Orphan Drugs Service Market Report 2026

Orphan Drugs Service Market Report 2026
Global Outlook – By Type (Congenital Disease, Genetic Disease, Tumors And Cancer, Chronic, Other Types), By Therapeutic Area (Oncology, Metabolic Disorders, Neurological Disorders, Hematology, Infectious Diseases, Other Rare Diseases), By Application (Aldult, Children) – Market Size, Trends, Strategies, and Forecast to 2035
Orphan Drugs Service Market Overview
• Orphan Drugs Service market size has reached to $43.4 billion in 2025 • Expected to grow to $93.93 billion in 2030 at a compound annual growth rate (CAGR) of 16.6% • Growth Driver: Growing Investment In Rare Diseases Fueling The Growth Of The Market Due To Increasing Funding For Research, Development, And Patient Access • Market Trend: Innovative Therapies Aim To Address Severe Rare Conditions • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Orphan Drugs Service Market?
Orphan drugs service refers to a specialized area of healthcare and pharmaceutical support that focuses on facilitating the research, development, approval, and availability of medications specifically intended to treat rare diseases, which affect only a small and often underserved portion of the population. This service plays a critical role in ensuring that these essential medications can navigate the complex regulatory and development processes, ultimately making them accessible to patients who have limited or no alternative treatment options. The main types of orphan drug services are congenital disease, genetic disease, tumors and cancer, chronic, and others. Congenital diseases refer to medical conditions that are present from birth, either caused by genetic abnormalities, developmental issues during pregnancy, or environmental factors affecting fetal development. These are used in various therapeutic areas, including oncology, metabolic disorders, neurological disorders, hematology, infectious diseases, and others, and find applications in adults and children.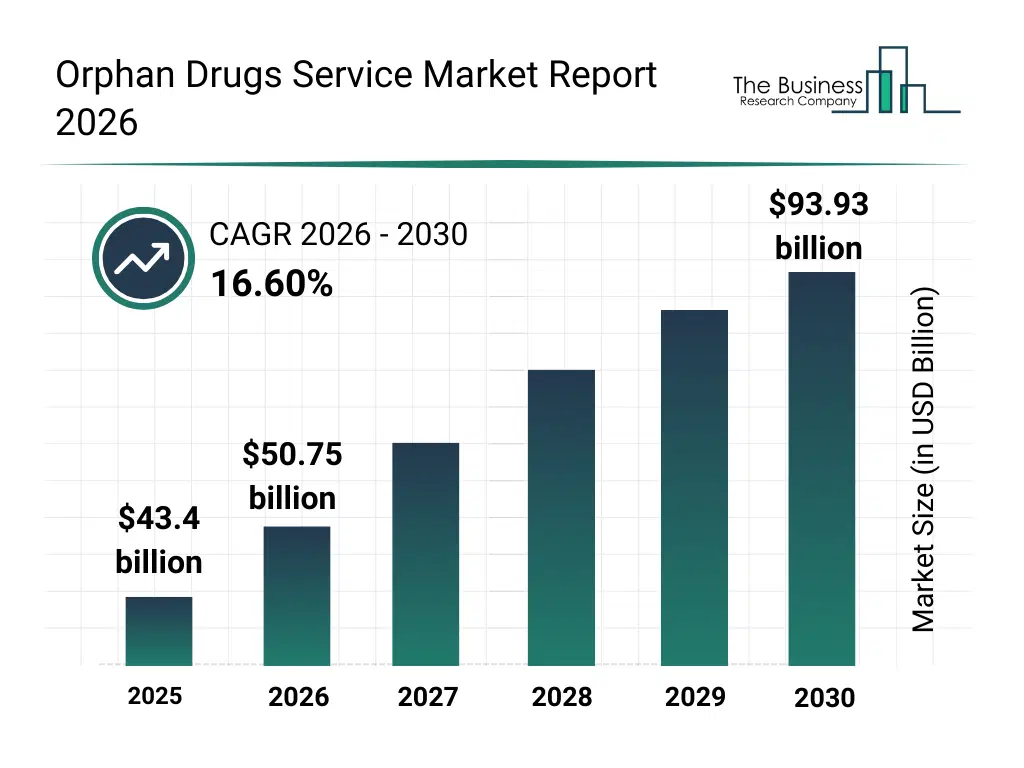
What Is The Orphan Drugs Service Market Size and Share 2026?
The orphan drugs service market size has grown rapidly in recent years. It will grow from $43.4 billion in 2025 to $50.75 billion in 2026 at a compound annual growth rate (CAGR) of 16.9%. The growth in the historic period can be attributed to growth in rare disease identification, expansion of orphan drug legislation, increasing investment in rare disease research, availability of regulatory incentives, rising role of patient advocacy groups.What Is The Orphan Drugs Service Market Growth Forecast?
The orphan drugs service market size is expected to see rapid growth in the next few years. It will grow to $93.93 billion in 2030 at a compound annual growth rate (CAGR) of 16.6%. The growth in the forecast period can be attributed to increasing demand for personalized rare disease therapies, rising adoption of advanced clinical trial technologies, expansion of global orphan drug pipelines, growing use of real-world evidence, continued regulatory support for orphan drug development. Major trends in the forecast period include increasing demand for specialized regulatory and development services, growing focus on accelerated approval pathways, rising use of data-driven rare disease research platforms, expansion of patient-centric clinical development models, enhanced collaboration between stakeholders.Global Orphan Drugs Service Market Segmentation
1) By Type: Congenital Disease, Genetic Disease, Tumors And Cancer, Chronic, Other Types 2) By Therapeutic Area: Oncology, Metabolic Disorders, Neurological Disorders, Hematology, Infectious Diseases, Other Rare Diseases 3) By Application: Aldult, Children Subsegments: 1) By Congenital Disease: Heart Defects, Cleft Lip And Palate, Spina Bifida, Down Syndrome 2) By Genetic Disease: Cystic Fibrosis, Huntington Disease, Sickle Cell Anemia, Muscular Dystrophy 3) By Tumors And Cancer: Leukemia, Lymphoma, Neuroblastoma, Sarcoma 4) By Chronic: Rare Metabolic Disorders, Rare Endocrine Disorders, Rare Cardiovascular Disorders, Rare Neurological Disorders 5) By Other Types: Rare Infectious Diseases, Rare Immunological Disorders, Rare Dermatological Conditions, Rare Ophthalmological DisordersWhat Is The Driver Of The Orphan Drugs Service Market?
The growing investment in rare diseases is expected to propel the growth of the orphan drugs service market going forward. A rare disease is a health condition that affects a small percentage of the population, typically fewer than 1 in 2,000 people. These conditions are often chronic and progressive and can be life-threatening or disabling. Investment in rare diseases is primarily due to the growing recognition of unmet medical needs and the potential for high returns through regulatory incentives such as orphan drug designation, market exclusivity, and reduced development costs. Growing investment in rare diseases enhances orphan drug services by providing increased funding for research and development, making it possible to develop therapies for previously neglected conditions. It accelerates drug availability by supporting clinical trials, regulatory approvals, and patient access, improving treatment options for those with rare diseases. For instance, in April 2024, according to Global Genes, a US-based nonprofit organization, companies developing drugs for rare diseases raised $7.1 billion in the first quarter of 2024, marking a 307% increase compared to the $1.8 billion raised in the same period in 2023. Therefore, the growing investment in rare diseases is driving the growth of the orphan drugs service industry.Key Players In The Global Orphan Drugs Service Market
Major companies operating in the orphan drugs service market are Johnson & Johnson Services Inc., F. Hoffmann-La Roche Ltd., Pfizer Inc., AbbVie Inc., Bayer AG, Sanofi S.A., Novartis AG, AstraZeneca PLC, The Bristol-Myers Squibb Company, GSK plc., Eli Lilly and Company, Takeda Pharmaceutical Company Limited, Amgen Inc., IQVIA Inc., CSL Behring GmbH, Lonza Group AG, BioMarin Pharmaceutical Inc., Fortrea Holdings Inc., Medpace Inc., Sarepta Therapeutics, Caidya Inc., Cevidra Inc.Global Orphan Drugs Service Market Trends and Insights
Major companies operating in the orphan drugs service market are focusing on obtaining regulatory approvals to expedite the development and availability of treatments for rare diseases. Regulatory approvals are official permissions from government agencies that allow a drug or treatment to be legally tested, manufactured, and marketed for patient use. For instance, in February 2023, Askbio GmbH, a US-based gene therapy company, received the European Commission orphan drug designation for AB-1003, a novel investigational gene therapy for limb-girdle muscular dystrophy. This adeno-associated virus (AAV)-based therapy aims to restore the FKRP enzyme activity in muscle cells via a one-time intravenous infusion. AB-1003 has received orphan drug designation from both the European Commission and the U.S. Food and Drug Administration (FDA), along with rare pediatric disease and fast track designations from the FDA, highlighting the urgent unmet medical need. It is currently in a Phase 1/2 clinical trial (LION-CS101), evaluating safety and efficacy in adults with genetically confirmed limb-girdle muscular dystrophy type 2I/R9 (LGMD2I/R9).What Are Latest Mergers And Acquisitions In The Orphan Drugs Service Market?
In April 2025, Norgine B.V., a Netherlands-based pharmaceutical company, acquired Theravia Pharma for an undisclosed amount. With this acquisition, Norgine aims to bolster its growth trajectory and rare disease portfolio by expanding its range of life-changing therapies for patients with high unmet medical needs in Europe and ANZ, establishing Norgine as a leading partner in rare and specialty pharmaceuticals. Theravia Pharma is a France-based pharmaceutical company specializing in rare diseases and orphan drug services.Regional Insights
North America was the largest region in the orphan drugs service market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Orphan Drugs Service Market?
The orphan drugs service market includes revenues earned by entities through clinical trial management, pharmacovigilance and safety monitoring, distribution and supply chain services, patient support and access programs, and data management and biostatistics services. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Orphan Drugs Service Market Report 2026?
The orphan drugs service market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the orphan drugs service industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Orphan Drugs Service Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $50.75 billion |
| Revenue Forecast In 2035 | $93.93 billion |
| Growth Rate | CAGR of 16.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Therapeutic Area, Application |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Johnson & Johnson Services Inc., F. Hoffmann-La Roche Ltd., Pfizer Inc., AbbVie Inc., Bayer AG, Sanofi S.A., Novartis AG, AstraZeneca PLC, The Bristol-Myers Squibb Company, GSK plc., Eli Lilly and Company, Takeda Pharmaceutical Company Limited, Amgen Inc., IQVIA Inc., CSL Behring GmbH, Lonza Group AG, BioMarin Pharmaceutical Inc., Fortrea Holdings Inc., Medpace Inc., Sarepta Therapeutics, Caidya Inc., Cevidra Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Orphan Drugs Service market was valued at $43.4 billion in 2025, increased to $50.75 billion in 2026, and is projected to reach $93.93 billion by 2030.
The global Orphan Drugs Service market is expected to grow at a CAGR of 16.6% from 2026 to 2035 to reach $93.93 billion by 2035.
Some Key Players in the Orphan Drugs Service market Include, Johnson & Johnson Services Inc., F. Hoffmann-La Roche Ltd., Pfizer Inc., AbbVie Inc., Bayer AG, Sanofi S.A., Novartis AG, AstraZeneca PLC, The Bristol-Myers Squibb Company, GSK plc., Eli Lilly and Company, Takeda Pharmaceutical Company Limited, Amgen Inc., IQVIA Inc., CSL Behring GmbH, Lonza Group AG, BioMarin Pharmaceutical Inc., Fortrea Holdings Inc., Medpace Inc., Sarepta Therapeutics, Caidya Inc., Cevidra Inc. .
Major trend in this market includes: Innovative Therapies Aim To Address Severe Rare Conditions. For further insights on this market.
Request for SampleNorth America was the largest region in the orphan drugs service market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the orphan drugs service market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
