
Nitinol Based Medical Device Market Report 2026
Global Outlook – By Product Type (Stents, Guidewires, Retriever Device, Catheters, Other Product Types), By Application (Cardiovascular, Urology, Dentistry, Other Applications), By End Use (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Nitinol Based Medical Device Market Overview
• Nitinol Based Medical Device market size has reached to $20.91 billion in 2025 • Expected to grow to $34.36 billion in 2030 at a compound annual growth rate (CAGR) of 10.4% • Growth Driver: Rising Prevalence of Cardiovascular Diseases Fueling the Growth Due to Aging Populations and Increased CVD Prevalence • Market Trend: Innovative Nitinol-Based Solutions Enhancing Surgical Efficiency And Patient Recovery • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Nitinol Based Medical Device Market?
Nitinol-based medical device is a healthcare device made from a nickel-titanium alloy known for its shape memory and superelastic properties. These devices are designed to adapt to body movements or temperatures, making them ideal for minimally invasive procedures. Common applications include orthopedic implants, surgical tools, and dental devices. The main product types in nitinol-based medical devices are stents, guidewires, retrieval devices, catheters, and others. Stents are small, tube-shaped implants used to keep narrowed or blocked blood vessels open. These devices are applied across various medical applications such as cardiovascular, urology, dentistry, and others. They are primarily used in hospitals, ambulatory surgical centers, specialty clinics, and others.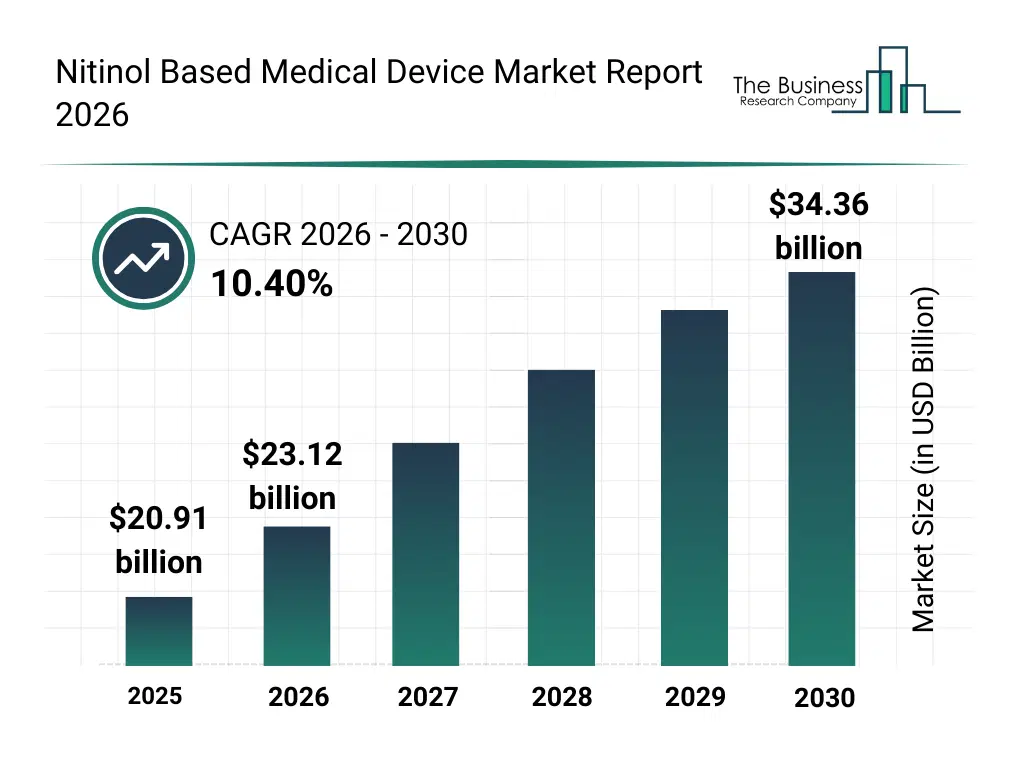
What Is The Nitinol Based Medical Device Market Size and Share 2026?
The nitinol based medical device market size has grown rapidly in recent years. It will grow from $20.91 billion in 2025 to $23.12 billion in 2026 at a compound annual growth rate (CAGR) of 10.6%. The growth in the historic period can be attributed to expansion of minimally invasive surgical procedures, availability of advanced nickel-titanium alloys, growth in cardiovascular intervention volumes, adoption of flexible implant technologies, rising use of guidewire-based interventions.What Is The Nitinol Based Medical Device Market Growth Forecast?
The nitinol based medical device market size is expected to see rapid growth in the next few years. It will grow to $34.36 billion in 2030 at a compound annual growth rate (CAGR) of 10.4%. The growth in the forecast period can be attributed to increasing demand for next-generation implantable devices, rising focus on patient-specific device designs, expansion of complex endovascular procedures, growing investments in advanced biomaterials, innovation in shape-memory alloy processing. Major trends in the forecast period include increasing adoption of nitinol-based minimally invasive devices, rising use of superelastic medical components, growing demand for shape-memory implants, expansion of cardiovascular and neurovascular applications, enhanced focus on device durability and flexibility.Global Nitinol Based Medical Device Market Segmentation
1) By Product Type: Stents, Guidewires, Retriever Device, Catheters, Other Product Types 2) By Application: Cardiovascular, Urology, Dentistry, Other Applications 3) By End Use: Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Other End Users Subsegments: 1) By Stents: Peripheral Stents, Coronary Stents, Neurovascular Stents, Other Stents 2) By Guidewires: Diagnostic Guidewires, Interventional Guidewires 3) By Retriever Devices: Thrombectomy Devices, Foreign Body Retrieval Devices 4) By Catheters: Balloon Catheters, Guiding Catheters, Microcatheters, Diagnostic Catheters 5) By Other Product Types: Orthodontic Archwires, Orthopedic Devices, Embolic Protection Devices, Other Specialized DevicesWhat Is The Driver Of The Nitinol Based Medical Device Market?
The increasing prevalence of cardiovascular diseases is expected to propel the growth of the nitinol-based medical device market going forward. Cardiovascular disease (CVD) refers to a range of heart and blood vessel disorders, including conditions such as coronary artery disease, heart attacks, heart failure, arrhythmias, and stroke. These diseases primarily involve the narrowing or blockage of blood vessels, which restricts blood flow and oxygen to the heart and other organs. The growth of cardiovascular diseases is due to aging populations, particularly in developed countries, contribute to the increase, as the risk of cardiovascular issues grows with age. Nitinol-based medical devices have become highly beneficial in the treatment and management of cardiovascular diseases due to the unique properties of nitinol, an alloy of nickel and titanium. For instance, in January 2024, according to the American Heart Association, a US-based nonprofit organization, the number of deaths caused by cardiovascular diseases reached 931,578, compared to 928,741 in 2023. Moreover, the age-adjusted death rate from cardiovascular disease also saw a modest uptick, reaching 233.3 per 100,000, marking a 4.0% increase from the previous year's rate of 224.4 per 100,000. Therefore, the increasing prevalence of vascular heart diseases is driving the cardiovascular catheters market.Key Players In The Global Nitinol Based Medical Device Market
Major companies operating in the nitinol based medical device market are Medtronic plc, Cook Medical, Teleflex Incorporated, Cordis, Endologix, Arthrex, Balton, EndoSmart GmbH, Ingpuls Medical, GEE Nitinol, Confluent Medical Technologies, Seisa Medical, Fort Wayne Metals, Norman Noble Inc., Resonetics, Vascotube, Nitinol Devices & Components (Johnson Matthey), SAES Smart Materials, Memry Corporation, Euroflex GmbH, Admedes Schuessler GmbH, ATI Specialty MaterialsGlobal Nitinol Based Medical Device Market Trends and Insights
Major companies operating in the nitinol-based medical device market are focusing on developing self-expanding nitinol stent-electrode to advance the market offering. A self-expanding nitinol stent-electrode is a hybrid medical device composed of a stent structure made from nitinol (a nickel-titanium alloy) that automatically expands to a predetermined shape upon deployment in the body and contains embedded electrodes designed for electrical stimulation or recording from biological tissues, such as nerves, blood vessels, or the brain. For instance, in September 2024, Synchron, a US-based company developing implantable neurotechnology, stated that Synchron's Stentrode BCI implant uses a self-expanding nitinol stent-electrode array delivered via the jugular vein to the superior sagittal sinus, adjacent to the motor cortex, enabling minimally invasive placement in ~20 minutes. The nitinol design allows thin construction (1.3mm lead) with low radial force to avoid venous damage, while platinum electrodes decode motor intent for digital controls.What Are Latest Mergers And Acquisitions In The Nitinol Based Medical Device Market?
In October 2023, Resonetics, LLC, a U.S. based medical technology company that provides advanced engineering, prototyping, and medical device contract manufacturing services, acquired SAES Smart Materials, Inc. (along with Memry Corporation). With this acquisition, Resonetics aimed to broaden its vertically integrated nitinol supply chain and strengthen its melt to component manufacturing capabilities, enhancing its ability to serve high-growth medical device markets. SAES Smart Materials, Inc., a U.S.-based manufacturing company, produces nitinol raw materials and semi finished nitinol forms (nickel–titanium alloys). Memry Corporation, a U.S.-based medical device manufacturing company, provides nitinol melting, complex components, and finished nitinol products for the medical device industry.Regional Outlook
North America was the largest region in the nitinol-based medical device market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Nitinol Based Medical Device Market?
The nitinol-based medical device market consists of sales of products including nitinol, orthopedic fixation devices, endoscopic components, surgical sutures, dental archwires, embolic protection devices, and spinal implants. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Nitinol Based Medical Device Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $23.12 billion |
| Revenue Forecast In 2035 | $34.36 billion |
| Growth Rate | CAGR of 10.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Application, End Use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Medtronic plc, Cook Medical, Teleflex Incorporated, Cordis, Endologix, Arthrex, Balton, EndoSmart GmbH, Ingpuls Medical, GEE Nitinol, Confluent Medical Technologies, Seisa Medical, Fort Wayne Metals, Norman Noble Inc., Resonetics, Vascotube, Nitinol Devices & Components (Johnson Matthey), SAES Smart Materials, Memry Corporation, Euroflex GmbH, Admedes Schuessler GmbH, ATI Specialty Materials |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
