic screening, blood gas and electrolyte analysis, glucose level monitoring, and sepsis detection. The market value includes the value of related goods sold by the service provider or included within the service offering. The neonatal point of care testing market also includes sales of portable diagnostic analyzers, oxygen saturation monitors, hematology analyzers, and lactate analyzers. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Neonatal Point Of Care Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.52 billion |
| Revenue Forecast In 2035 | $2.59 billion |
| Growth Rate | CAGR of 14.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Test Type, End-Users |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Abbott Laboratories, Siemens Healthineers AG, GE Healthcare Technologies Inc., Koninklijke Philips N.V., Roche Diagnostics International AG, Danaher Corporation, Dräger AG & Co. KGaA, Masimo Corporation, Radiometer Medical ApS, Nova Biomedical Corporation, bioMérieux SA, Sysmex Corporation, Nihon Kohden Corporation, Natus Medical Incorporated, Contec Medical Systems Co. Ltd., Edan Instruments Inc., Trivitron Healthcare, Bistos Co. Ltd., Bilimetrix S.r.l., GINEVRI Srl |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Neonatal Point Of Care Testing Market Report 2026 market was valued at $1.32 billion in 2025, increased to $1.52 billion in 2026, and is projected to reach $2.59 billion by 2030.
The expected CAGR for the Neonatal Point Of Care Testing Market Report 2026 market during the forecast period 2025–2030 is 14.3%.
Major growth driver of the market includes: Rising Demand For Remote Patient Monitoring Driving The Growth Of The Market Due To Increasing Access To Timely Newborn Care in the Neonatal Point Of Care Testing Market Report 2026 market. For further insights on this market,
The neonatal point of care testing market covered in this report is segmented –
1) By Product: Instruments, Assay
2) By Test Type: Neonatal Bilirubin Testing, Blood Glucose Testing, Infectious Disease Screening, Electrolyte Testing, Other Test Types
3) By End-Users: Hospitals, Maternity And Specialty Clinics, Other End-Users Subsegments:
1) By Instruments: Blood Gas Analyzers, Bilirubinometers, Glucose Meters
2) By Assay: Bilirubin Assays, Glucose Assays, Infectious Disease Assays, Electrolyte Assays, Blood Gas Assays
1) By Product: Instruments, Assay
2) By Test Type: Neonatal Bilirubin Testing, Blood Glucose Testing, Infectious Disease Screening, Electrolyte Testing, Other Test Types
3) By End-Users: Hospitals, Maternity And Specialty Clinics, Other End-Users Subsegments:
1) By Instruments: Blood Gas Analyzers, Bilirubinometers, Glucose Meters
2) By Assay: Bilirubin Assays, Glucose Assays, Infectious Disease Assays, Electrolyte Assays, Blood Gas Assays
Major trend in this market includes: Strategic Collaborations Driving Expanded Access And Faster Diagnostics In Newborn Health For further insights on this market,
Request for SampleMajor companies operating in the Neonatal Point Of Care Testing Market Report 2026 market are Major companies operating in the neonatal point of care testing market are Abbott Laboratories, Siemens Healthineers AG, GE Healthcare Technologies Inc., Koninklijke Philips N.V., Roche Diagnostics International AG, Danaher Corporation, Dräger AG & Co. KGaA, Masimo Corporation, Radiometer Medical ApS, Nova Biomedical Corporation, bioMérieux SA, Sysmex Corporation, Nihon Kohden Corporation, Natus Medical Incorporated, Contec Medical Systems Co. Ltd., Edan Instruments Inc., Trivitron Healthcare, Bistos Co. Ltd., Bilimetrix S.r.l., GINEVRI Srl
North America was the largest region in the neonatal point of care testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the neonatal point of care testing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
