
Nivolumab Market Report 2026
Global Outlook – By Type (Injection 4mL, Injection 10mL), By Route Of Administration (Intravenous, Other Routes Of Administration), By Application (Melanoma, Non-small Cell Lung Cancer (NSCLC), Malignant Pleural Mesothelioma, Classical Hodgkin Lymphoma (CHL), Squamous Cell Carcinoma Of The Head And Neck (SCCHN), Renal Cell Carcinoma (RCC), Other Applications), By End-User (Clinic, Hospital, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Nivolumab Market Overview
• Nivolumab market size has reached to $1.92 billion in 2025 • Expected to grow to $3.63 billion in 2030 at a compound annual growth rate (CAGR) of 13.8% • Growth Driver: Increased Incidence Of Non-Small Cell Lung Cancer (NSCLC) Is Ascending The Nivolumab Market Growth • Market Trend: Adoption Of Subcutaneous Immunotherapies Enhancing Convenience And Treatment Flexibility In Oncology • North America was the largest region in 2025.What Is Covered Under Nivolumab Market?
Nivolumab refers to an immunoglobulin G4 (IgG4) completely human antibody that targets programmed cell death protein 1 (PD1) and is licensed at a dose of 3 mg/kg for metastatic or unresectable melanoma, metastatic non-small cell lung cancer following platinum-based chemotherapy and metastatic renal cell carcinoma in the second-line context. It is a targeted therapy medication used to manage and treat a variety of cancers. It functions by boosting the immune system. The main types of nivolumab are injection 4ml and injection 10ml. Nivolumab 40 mg/4 mL (10 mg/mL) solution is available in a single-use vial. This formulation is used for the treatment of melanoma, non-small cell lung cancer (NSCLC) and Hodgkin lymphoma. It is administered through intravenous and other routes for melanoma, non-small cell lung cancer (NSCLC), malignant pleural mesothelioma, classical Hodgkin lymphoma (CHL), squamous cell carcinoma of the head and neck (SCCHN), renal cell carcinoma (RCC) and others. It is used by clinic, hospital and others.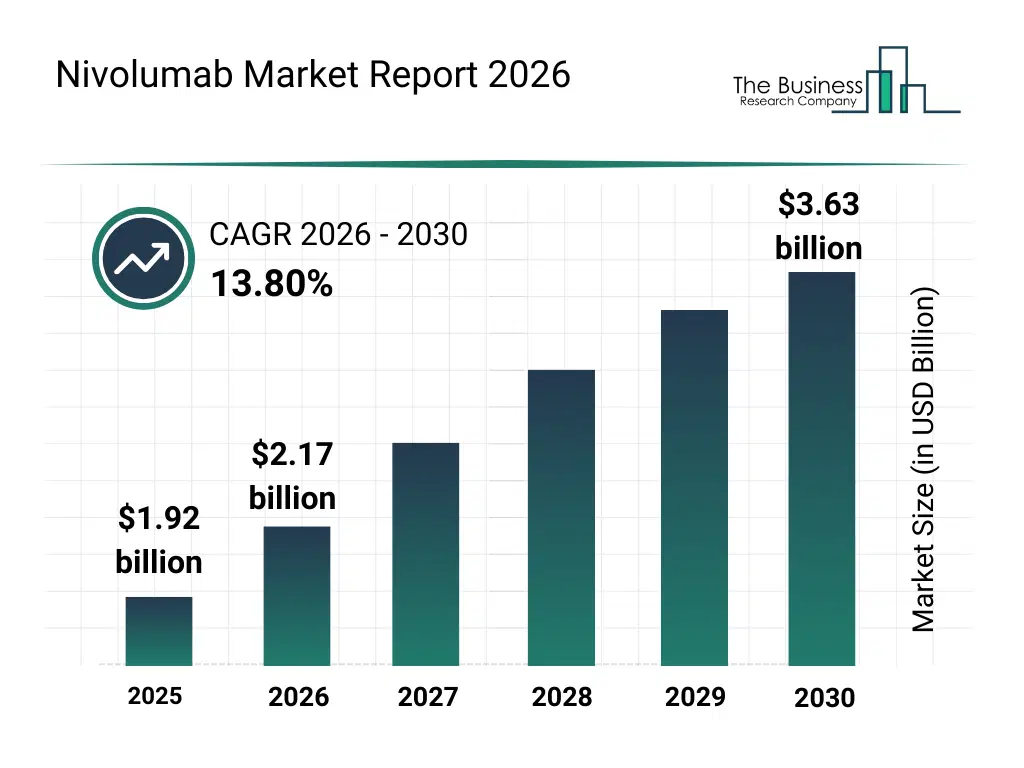
What Is The Nivolumab Market Size and Share 2026?
The nivolumab market size has grown rapidly in recent years. It will grow from $1.92 billion in 2025 to $2.17 billion in 2026 at a compound annual growth rate (CAGR) of 12.9%. The growth in the historic period can be attributed to rising prevalence of melanoma and nsclc, early approvals of nivolumab for metastatic cancers, increasing hospital infrastructure for oncology treatment, growth in oncology research and clinical trials, adoption of monoclonal antibody therapies.What Is The Nivolumab Market Growth Forecast?
The nivolumab market size is expected to see rapid growth in the next few years. It will grow to $3.63 billion in 2030 at a compound annual growth rate (CAGR) of 13.8%. The growth in the forecast period can be attributed to expansion of nivolumab indications, rising geriatric cancer population, integration with combination therapies, increasing investment in biologics manufacturing, growth in emerging markets oncology infrastructure. Major trends in the forecast period include immuno-oncology advancements, combination therapy development, personalized cancer treatment, expansion of hospital-based oncology services, increasing adoption of targeted therapies.Global Nivolumab Market Segmentation
1) By Type: Injection 4mL, Injection 10mL 2) By Route Of Administration: Intravenous, Other Routes Of Administration 3) By Application: Melanoma, Non-small Cell Lung Cancer (NSCLC), Malignant Pleural Mesothelioma, Classical Hodgkin Lymphoma (CHL), Squamous Cell Carcinoma Of The Head And Neck (SCCHN), Renal Cell Carcinoma (RCC), Other Applications 4) By End-User: Clinic, Hospital, Other End-UsersWhat Are The Drivers Of The Nivolumab Market?
The increased incidence of non-small cell lung cancer (NSCLC) is expected to propel the growth of the Nivolumab market in the coming future. Non-small cell lung cancer (NSCLC) is a kind of lung cancer that begins in the lungs' epithelial cells. Nivolumab is an approved drug that is used to treat people with advanced non-small cell lung cancer (NSCLC). It is due to its effectiveness in treating non-small cell lung cancer (NSCLC). For instance, in March 2023, according to the American Society of Clinical Oncology, a US-based professional society representing oncologists from all subspecialties, a projected 238,340 people (117,550 men and 120,790 women) are going to be diagnosed with lung cancer in the United States, while NSCLC is the most frequent kind of lung cancer, representing 81% of all lung cancer cases. Therefore, the increased incidence of non-small cell lung cancer (NSCLC) is driving the Nivolumab industry. The increasing adoption of personalized medicine is expected to drive the growth of the Nivolumab market going forward. Personalized medicine is a medical approach that tailor’s treatment and prevention strategies to an individual’s unique genetic makeup, lifestyle, and environment rather than using a one-size-fits-all method. Personalized medicine is increasing due to advances in genetic testing that allow treatments to be tailored to individual patient profiles. Nivolumab is used as personalized medicine by targeting specific immune pathways based on a patient’s tumor characteristics to deliver more tailored and effective cancer treatment. For instance, in February 2024, Personalized Medicine Coalition, a US-based non-profit organization, reported that in 2023, the FDA approved 16 new personalized treatments for patients with rare diseases, a significant increase from the six approved in 2022. Therefore, the increasing adoption of personalized medicine is driving the growth of the Nivolumab industry.Key Players In The Global Nivolumab Market
Major companies operating in the nivolumab market are Bristol-Myers Squibb Company, Ono Pharmaceutical Co. Ltd., Xbrane Biopharma AB, NeuClone Therapeutics Inc., Dr. Reddy’s Laboratories Ltd., Biocon Limited, Samsung Bioepis Co. Ltd., Celltrion Inc., Amneal Pharmaceuticals LLC, Lupin Ltd., Intas Pharmaceuticals Ltd., Hetero Biopharma Ltd., Reliance Life Sciences Pvt. Ltd., Zydus Lifesciences Limited, mAbxience, Shanghai Henlius Biotech Inc., Innovent Biologics Inc., WuXi Biologics, Lonza Group AG, Samsung BiologicsGlobal Nivolumab Market Trends and Insights
Major companies operating in the nivolumab market are focusing on advancing new formulations and expanding treatment indications to enhance patient convenience and strengthen their immuno-oncology portfolios. For instance, in December 2024, the Food and Drug Administration, a US-based federal agency, has approved Opdivo Qvantig, a subcutaneous formulation of nivolumab combined with hyaluronidase-nvhy, for use across all previously approved adult solid-tumor indications of intravenous nivolumab. This new option allows monotherapy, maintenance therapy after Opdivo plus Yervoy, or combination treatment with chemotherapy or cabozantinib. The approval is supported by the Phase 3 CHECKMATE-67T trial, which demonstrated comparable drug exposure, similar safety, and a slightly higher response rate than IV nivolumab in advanced renal cell carcinoma patients. With flexible dosing schedules and maintained efficacy, the subcutaneous version offers a more convenient alternative for patients and strengthens Bristol Myers Squibb’s immuno-oncology portfolio.Regional Insights
North America was the largest region in the nivolumab market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Nivolumab Market?
The nivolumab market consists of sales of opdivo, nivolumab injection, generic Nivolumab. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Nivolumab Market Report 2026?
The nivolumab market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the nivolumab industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Nivolumab Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.17 billion |
| Revenue Forecast In 2035 | $3.63 billion |
| Growth Rate | CAGR of 12.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Route Of Administration, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Bristol-Myers Squibb Company, Ono Pharmaceutical Co. Ltd., Xbrane Biopharma AB, NeuClone Therapeutics Inc., Dr. Reddy’s Laboratories Ltd., Biocon Limited, Samsung Bioepis Co. Ltd., Celltrion Inc., Amneal Pharmaceuticals LLC, Lupin Ltd., Intas Pharmaceuticals Ltd., Hetero Biopharma Ltd., Reliance Life Sciences Pvt. Ltd., Zydus Lifesciences Limited, mAbxience, Shanghai Henlius Biotech Inc., Innovent Biologics Inc., WuXi Biologics, Lonza Group AG, Samsung Biologics |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
