
Next-Generation Gynecological Cancer Diagnostics Market Report 2026
Global Outlook – By Cancer Type (Cervical Cancer, Ovarian Cancer, Uterine Cancer, Vaginal Cancer, Vulvar Cancer), By Function (Therapeutic Monitoring, Companion Diagnostics, Prognostics, Cancer Screening, Risk Analysis), By Technology (Next-Generation Sequencing, qPCR And Multiplexing, Lab-On-A-Chip (LOAC) And Reverse Transcriptase-PCR (RT-PCR), Protein Microarrays, DNA Microarrays), By Application (Biomarker Development, CTC Analysis, Proteomic Analysis, Epigenetic Analysis, Genetic Analysis), By End User (Hospitals And Ambulatory Centers, Diagnostic Laboratories, Academic And Research Institutes) - Market Size, Trends, And Global Forecast 2026-2035
Next-Generation Gynecological Cancer Diagnostics Market Overview
• Next-Generation Gynecological Cancer Diagnostics market size has reached to $3.01 billion in 2025 • Expected to grow to $5.12 billion in 2030 at a compound annual growth rate (CAGR) of 11.3% • Growth Driver: Rising Adoption Of Personalized Medicine To Drive Market Growth • Market Trend: Innovative Vaccines Reshaping The Gynecological Cancer Diagnostics Market • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Next-Generation Gynecological Cancer Diagnostics Market?
Next-generation gynecological cancer diagnostics refer to advanced and innovative methods for the early detection, accurate diagnosis, and personalized treatment of any cancer that starts in a woman's reproductive organs. Gynecological cancer begins in distinct locations in a woman's pelvis, the region below the stomach, and between the hip bones. This strategy aims to enhance patient outcomes by facilitating personalized treatment plans and targeted therapies that are informed by accurate biomarker analysis. The main types of next-generation gynecological cancers are cervical cancer, ovarian cancer, uterine cancer, vaginal cancer, and vulvar cancer. Cervical cancer refers to a type of gynecological malignancy that develops in the cells of the cervix, the lower part of the uterus. The several types of functions include therapeutic monitoring, companion diagnostics, prognostics, cancer screening, and risk analysis, in which various types of technologies are involved, such as next-generation sequencing, QPCR and multiplexing, lab-on-a-chip (LOAC) and reverse transcriptase-PCR (RT-PCR), protein microarrays, and DNA microarrays. These are used for several applications, such as biomarker development, CTC analysis, proteomic analysis, epigenetic analysis, and genetic analysis, and are used by various end users, including hospitals and ambulatory centers, diagnostic laboratories, and academic and research institutes.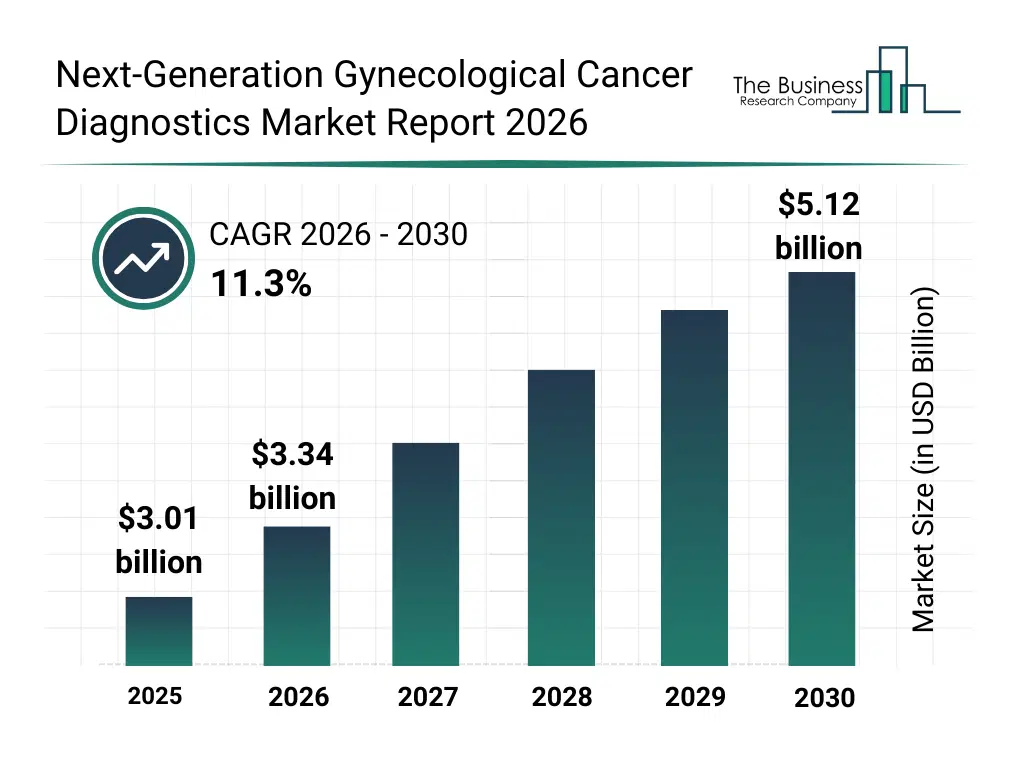
What Is The Next-Generation Gynecological Cancer Diagnostics Market Size and Share 2026?
The next-generation gynecological cancer diagnostics market size has grown rapidly in recent years. It will grow from $3.01 billion in 2025 to $3.34 billion in 2026 at a compound annual growth rate (CAGR) of 11.1%. The growth in the historic period can be attributed to increasing prevalence of gynecological cancers, rising awareness of women’s health screening, advancements in molecular diagnostics, expansion of hospital diagnostic infrastructure, government-supported cancer screening programs.What Is The Next-Generation Gynecological Cancer Diagnostics Market Growth Forecast?
The next-generation gynecological cancer diagnostics market size is expected to see rapid growth in the next few years. It will grow to $5.12 billion in 2030 at a compound annual growth rate (CAGR) of 11.3%. The growth in the forecast period can be attributed to growing demand for precision medicine, increasing adoption of AI-enabled diagnostics, expansion of genetic and epigenetic testing, rising investment in oncology R&D, improving access to advanced diagnostics in emerging markets. Major trends in the forecast period include rising adoption of biomarker-based screening, shift toward personalized gynecological oncology diagnostics, expansion of companion diagnostics in treatment planning, growing use of non-invasive and liquid biopsy techniques, increased focus on early-stage cancer detection.Global Next-Generation Gynecological Cancer Diagnostics Market Segmentation
1) By Cancer Type: Cervical Cancer, Ovarian Cancer, Uterine Cancer, Vaginal Cancer, Vulvar Cancer 2) By Function: Therapeutic Monitoring, Companion Diagnostics, Prognostics, Cancer Screening, Risk Analysis 3) By Technology: Next-Generation Sequencing, qPCR And Multiplexing, Lab-On-A-Chip (LOAC) And Reverse Transcriptase-PCR (RT-PCR), Protein Microarrays, DNA Microarrays 4) By Application: Biomarker Development, CTC Analysis, Proteomic Analysis, Epigenetic Analysis, Genetic Analysis 5) By End User: Hospitals And Ambulatory Centers, Diagnostic Laboratories, Academic And Research Institutes Subsegments: 1) By Cervical Cancer: HPV Testing, Pap Smear Testing, Liquid-Based Cytology, Biomarker Testing 2) By Ovarian Cancer: CA-125 Testing, HE4 Testing, Genetic Testing (BRCA1 Or BRCA2), Imaging Techniques 3) By Uterine Cancer: Endometrial Biopsy, Ultrasound Imaging, Genetic Testing (PMS2, MLH1), Biomarker Testing 4) By Vaginal Cancer: Pap Smear Testing, HPV Testing, Biopsy Procedures 5) By Vulvar Cancer: Visual Inspection, Biopsy, HPV Testing, Genetic TestingWhat Is The Driver Of The Next-Generation Gynecological Cancer Diagnostics Market?
The rising adoption of personalized medicine is expected to propel the growth of the next-generation gynecological cancer diagnostics market going forward. Personalized medicine is an innovative healthcare approach that tailors medical treatment and interventions to the individual genetic, molecular, and clinical characteristics of each patient, enabling more precise diagnoses, targeted therapies, and improved patient outcomes. The increasing adoption of personalized medicine is driven by advances in biomarker identification, regulatory support, and the growing availability of molecularly targeted therapies. Next-generation gynecological cancer diagnostics support clinical decision-making by enabling tumor profiling, predicting treatment response, and guiding personalized therapeutic strategies. For instance, in February 2024, according to the Personalized Medicine Coalition, a US-based nonprofit organization, the USFDA approved 16 new personalized treatments for rare disease patients in 2023, a significant increase from 6 personalized treatments in 2022. Therefore, the rising adoption of personalized medicine is driving the growth of the next-generation gynecological cancer diagnostics industry.Key Players In The Global Next-Generation Gynecological Cancer Diagnostics Market
Major companies operating in the next-generation gynecological cancer diagnostics market are F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthcare GmbH, Agena Bioscience Inc., Koninklijke Philips N.V., Becton, Dickinson and Company, GE HealthCare Technologies Inc., Laboratory Corporation Of America Holdings, Quest Diagnostics Incorporated, Agilent Technologies Inc., Hologic Inc., Illumina Inc., PerkinElmer Inc., Sysmex Corporation, Qiagen N.V., ARUP Laboratories, Almac Group, BGI Genomics Co Ltd, OPKO HEALTH Inc., BGI Genomics Co. Ltd., Myriad Genetics Inc., Fulgent Genetics Inc., Cepheid, Invitae Corporation, Centogene NVGlobal Next-Generation Gynecological Cancer Diagnostics Market Trends and Insights
Major companies operating in the gynecological cancer diagnostics market are developing new products such as cervical cancer vaccines to gain a competitive edge in the market. Vaccine is a biological preparation that provides active acquired immunity to a particular disease. For instance, in July 2023, Serum Institute of India Pvt. Ltd., an India-based biotechnology company, launched Cervavac, the country’s first domestically-made vaccine to protect against cervical cancer. Cervavac protects against the Human Papilloma Virus, the main cause of cervical cancer, and will be accessible to both men and women. Cervavac acts prophylactically, stimulating the immune system to prevent HPV infection and providing cross-protection against multiple high-risk HPV types. It offers high efficacy against HPV types 16 and 18 and proven safety. Cervavac is offered at an affordable price range of 200 to 400 rupees (about $2.50 to $5), enhancing its accessibility, especially for marginalized communities, and contributing to the goal of making cervical cancer prevention more cost-effective in India.What Are Latest Mergers And Acquisitions In The Next-Generation Gynecological Cancer Diagnostics Market?
In December 2023, Pfizer Inc., a US-based pharmaceutical company acquired Seagen Inc. for $43 billion. This acquisition aims to enhance Pfizer's oncology portfolio, focusing specifically on the development of innovative cancer therapies. The deal reflects Pfizer's commitment to expanding its capabilities in cancer treatment and leveraging Seagen's expertise and technologies in the oncology space. Seagen Inc. is a US-based company that provides antibody-drug conjugate-based targeted gynecological cancer therapeutics.Regional Outlook
North America a was the largest region in the next-generation gynecological cancer diagnostics market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Next-Generation Gynecological Cancer Diagnostics Market?
The next-generation gynecological cancer diagnostics market includes revenues earned by entities by providing blood tests, next-generation sequencing, imaging tests, biopsy and pathology, minimally invasive procedures, personalized medicine, and molecular profiling. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Next-Generation Gynecological Cancer Diagnostics Market Report 2026?
The next-generation gynecological cancer diagnostics market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the next-generation gynecological cancer diagnostics industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Next-Generation Gynecological Cancer Diagnostics Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.34 billion |
| Revenue Forecast In 2035 | $5.12 billion |
| Growth Rate | CAGR of 11.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Cancer Type, Function, Technology, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthcare GmbH, Agena Bioscience Inc., Koninklijke Philips N.V., Becton, Dickinson and Company, GE HealthCare Technologies Inc., Laboratory Corporation Of America Holdings, Quest Diagnostics Incorporated, Agilent Technologies Inc., Hologic Inc., Illumina Inc., PerkinElmer Inc., Sysmex Corporation, Qiagen N.V., ARUP Laboratories, Almac Group, BGI Genomics Co Ltd, OPKO HEALTH Inc., BGI Genomics Co. Ltd., Myriad Genetics Inc., Fulgent Genetics Inc., Cepheid, Invitae Corporation, Centogene NV |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
