
Ornithine Transcarbamylase Deficiency (OTC Deficiency) Market Report 2026
Global Outlook – By Product Type (DTX-301, SEL-313, SHP-641, PRX-OTC), By Treatment Type (Gene Therapy, Liver Transplant, Dietary Management, Other Treatement Types), By Diagnosis (Genetic Testing, Ammonia Level Testing), By End-User (Hospitals, Specialty Clinics, Research Institutes, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Ornithine Transcarbamylase Deficiency (OTC Deficiency) Market Overview
• Ornithine Transcarbamylase Deficiency (OTC Deficiency) market size has reached to $0.89 billion in 2025 • Expected to grow to $1.23 billion in 2030 at a compound annual growth rate (CAGR) of 6.6% • Growth Driver: Rising Demand For Gene Therapies Driving The Growth Of The Market Due To Increasing Prevalence Of Genetic Disorders • Market Trend: Advancements In mRNA Therapeutics Accelerating Innovation In The Market Due To Enhanced Treatment Efficacy • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Ornithine Transcarbamylase Deficiency (OTC Deficiency) Market?
Ornithine transcarbamylase (OTC) deficiency refers to a rare genetic disorder caused by mutations in the OTC gene, leading to a deficiency of the ornithine transcarbamylase enzyme, which is essential for the urea cycle to remove ammonia from the body. The condition results in hyperammonemia, causing symptoms such as vomiting, lethargy, seizures, and coma in severe cases, particularly in newborns. The main product types in ornithine transcarbamylase (OTC) deficiency include DTX-301, SEL-313, SHP-641, and PRX-OTC. DTX-301 is an investigational AAV8-based gene therapy designed to deliver a functional OTC gene to liver cells for treating OTC deficiency. The treatment types include gene therapy, liver transplant, dietary management, and other treatment types. Diagnosis methods include genetic testing and ammonia level testing. The end users of OTC deficiency treatments include hospitals, specialty clinics, research institutes, and other end users.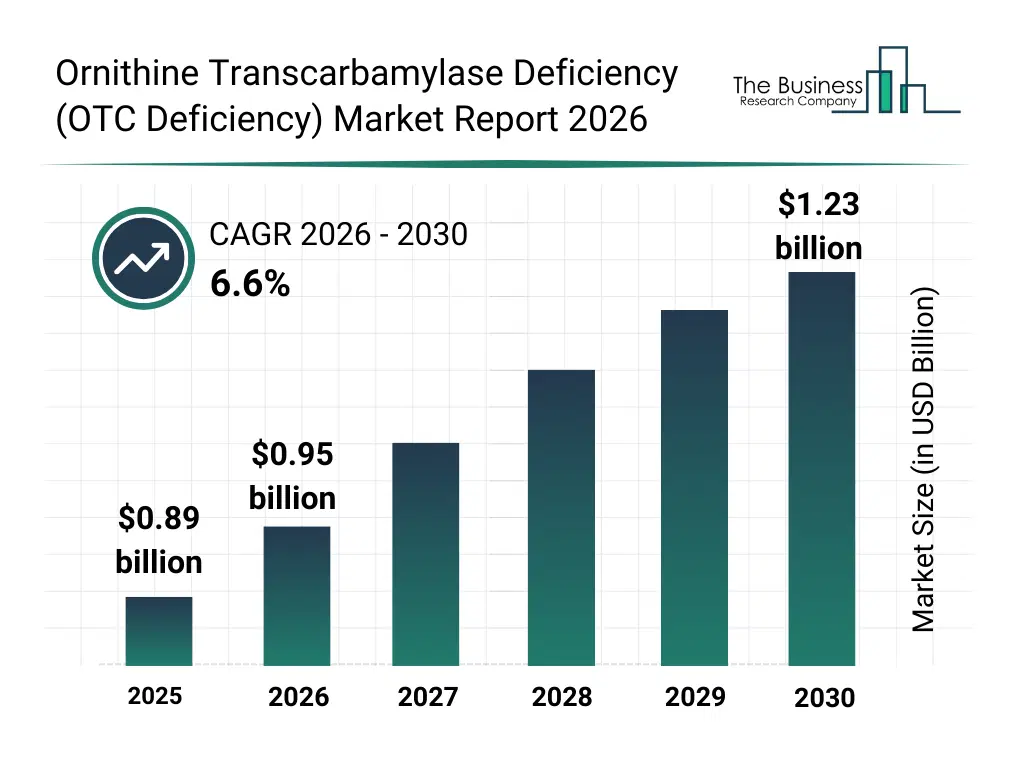
What Is The Ornithine Transcarbamylase Deficiency (OTC Deficiency) Market Size and Share 2026?
The ornithine transcarbamylase deficiency (otc deficiency) market size has grown strongly in recent years. It will grow from $0.89 billion in 2025 to $0.95 billion in 2026 at a compound annual growth rate (CAGR) of 7.0%. The growth in the historic period can be attributed to limited treatment options and high disease burden, growing awareness of otc deficiency in neonates, early adoption of liver transplant and dietary management, increasing research in rare genetic disorders, rising prevalence of hyperammonemia-related complications.What Is The Ornithine Transcarbamylase Deficiency (OTC Deficiency) Market Growth Forecast?
The ornithine transcarbamylase deficiency (otc deficiency) market size is expected to see strong growth in the next few years. It will grow to $1.23 billion in 2030 at a compound annual growth rate (CAGR) of 6.6%. The growth in the forecast period can be attributed to expanding pipeline of gene and mRNA-based therapies, increasing availability of enzyme replacement and protein engineering solutions, growing adoption of precision medicine approaches, rising investments in rare disease research, expansion of diagnostic testing and genetic screening programs. Major trends in the forecast period include rising adoption of gene therapy for otc deficiency, increasing focus on personalized and targeted treatment approaches, growing awareness about early diagnosis through genetic testing, rising demand for enzyme replacement and protein engineering therapies, increasing clinical emphasis on ammonia level monitoring and management.Global Ornithine Transcarbamylase Deficiency (OTC Deficiency) Market Segmentation
1) By Product Type: DTX-301, SEL-313, SHP-641, PRX-OTC 2) By Treatment Type: Gene Therapy, Liver Transplant, Dietary Management, Other Treatement Types 3) By Diagnosis: Genetic Testing, Ammonia Level Testing 4) By End-User: Hospitals, Specialty Clinics, Research Institutes, Other End-Users Subsegments: 1) By DTX-301: Adeno-Associated Virus (AAV)-Based Gene Therapy, Intravenous Dosing Formulation 2) By SEL-313: Immune Tolerance-Inducing Therapy, Enzyme Replacement Therapy (ERT) Combination 3) By SHP-641: Small Molecule Therapy, mRNA-Based Therapeutic Approach 4) By PRX-OTC: Recombinant Enzyme Replacement Therapy, Protein Engineering-Based TherapyWhat Is The Driver Of The Ornithine Transcarbamylase Deficiency (OTC Deficiency) Market?
The growing demand for gene therapies is expected to propel the growth of the ornithine transcarbamylase deficiency market going forward. Gene therapy refers to a medical treatment or technique that involves altering or manipulating a person's genetic material to treat or prevent diseases. The demand for gene therapies is attributed to the rising prevalence of genetic disorders, as these conditions often have limited treatment options, and gene therapy offers a promising approach to addressing the root cause rather than just managing symptoms. Ornithine transcarbamylase deficiency demonstrates the promise of gene therapies by correcting the genetic mutation that disrupts the urea cycle, allowing for targeted treatment to restore metabolic function and prevent toxic ammonia accumulation. For instance, in January 2024, according to the American Society of Gene & Cell Therapy, a US-based organization for gene and cell therapy, in Q4 2023, the number of gene therapies in Phase III increased by 10% from the previous quarter, marking the first rise since Q3 2022. Therefore, the growing demand for gene therapies is driving the growth of the ornithine transcarbamylase deficiency market.Key Players In The Global Ornithine Transcarbamylase Deficiency (OTC Deficiency) Market
Major companies operating in the ornithine transcarbamylase deficiency (otc deficiency) market are Thermo Fisher Scientific, Novartis AG, GlaxoSmithKline plc, Roche Holding AG, Eli Lilly and Company, Amgen Inc., Merck & Co. Inc, Biogen Idec, Bausch Health Companies Inc, Alexion Pharmaceuticals, Sobi, BioMarin Pharmaceutical Inc, Glenmark Pharmaceuticals Ltd., Nutricia, Vertex Pharmaceuticals Incorporated, Ultragenyx Pharmaceutical Inc, Arcturus Therapeutics, Regenxbio, OrphanPacific Inc, Ucyclyd Pharma Inc.Global Ornithine Transcarbamylase Deficiency (OTC Deficiency) Market Trends and Insights
Major companies operating in the ornithine transcarbamylase deficiency (OTC Deficiency) market are focusing on developing innovative therapies such as mRNA therapeutics to enhance treatment efficacy and address the underlying genetic cause of OTC deficiency. mRNA therapeutics are a class of treatments that use messenger RNA (mRNA) to instruct cells to produce specific proteins for therapeutic purposes. These therapies leverage the body's natural protein synthesis machinery to treat diseases, including genetic disorders, infectious diseases, and cancers. For instance, in June 2023, Arcturus Therapeutics Holdings Inc., a US-based biotechnology company, received Fast Track Designation from the U.S. Food and Drug Administration (FDA) for ARCT-810, its mRNA therapeutic candidate for ornithine transcarbamylase (OTC) deficiency. This designation is intended to accelerate the drug's development and review process, offering potential benefits such as more frequent FDA interactions and priority review. ARCT-810 is designed to enable patients with OTC deficiency, a urea cycle disorder, to produce functional OTC enzyme in their liver cells, addressing the underlying cause of the condition and potentially improving their quality of life.What Are Latest Mergers And Acquisitions In The Ornithine Transcarbamylase Deficiency (OTC Deficiency) Market?
In November 2023, Zevra Therapeutics Inc., a US-based pharmaceutical company, acquired Acer Therapeutics Inc. for $91 million. With this acquisition, Zevra Therapeutics aims to enhance its position in the rare disease sector by expanding its portfolio with valuable assets and increasing revenue potential. Acer Therapeutics Inc. is a US-based pharmaceutical company that develops therapies for ornithine transcarbamylase deficiency (OTC deficiency).Regional Outlook
North America was the largest region in the ornithine transcarbamylase deficiency (OTC Deficiency) market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Ornithine Transcarbamylase Deficiency (OTC Deficiency) Market?
The ornithine transcarbamylase deficiency market consists of revenues earned by entities by providing services such as genetic testing & diagnosis services, dietary management & nutrition services, clinical trials & research programs, medical consultation and genetic counseling. The market value includes the value of related goods sold by the service provider or included within the service offering. The ornithine transcarbamylase deficiency (OTC deficiency) market also includes sales of amino acid and protein supplements, gene therapies, nitrogen scavenging drugs, diagnostic and monitoring tests. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Ornithine Transcarbamylase Deficiency (OTC Deficiency) Market Report 2026?
The ornithine transcarbamylase deficiency (otc deficiency) market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the ornithine transcarbamylase deficiency (otc deficiency) industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Ornithine Transcarbamylase Deficiency (OTC Deficiency) Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $0.95 billion |
| Revenue Forecast In 2035 | $1.23 billion |
| Growth Rate | CAGR of 7.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Treatment Type, Diagnosis, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific, Novartis AG, GlaxoSmithKline plc, Roche Holding AG, Eli Lilly and Company, Amgen Inc., Merck & Co. Inc, Biogen Idec, Bausch Health Companies Inc, Alexion Pharmaceuticals, Sobi, BioMarin Pharmaceutical Inc, Glenmark Pharmaceuticals Ltd., Nutricia, Vertex Pharmaceuticals Incorporated, Ultragenyx Pharmaceutical Inc, Arcturus Therapeutics, Regenxbio, OrphanPacific Inc, Ucyclyd Pharma Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
