
Non-Viral Drug Delivery Systems Market Report 2026
Global Outlook – By Type Of Molecule Delivered (Biologics, Small Molecules), By Type Of Biologics Delivered (Ribonucleic Acid (RNA), Deoxyribonucleic Acid (DNA), Proteins or Peptides, Antibodies), By End User (Pharmaceutical And Biotechnology Companies, Academic And Research Institutes, Contract Development And Manufacturing Organizations (CDMOs), Hospitals And Clinics) – Market Size, Trends, Strategies, and Forecast to 2035
Non-Viral Drug Delivery Systems Market Overview
• Non-Viral Drug Delivery Systems market size has reached to $9.84 billion in 2025 • Expected to grow to $19.07 billion in 2030 at a compound annual growth rate (CAGR) of 14.1% • Growth Driver: Surge In Demand For Personalized Medicines Fueling The Growth Of The Market Due To Targeted Treatment Approaches • Market Trend: Advanced Lipid Mix Technology Accelerates RNA Therapeutic Development And Delivery • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Non-Viral Drug Delivery Systems Market?
Non-viral drug delivery systems refer to methods of delivering therapeutic agents into the body without using viral vectors. These systems use physical, chemical, or mechanical techniques such as nanoparticles, liposomes, or polymer-based carriers to safely transport drugs to targeted sites. They aim to reduce risks associated with viral delivery, such as immune reactions and insertional mutagenesis. This approach enhances treatment efficacy and patient safety across various medical conditions. The main types of molecule-delivered non-viral drug delivery systems include biologics and small molecules. Biologics refer to therapeutics derived from living organisms, including proteins, peptides, monoclonal antibodies, and nucleic acid-based treatments such as ribonucleic acid (RNA) and deoxyribonucleic acid (DNA). The types of biologics delivered are ribonucleic acid (RNA) and deoxyribonucleic acid (DNA) proteins or peptides and antibodies. The key end users are pharmaceutical and biotechnology companies, academic and research institutes, contract development and manufacturing organizations (CDMOs), and hospitals and clinics.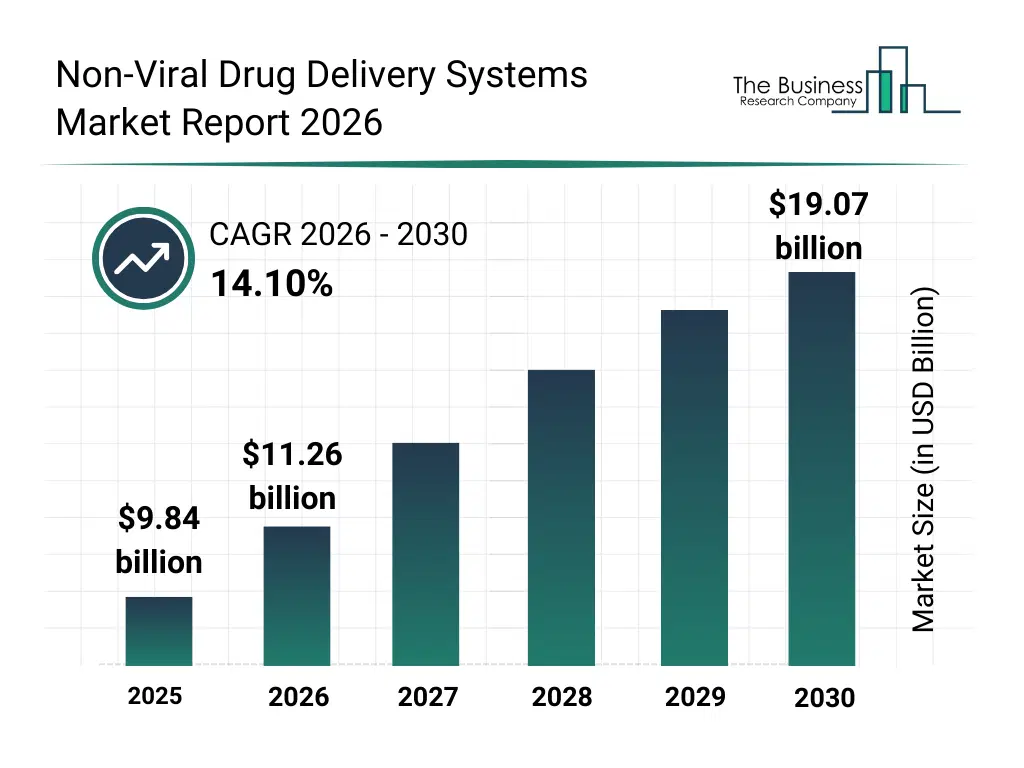
What Is The Non-Viral Drug Delivery Systems Market Size and Share 2026?
The non-viral drug delivery systems market size has grown rapidly in recent years. It will grow from $9.84 billion in 2025 to $11.26 billion in 2026 at a compound annual growth rate (CAGR) of 14.4%. The growth in the historic period can be attributed to reliance on viral delivery methods, limited awareness of non-viral techniques, emerging biologics development, initial regulatory frameworks for drug delivery, early academic research focus on nanoparticles.What Is The Non-Viral Drug Delivery Systems Market Growth Forecast?
The non-viral drug delivery systems market size is expected to see rapid growth in the next few years. It will grow to $19.07 billion in 2030 at a compound annual growth rate (CAGR) of 14.1%. The growth in the forecast period can be attributed to advancements in polymer-based carriers, growth of rna and dna therapeutics, increasing investment in personalized medicine, expansion of contract development and manufacturing organizations (cdmos), rising collaborations between pharma and academic institutions. Major trends in the forecast period include rising adoption of non-viral delivery platforms for targeted therapies, increasing use of nanoparticles and liposome-based carriers, focus on reducing immune response and enhancing safety, growth in personalized medicine and targeted therapeutics, expansion of cdmos and academic research collaborations.Global Non-Viral Drug Delivery Systems Market Segmentation
1) By Type Of Molecule Delivered: Biologics, Small Molecules 2) By Type Of Biologics Delivered: Ribonucleic Acid (RNA), Deoxyribonucleic Acid (DNA), Proteins or Peptides, Antibodies 3) By End User: Pharmaceutical And Biotechnology Companies, Academic And Research Institutes, Contract Development And Manufacturing Organizations (CDMOs), Hospitals And Clinics Subsegments: 1) By Biologics: Peptides, Proteins, Monoclonal Antibodies, RNA-based Therapeutics, DNA-based Therapies 2) By Small Molecules: Chemotherapeutics, Anti-inflammatories, Antivirals, Antibiotics, Hormonal DrugsWhat Is The Driver Of The Non-Viral Drug Delivery Systems Market?
The increasing demand for personalized medicines is expected to propel the growth of the non-viral drug delivery systems market going forward. Personalized medicine is a medical approach that tailors treatment to an individual’s genetic makeup, lifestyle, and environment. The demand for personalized medicine is rising as it leads to more effective treatments by targeting the specific causes of disease in each patient. Non-viral systems help personalized medicine by safely delivering drugs or genes to cells, reducing risks, and improving treatment precision. For instance, in February 2024, according to the Personalized Medicine Coalition, a US-based non-profit organization, in 2023, the FDA approved 16 new personalized therapies for patients with rare diseases, an increase from 6 approvals in 2022. Therefore, the increasing demand for personalized medicines is driving the growth of the non-viral drug delivery systems industry.Key Players In The Global Non-Viral Drug Delivery Systems Market
Major companies operating in the non-viral drug delivery systems market are Evonik Industries AG, Cytiva, Arcturus Therapeutics Holdings Inc., CureVac N.V., PolyPid Ltd, Ethris GmbH, Entos Pharmaceuticals Inc., Liquidia Technologies Inc., ReCode Therapeutics Inc., Enable Injections Inc., GenEdit Inc., N4 Pharma Plc, Hopewell Therapeutics Inc., Tiba Biotech LLC, Vesigen Therapeutics Inc., Carmine Therapeutics Inc., Evox Therapeutics Ltd, InnoCore Pharmaceuticals BV, Nanomerics Ltd, DelSiTech Ltd, Curapath Inc.Global Non-Viral Drug Delivery Systems Market Trends and Insights
Major companies operating in the non-viral drug delivery system market are focusing on advanced technologies such as pre-optimized ionizable lipid mix systems to streamline the development and delivery of RNA-based therapeutics. A pre-optimized ionizable lipid mix system is a formulation technology that enables the efficient encapsulation of RNA molecules into lipid nanoparticles, ensuring high stability, payload protection, and targeted delivery for gene-based drug applications. For instance, in September 2024, Cytiva, a US-based biotechnology company, launched an RNA delivery LNP kit compatible with its NanoAssemblr Ignite and Ignite+ platforms. This system includes a ready-to-use ionizable lipid mix that allows for rapid screening of RNA payloads and effective delivery validation, especially in the context of infectious disease vaccine development. It integrates smoothly with automated microfluidic platforms, supports scalable manufacturing through GMP-grade components, and includes protocols and proof-of-concept data to guide users from early discovery to clinical evaluation.What Are Latest Mergers And Acquisitions In The Non-Viral Drug Delivery Systems Market?
In September 2023, SOHM Inc., a US-based pharmaceutical company, acquired ABBIE from CGA 369 for $10 million. This acquisition strengthens SOHM’s capabilities in delivering large genetic payloads using integrase-based, non-viral tools. ABBIE is being developed for off-the-shelf gene-editing kits, with commercialization targeted for 2024 and human trials anticipated by late 2025. CGA 369 is a US-based biotechnology company specializing in non-viral, integrase-based gene-editing and genetic payload delivery systems.Regional Insights
North America was the largest region in the non-viral drug delivery systems market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Non-Viral Drug Delivery Systems Market?
The non-viral drug delivery systems market consists of sales of exosomes, cell-penetrating peptides, and nanocrystals. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Non-Viral Drug Delivery Systems Market Report 2026?
The non-viral drug delivery systems market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the non-viral drug delivery systems industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Non-Viral Drug Delivery Systems Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $11.26 billion |
| Revenue Forecast In 2035 | $19.07 billion |
| Growth Rate | CAGR of 14.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type Of Molecule Delivered, Type Of Biologics Delivered, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Evonik Industries AG, Cytiva, Arcturus Therapeutics Holdings Inc., CureVac N.V., PolyPid Ltd, Ethris GmbH, Entos Pharmaceuticals Inc., Liquidia Technologies Inc., ReCode Therapeutics Inc., Enable Injections Inc., GenEdit Inc., N4 Pharma Plc, Hopewell Therapeutics Inc., Tiba Biotech LLC, Vesigen Therapeutics Inc., Carmine Therapeutics Inc., Evox Therapeutics Ltd, InnoCore Pharmaceuticals BV, Nanomerics Ltd, DelSiTech Ltd, Curapath Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
