
Neurology Clinical Trials Market Report 2026
Global Outlook – By Phase (Phase I, Phase II, Phase III, Phase IV), By Study Design (Interventional, Observational, Expanded Access), By Indication (Epilepsy, Parkinson’s Disease (PD), Huntington’s Disease, Stroke, Traumatic Brain Injury (TBI), Amyotrophic Lateral Sclerosis (ALS), Multiple Sclerosis, Other Indications), By End-User (Hospitals, Clinics, Research Institutes, Other End- Users) - Market Size, Trends, And Global Forecast 2026-2035
Neurology Clinical Trials Market Overview
• Neurology Clinical Trials market size has reached to $5.8 billion in 2025 • Expected to grow to $8.05 billion in 2030 at a compound annual growth rate (CAGR) of 6.7% • Growth Driver: Rising Prevalence Of Neurological Disorders Fueling The Growth Of The Market Due To Increasing Aging Population • Market Trend: Innovative Therapies Using Monoclonal Antibodies Drive Progress In Neurological Disorder Treatment • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Neurology Clinical Trials Market?
Neurology clinical trials refer to research studies conducted to evaluate the safety, efficacy, and outcomes of interventions such as drugs, therapies, or diagnostic tools for neurological disorders. These trials aim to advance treatment options for conditions affecting the brain, spinal cord, and nervous system. The main phases of neurology clinical trials are Phase I, Phase II, Phase III, and Phase IV. Phase I refers to the first stage of clinical trials involving a small group of healthy volunteers or patients to assess a drug's safety, dosage range, and side effects. In neurology, Phase I trials are essential for determining the tolerability of novel treatments targeting complex brain and nerve functions. Various study designs include interventional, observational, and expanded access, and trials focus on various indications such as epilepsy, Parkinson’s disease (PD), Huntington’s disease, stroke, traumatic brain injury (TBI), amyotrophic lateral sclerosis (ALS), muscle regeneration, and others. Key end-users of these trials include hospitals, clinics, research institutes, and others.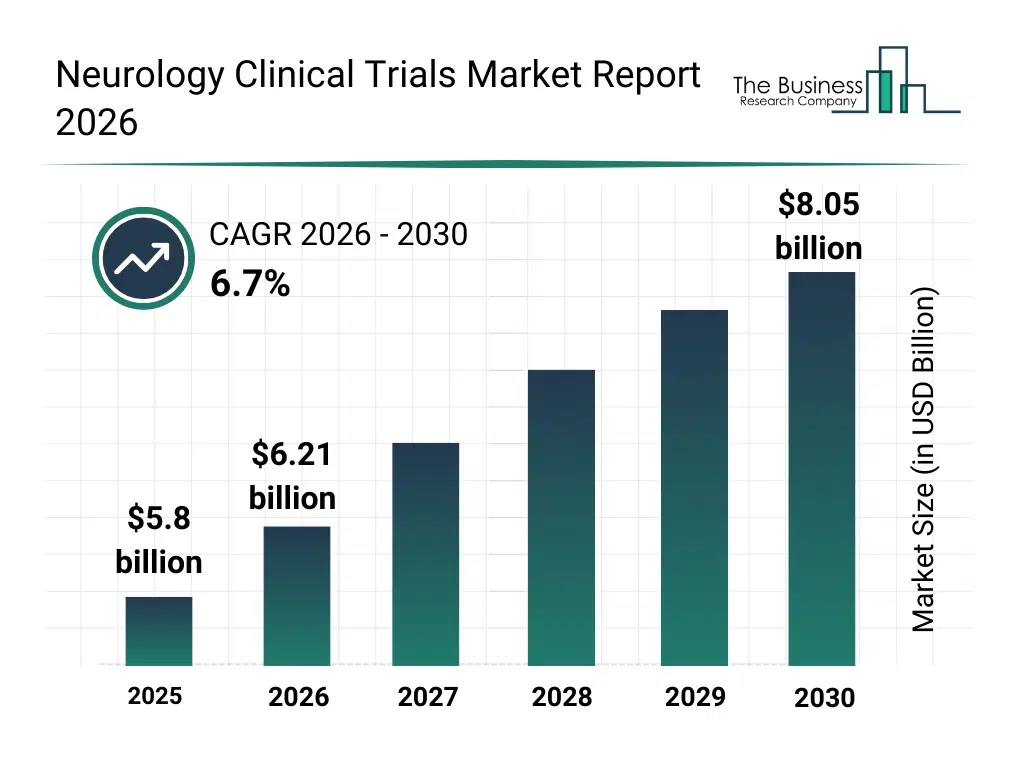
What Is The Neurology Clinical Trials Market Size and Share 2026?
The neurology clinical trials market size has grown strongly in recent years. It will grow from $5.8 billion in 2025 to $6.21 billion in 2026 at a compound annual growth rate (CAGR) of 7.0%. The growth in the historic period can be attributed to growth in neurological disease prevalence, expansion of pharmaceutical research pipelines, increased investment in neurology drug development, availability of advanced imaging tools, rising participation in clinical research.What Is The Neurology Clinical Trials Market Growth Forecast?
The neurology clinical trials market size is expected to see strong growth in the next few years. It will grow to $8.05 billion in 2030 at a compound annual growth rate (CAGR) of 6.7%. The growth in the forecast period can be attributed to increasing use of AI-driven trial analytics, rising demand for faster trial execution, expansion of patient-centric trial models, growing integration of remote monitoring technologies, increasing focus on rare neurological conditions. Major trends in the forecast period include increasing adoption of adaptive clinical trial designs, rising use of digital patient monitoring tools, growing focus on biomarker-based neurology trials, expansion of decentralized clinical trial models, enhanced emphasis on real-world evidence generation.Global Neurology Clinical Trials Market Segmentation
1) By Phase: Phase I, Phase II, Phase III, Phase IV 2) By Study Design: Interventional, Observational, Expanded Access 3) By Indication: Epilepsy, Parkinson’s Disease (PD), Huntington’s Disease, Stroke, Traumatic Brain Injury (TBI), Amyotrophic Lateral Sclerosis (ALS), Multiple Sclerosis, Other Indications 4) By End-User: Hospitals, Clinics, Research Institutes, Other End- Users Subsegments: 1) By Phase I: Drug Safety Evaluation, Pharmacokinetics Assessment, Dosage Range Finding, Tolerability Studies 2) By Phase II: Efficacy Evaluation, Dose Response Studies, Therapeutic Effectiveness, Biomarker Identification 3) By Phase III: Large Scale Efficacy Studies, Safety Monitoring, Comparative Effectiveness, Risk Benefit Analysis 4) By Phase IV: Post Marketing Surveillance, Long Term Safety Follow Up, Real World Evidence Collection, Quality Of Life AssessmentWhat Is The Driver Of The Neurology Clinical Trials Market?
The increasing prevalence of neurological disorders is expected to propel the growth of the neurology clinical trials market going forward. Neurological disorders refer to conditions that affect the brain, spinal cord, or nerves, impairing the nervous system's normal function. Neurological disorders are increasing primarily due to the aging population, as older adults are more susceptible to age-related conditions such as Alzheimer's and Parkinson's disease. Neurology clinical trials help in neurological disorders by testing new treatments, drugs, or interventions to improve diagnosis, manage symptoms, slow disease progression, and enhance patient outcomes. For instance, in September 2024, according to the Australian Institute of Health and Welfare, an Australia-based government agency, the number of Australians living with dementia is expected to more than double, increasing from 411,100 in 2023 to 849,300 by 2058. Additionally, by 2058, an estimated 315,500 men and 533,800 women will be living with dementia. Therefore, the increasing prevalence of neurological disorders is driving the growth of the neurology clinical trials industry.Key Players In The Global Neurology Clinical Trials Market
Major companies operating in the neurology clinical trials market are AbbVie Inc., Sanofi S.A., Novartis AG, Eli Lilly and Company, Teva Pharmaceutical Industries Ltd., IQVIA Holdings Inc., Biogen Inc., ICON plc., Syneos Health, Charles River Laboratories International Inc., Parexel International Corporation, CMIC Holdings Co. Ltd., Medpace Holdings Inc., PSI CRO AG., Supernus Pharmaceuticals Inc., Worldwide Clinical Trials LLC., Ergomed plc., Biotrial S.A.S., Clinilabs Drug Development Corporation, Lindus Health Ltd.Global Neurology Clinical Trials Market Trends and Insights
Major companies operating in the neurology clinical trials market, focusing on developing innovative solutions such as monoclonal antibodies to target specific neurological pathways, improve treatment efficacy, and accelerate the development of novel therapies. Monoclonal antibody refers to a laboratory-produced molecule engineered to recognize and bind to specific proteins or antigens, often used to target disease mechanisms with high precision in treatments and diagnostics. For instance, in July 2024, Eli Lilly and Company, a U.S.-based pharmaceutical company, launched Kisunla (donanemab-azbt) for the treatment of early symptomatic Alzheimer's disease. That slows cognitive and functional decline, allowing eligible patients more time to remain independent and participate in daily life. It targets and reduces brain amyloid plaques, with clinical trials showing up to a 35% slowing of disease progression versus placebo in those with less advanced disease.What Are Latest Mergers And Acquisitions In The Neurology Clinical Trials Market?
In March 2025, CLARIO, a US-based healthcare company, acquired NeuroRx for an undisclosed amount. With this acquisition, CLARIO aims to reinforce its leadership in imaging, expand its capabilities in neuroscience, and better support the development of life-changing drugs for neurological diseases. NeuroRx is a Canada-based clinical-stage pharmaceutical company involved in neurology clinical trials.Regional Outlook
North America was the largest region in the neurology clinical trials market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Neurology Clinical Trials Market?
The neurology clinical trials market consists of revenues earned by entities by providing services such as collecting and analyzing health data, monitoring patient safety, managing trial sites, regulatory consulting, and trial design and planning. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Neurology Clinical Trials Market Report 2026?
The neurology clinical trials market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the neurology clinical trials industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Neurology Clinical Trials Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $6.21 billion |
| Revenue Forecast In 2035 | $8.05 billion |
| Growth Rate | CAGR of 7.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Phase, Study Design, Indication, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | AbbVie Inc., Sanofi S.A., Novartis AG, Eli Lilly and Company, Teva Pharmaceutical Industries Ltd., IQVIA Holdings Inc., Biogen Inc., ICON plc., Syneos Health, Charles River Laboratories International Inc., Parexel International Corporation, CMIC Holdings Co. Ltd., Medpace Holdings Inc., PSI CRO AG., Supernus Pharmaceuticals Inc., Worldwide Clinical Trials LLC., Ergomed plc., Biotrial S.A.S., Clinilabs Drug Development Corporation, Lindus Health Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
