
Oncology Clinical Trials Market Report 2026
Global Outlook – By Phase (Phase I, Phase II, Phase III, Phase IV), By Study Design (Interventional, Observational, Expanded Access), By Therapeutic Modality (Immunotherapy, Targeted Therapy, Chemotherapy, Cell And Gene Therapy, Other Therapeutic Modalities), By Indication (Lung Cancer, Breast Cancer, Colorectal Cancer, Prostate Cancer, Blood Cancer, Other Indications), By Sponsor Type (Pharmaceutical And Biotech Companies, Academic And Research Institutes, Government Organizations, Other Sponsor Types) – Market Size, Trends, Strategies, and Forecast to 2035
Oncology Clinical Trials Market Overview
• Oncology Clinical Trials market size has reached to $14.99 billion in 2025 • Expected to grow to $20.4 billion in 2030 at a compound annual growth rate (CAGR) of 6.3% • Growth Driver: Increasing Cancer Incidence Fueling The Growth Of The Market Due To Rising Prevalence Of Unhealthy Lifestyle Habits • Market Trend: Artificial Intelligence-Driven Bioinformatics Breakthrough Speeds Patient Matching And Trial Efficiency • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Oncology Clinical Trials Market?
Oncology clinical trials are research studies that evaluate new treatments, drugs, or approaches for preventing, detecting, or managing cancer in humans. These trials aim to determine the safety, effectiveness, and potential side effects of these interventions before they become widely available. Oncology clinical trials help establish better clinical outcomes and advance cancer care. The main phases of oncology clinical trials are phase I, phase II, phase III, and phase IV. Phase I oncology clinical trials are early-stage studies designed primarily to evaluate the safety, tolerability, and optimal dosage of a new cancer treatment while assessing how the drug is absorbed, distributed, metabolized, and excreted in the body. Their study designs encompass interventional, observational, and expanded access, and their therapeutic modalities include immunotherapy, targeted therapy, chemotherapy, cell and gene therapy, and others. These trials are used for indications that include lung cancer, breast cancer, colorectal cancer, prostate cancer, blood cancer, and others, with sponsor types including pharmaceutical and biotech companies, academic and research institutes, government organizations, and others.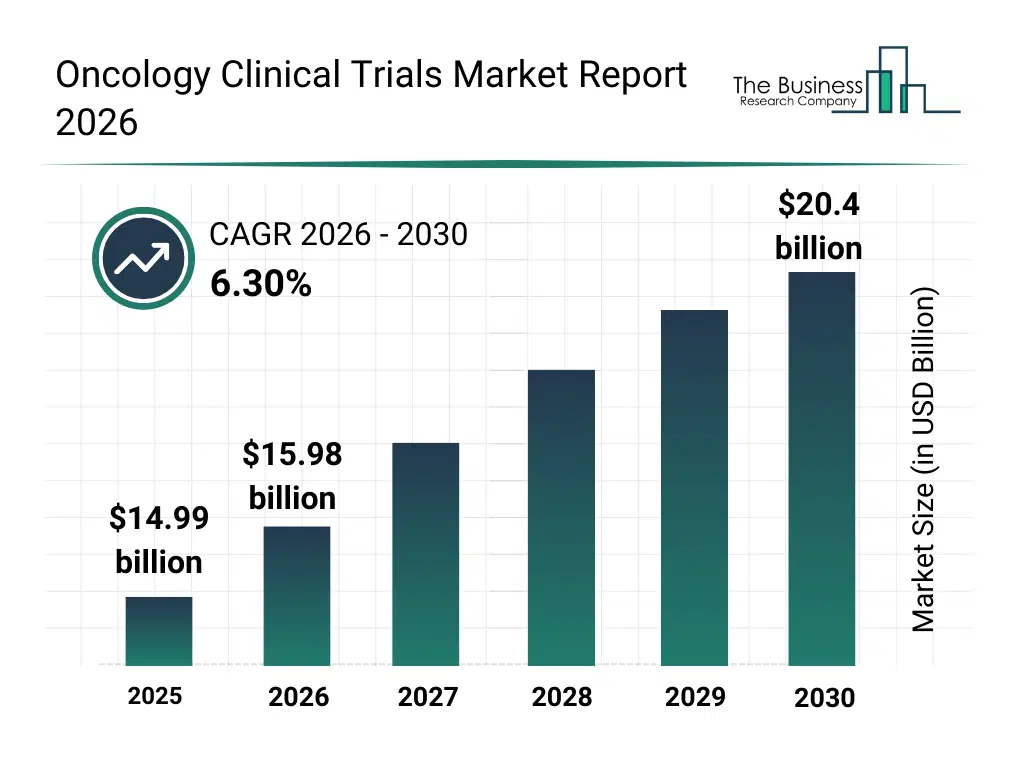
What Is The Oncology Clinical Trials Market Size and Share 2026?
The oncology clinical trials market size has grown strongly in recent years. It will grow from $14.99 billion in 2025 to $15.98 billion in 2026 at a compound annual growth rate (CAGR) of 6.6%. The growth in the historic period can be attributed to limited treatment options for cancer patients, increasing incidence of cancer globally, growing pharmaceutical and biotech R&D investments, rising number of academic clinical research centers, increasing awareness of clinical trial importance.What Is The Oncology Clinical Trials Market Growth Forecast?
The oncology clinical trials market size is expected to see strong growth in the next few years. It will grow to $20.4 billion in 2030 at a compound annual growth rate (CAGR) of 6.3%. The growth in the forecast period can be attributed to development of personalized oncology therapies, expansion of multicenter and global trials, adoption of digital patient monitoring tools, integration of AI and big data in trial design, increasing government and private funding for oncology research. Major trends in the forecast period include expansion of immunotherapy and targeted therapy clinical trials, growing focus on multicenter and randomized controlled trials, increased investment in post-marketing surveillance and long-term safety studies, rising adoption of cell and gene therapy trials, integration of biomarker and pharmacogenomics studies in oncology research.Global Oncology Clinical Trials Market Segmentation
1) By Phase: Phase I, Phase II, Phase III, Phase IV 2) By Study Design: Interventional, Observational, Expanded Access 3) By Therapeutic Modality: Immunotherapy, Targeted Therapy, Chemotherapy, Cell And Gene Therapy, Other Therapeutic Modalities 4) By Indication: Lung Cancer, Breast Cancer, Colorectal Cancer, Prostate Cancer, Blood Cancer, Other Indications 5) By Sponsor Type: Pharmaceutical And Biotech Companies, Academic And Research Institutes, Government Organizations, Other Sponsor Types Subsegments: 1) By Phase I: Safety And Dosage Studies, Pharmacokinetics And Pharmacodynamics Studies, Biomarker Identification Studies 2) By Phase II: Efficacy And Side Effects Studies, Dose-Response Relationship Studies, Therapeutic Effectiveness Studies 3) By Phase III: Large-Scale Efficacy Studies, Comparative Effectiveness Studies, Multicenter Randomized Controlled Trials 4) By Phase IV: Post Marketing Surveillance Studies, Long-Term Safety Studies, Quality Of Life StudiesWhat Is The Driver Of The Oncology Clinical Trials Market?
The increasing cancer incidence is expected to propel the growth of the oncology clinical trials market going forward. Cancer incidence refers to the number of new cancer cases occurring in a specific population during a defined period of time. The increasing cancer incidence is primarily due to unhealthy lifestyle habits, including poor diet, physical inactivity, tobacco use, and excessive alcohol consumption, elevating the risk of developing various cancers. Oncology clinical trials advance cancer treatment by evaluating new therapies and interventions, improving patient outcomes through evidence-based research. They accelerate medical innovation by testing safety and efficacy, driving progress in cancer prevention, diagnosis, and care. For instance, in October 2025, according to National Health Service (NHS), a UK-based government health agency, there were 354,820 new cancer diagnoses in 2023, an average of 972 per day, which is 8,605 more than in 2022. Therefore, the increasing cancer incidence is driving the growth of the oncology clinical trials.Key Players In The Global Oncology Clinical Trials Market
Major companies operating in the oncology clinical trials market are Pfizer Inc., Johnson & Johnson, Merck And Co. Inc., AbbVie Inc., Bayer AG, Novartis International AG, Sanofi S.A., Bristol-Myers Squibb Company, AstraZeneca plc, GSK plc, Takeda Pharmaceutical Company Limited, Eli Lilly and Company, Gilead Sciences Inc., Amgen Inc., Boehringer Ingelheim GmbH, Regeneron Pharmaceuticals Inc., Genentech Inc., BeiGene Ltd., Seagen Inc.Global Oncology Clinical Trials Market Trends and Insights
Major companies operating in the oncology clinical trials market are focusing on developing technologically advanced solutions, such as advanced bioinformatics platforms, to enhance data analysis, accelerate patient recruitment, and optimize trial outcomes. Advanced bioinformatics platforms are sophisticated software systems that use computational tools and algorithms to collect, analyze, and interpret complex biological and clinical data for research and healthcare applications. For instance, in September 2023, Deep 6 AI, a US-based healthcare technology company, launched an AI-powered genomics module aimed at speeding up enrollment in precision medicine and oncology clinical trials. The module leverages artificial intelligence and natural language processing to extract both structured and unstructured genomics data from electronic medical records, enabling real-time identification and accurate matching of patients with specific genetic markers. It provides researchers with the ability to search millions of records for more than 19,000 genes and 30,000 mutation names, significantly accelerating patient recruitment and enhancing the efficiency of clinical trial design.What Are Latest Mergers And Acquisitions In The Oncology Clinical Trials Market?
In September 2023, Coherus BioSciences Inc., a US-based biopharmaceutical company, acquired Surface Oncology Inc. for $66.9 million. With this acquisition, Coherus aims to strengthen its immuno-oncology pipeline, expand its tumor microenvironment-focused therapies, and enhance the clinical potential of its PD-1 inhibitor, toripalimab. Surface Oncology Inc. is a US-based clinical-stage immuno-oncology company conducting oncology trials for novel cancer therapies.Regional Insights
North America was the largest region in the oncology clinical trials market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Oncology Clinical Trials Market?
The oncology clinical trials market includes revenues earned by entities through designing and conducting cancer-related clinical research studies, providing patient recruitment and enrollment services, supplying oncology-specific laboratory testing and diagnostics, and managing data collection, monitoring, and regulatory compliance. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Oncology Clinical Trials Market Report 2026?
The oncology clinical trials market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the oncology clinical trials industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Oncology Clinical Trials Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $15.98 billion |
| Revenue Forecast In 2035 | $20.4 billion |
| Growth Rate | CAGR of 6.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Phase, Study Design, Therapeutic Modality, Indication, Sponsor Type |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Pfizer Inc., Johnson & Johnson, Merck And Co. Inc., AbbVie Inc., Bayer AG, Novartis International AG, Sanofi S.A., Bristol-Myers Squibb Company, AstraZeneca plc, GSK plc, Takeda Pharmaceutical Company Limited, Eli Lilly and Company, Gilead Sciences Inc., Amgen Inc., Boehringer Ingelheim GmbH, Regeneron Pharmaceuticals Inc., Genentech Inc., BeiGene Ltd., Seagen Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
