
Neutropenia Biologic Drug Treatment Market Report 2026
Global Outlook – By Drug Type (Filgrastim, Pegfilgrastim, Lenograstim, Lipegfilgrastim, Sargramostim), By Treatment Type (Antibiotic Drugs, Granulocyte-Colony-Stimulating Factor (G-CSF), Antifungal Drugs, Other Treatment Types), By Distribution Channel (Retail Pharmacies, Hospital Pharmacies, Online Pharmacies) – Market Size, Trends, Strategies, and Forecast to 2035
Neutropenia Biologic Drug Treatment Market Overview
• Neutropenia Biologic Drug Treatment market size has reached to $14.78 billion in 2025 • Expected to grow to $20.88 billion in 2030 at a compound annual growth rate (CAGR) of 7.2% • Growth Driver: Cancer Epidemic Driving Demand For Neutropenia Biologic Drugs • Market Trend: Advancing Neutropenia Treatment with Cost-Effective Biosimilars • North America was the largest region in 2025.What Is Covered Under Neutropenia Biologic Drug Treatment Market?
Neutropenia biologic drug treatment refers to drugs that are used to reduce fevers or potential infections as well as activate the bone marrow and boost neutrophil production. A lack of neutrophils, a particular type of white blood cell, causes neutropenia. The main types of drugs in neutropenia biologic drug treatment are filgrastim, pegfilgrastim, lenograstim, lipegfilgrastim, and sargramostim. Filgrastim is a recombinant form of granulocyte colony-stimulating protein that is given as an injection to boost neutrophil production after chemotherapy. The different types of treatments include antibiotic drugs, granulocyte-colony-stimulating factor (G-CSF), antifungal drugs, and others, and are distributed through various channels such as retail pharmacies, hospital pharmacies, and online pharmacies.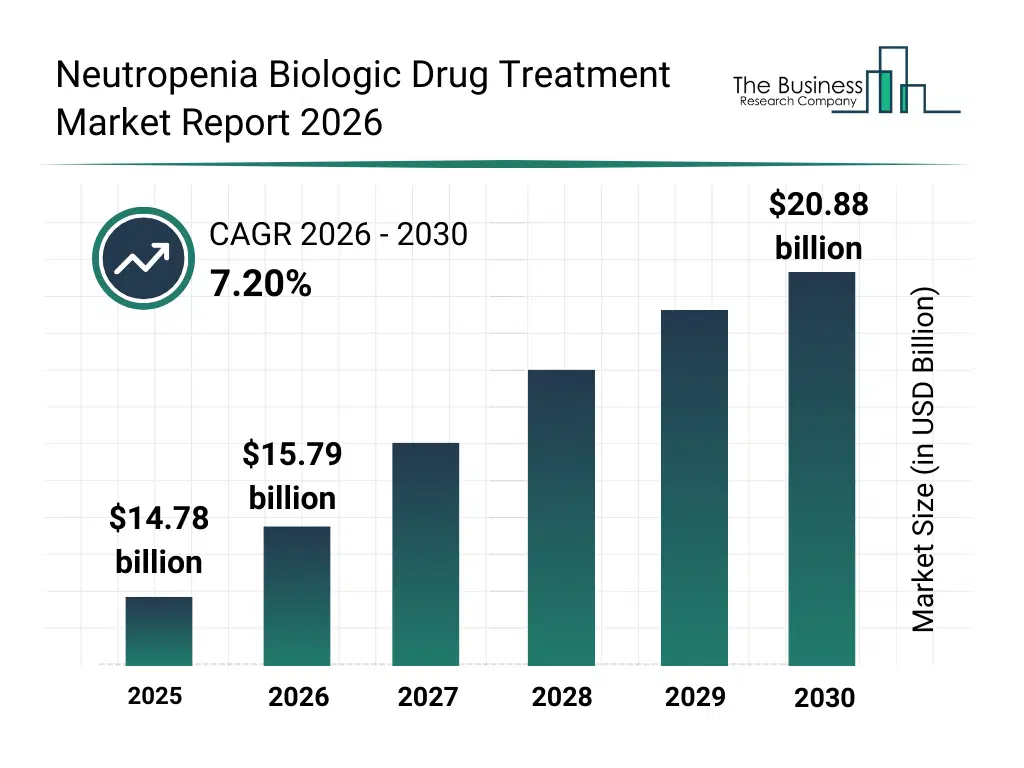
What Is The Neutropenia Biologic Drug Treatment Market Size and Share 2026?
The neutropenia biologic drug treatment market size has grown strongly in recent years. It will grow from $14.78 billion in 2025 to $15.79 billion in 2026 at a compound annual growth rate (CAGR) of 6.8%. The growth in the historic period can be attributed to growth in cancer chemotherapy usage, approval of filgrastim-based therapies, rising infection-related hospitalizations, adoption of supportive oncology care, hospital protocol standardization.What Is The Neutropenia Biologic Drug Treatment Market Growth Forecast?
The neutropenia biologic drug treatment market size is expected to see strong growth in the next few years. It will grow to $20.88 billion in 2030 at a compound annual growth rate (CAGR) of 7.2%. The growth in the forecast period can be attributed to increasing oncology patient population, biosimilar penetration growth, demand for long-acting formulations, expansion of outpatient cancer care, rising healthcare expenditure. Major trends in the forecast period include increasing use of g-csf biologics, rising adoption of long-acting biologic therapies, growth in chemotherapy-induced neutropenia management, focus on biosimilars for cost reduction, expansion of hospital and oncology pharmacy distribution.Global Neutropenia Biologic Drug Treatment Market Segmentation
1) By Drug Type: Filgrastim, Pegfilgrastim, Lenograstim, Lipegfilgrastim, Sargramostim 2) By Treatment Type: Antibiotic Drugs, Granulocyte-Colony-Stimulating Factor (G-CSF), Antifungal Drugs, Other Treatment Types 3) By Distribution Channel: Retail Pharmacies, Hospital Pharmacies, Online PharmaciesWhat Is The Driver Of The Neutropenia Biologic Drug Treatment Market?
The rise in the number of cancer cases across the globe is likely to contribute to the growth of the market for neutropenia biologic drugs. Due to a growing cancer incidence and an increase in the number of people opting for chemotherapy, there has been an increase in the number of people suffering from neutropenia. For instance, in July 2024, according to the Australian Institute of Health and Welfare, an Australia-based government agency, In 2023, an estimated 165,000 Australians were newly diagnosed with cancer, which equates to over 450 cases each day, and males represented about 55% of these diagnoses. Therefore, the rise in cancer incidence rates globally is anticipated to boost demand for the neutropenia biologic drug treatment industry over the forthcoming years.Key Players In The Global Neutropenia Biologic Drug Treatment Market
Major companies operating in the neutropenia biologic drug treatment market are Amgen Inc., Pfizer Inc., Novartis AG, Teva Pharmaceutical Industries Ltd., Sanofi S.A., GlaxoSmithKline plc, F. Hoffmann-La Roche Ltd, Merck & Co. Inc., Johnson & Johnson, Baxter International Inc., Mylan N.V., Takeda Pharmaceutical Company Limited, Bayer AG, Sun Pharmaceutical Industries Ltd., Lupin Limited, Fresenius SE & Co. KGaA, Kyowa Kirin Co. Ltd., CSL Limited, Dr. Reddy's Laboratories Ltd., Astellas Pharma Inc., Otsuka Pharmaceutical Co. Ltd.Global Neutropenia Biologic Drug Treatment Market Trends and Insights
Major companies operating in the neutropenia biologic drug treatment are developing biosimilars to offer more cost-effective alternatives to existing biologics. Biosimilars are biologic drugs designed to be nearly identical to an already approved reference biologic, with no significant differences in safety or effectiveness. For instance, in May 2023, Amneal Pharmaceuticals, a US-based pharmaceutical company, launched Fylnetra (pegfilgrastim-pbbk), a biosimilar to Neulasta, aimed at treating and preventing febrile neutropenia in patients undergoing chemotherapy. It binds to specific receptors on hematopoietic cells, stimulating the proliferation and differentiation of neutrophil precursors in the bone marrow. This action increases the production of neutrophils, which are crucial for fighting infections, particularly in patients undergoing myelosuppressive chemotherapy, thereby reducing the incidence of febrile neutropenia associated with cancer treatments.What Are Latest Mergers And Acquisitions In The Neutropenia Biologic Drug Treatment Market?
In April 2023, Assertio Holdings, Inc., a US-based specialty pharmaceutical company, acquired Spectrum Pharmaceuticals, Inc. for approximately $250 million. Through this acquisition, Assertio aims to strengthen its oncology portfolio, enhance long-term revenue growth, improve profitability, and expand its commercial capabilities across specialty therapeutics. Spectrum Pharmaceuticals, Inc. is a US-based biotechnology company focused on developing and commercializing novel and targeted oncology therapies.Regional Insights
North America was the largest region in the neutropenia biologic drug treatment market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Neutropenia Biologic Drug Treatment Market?
The neutropenia biologic drug treatment market consists of sales of cefepime, ceftazidime, piperacillin-tazobactam, and meropenem. Values in this market are 'factory gate values', that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Neutropenia Biologic Drug Treatment Market Report 2026?
The neutropenia biologic drug treatment market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the neutropenia biologic drug treatment industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Neutropenia Biologic Drug Treatment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $15.79 billion |
| Revenue Forecast In 2035 | $20.88 billion |
| Growth Rate | CAGR of 6.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Drug Type, Treatment Type, Distribution Channel |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Amgen Inc., Pfizer Inc., Novartis AG, Teva Pharmaceutical Industries Ltd., Sanofi S.A., GlaxoSmithKline plc, F. Hoffmann-La Roche Ltd, Merck & Co. Inc., Johnson & Johnson, Baxter International Inc., Mylan N.V., Takeda Pharmaceutical Company Limited, Bayer AG, Sun Pharmaceutical Industries Ltd., Lupin Limited, Fresenius SE & Co. KGaA, Kyowa Kirin Co. Ltd., CSL Limited, Dr. Reddy's Laboratories Ltd., Astellas Pharma Inc., Otsuka Pharmaceutical Co. Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
