
Orthopedic Regenerative Surgical Products Market Report 2026
Global Outlook – By Product (Allograft, Synthetic, Cell-based, Viscosupplements), By Application (Orthopedic Pain Management, Trauma Repair, Cartilage and Tendon Repair), By End User (Hospitals, Ambulatory Surgical Centers, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Orthopedic Regenerative Surgical Products Market Overview
• Orthopedic Regenerative Surgical Products market size has reached to $4.88 billion in 2025 • Expected to grow to $7.39 billion in 2030 at a compound annual growth rate (CAGR) of 8.6% • Growth Driver: Elevating Prevalence Of Orthopedic Diseases Fuels Growth In Orthopedic Regenerative Surgical Products Market • Market Trend: Increasing Focus On Product Innovations To Provide Reliable Services To Their Customers • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Orthopedic Regenerative Surgical Products Market?
Orthopedic regenerative surgical products are used for the treatment of joint pains, gout, articular defects, fibromyalgia, and osteoarthritis, as well as trauma and joint replacement, joint problems, regenerative orthopedic medicine using platelet-rich plasma (PRP) which is frequently the nonsurgical treatment of choice to assist patients in healing and repairing many musculoskeletal (orthopedic) injuries such as joint, tendon, ligament, and muscular injuries. The orthopedic regenerative surgical products are curing many other musculoskeletal injuries. The main type of orthopedic regenerative surgical products is allograft, synthetic, cell-based, and viscosupplements. Allograft is a tissue transplant between genetically distinct individuals of the same species. An allograft is not the same as an autograft, which uses tissue from the same person's body and is thus genetically identical. Anterior tibialis tendon, frozen femoral head, freeze-dried bone chips, DBM putty, acellular dermis, and amniotic membrane are examples of human allografts. These are used by hospitals, ambulatory surgical centers, and others, in the treatment of orthopedic pain management, trauma repair, cartilage and tendon repair, and joint reconstruction.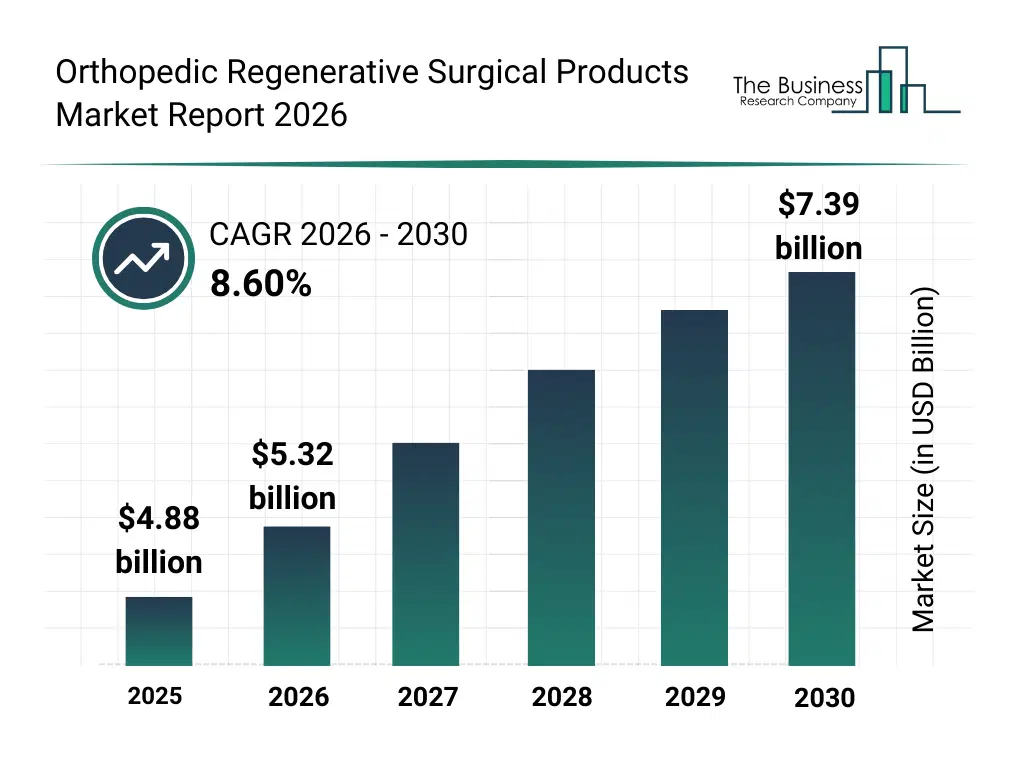
What Is The Orthopedic Regenerative Surgical Products Market Size and Share 2026?
The orthopedic regenerative surgical products market size has grown strongly in recent years. It will grow from $4.88 billion in 2025 to $5.32 billion in 2026 at a compound annual growth rate (CAGR) of 9.1%. The growth in the historic period can be attributed to increasing prevalence of musculoskeletal disorders, rising demand for joint preservation treatments, expansion of sports injury management, growing acceptance of regenerative therapies, improvements in biologic processing techniques.What Is The Orthopedic Regenerative Surgical Products Market Growth Forecast?
The orthopedic regenerative surgical products market size is expected to see strong growth in the next few years. It will grow to $7.39 billion in 2030 at a compound annual growth rate (CAGR) of 8.6%. The growth in the forecast period can be attributed to increasing adoption of stem cell-based treatments, rising focus on non-surgical orthopedic interventions, expansion of regenerative medicine clinics, growing investments in biologics research, increasing demand for personalized orthopedic therapies. Major trends in the forecast period include increasing adoption of platelet-rich plasma therapies, rising demand for cell-based regenerative solutions, growing use of synthetic bone grafts, expansion of minimally invasive regenerative procedures, enhanced focus on tissue repair outcomes.Global Orthopedic Regenerative Surgical Products Market Segmentation
1) By Product: Allograft, Synthetic, Cell-based, Viscosupplements 2) By Application: Orthopedic Pain Management, Trauma Repair, Cartilage and Tendon Repair 3) By End User: Hospitals, Ambulatory Surgical Centers, Other End Users Subsegments: 1) By Allograft: Bone Allograft, Soft Tissue Allograft, Osteoinductive Allograft, Osteoconductive Allograft 2) By Synthetic: Synthetic Bone Grafts, Synthetic Soft Tissue Grafts, Composite Synthetic Grafts 3) By Cell-based: Stem Cell Therapies, Platelet-Rich Plasma (PRP), Mesenchymal Stem Cells (MSCs) 4) By Viscosupplements: Hyaluronic Acid Injections, Sodium HyaluronateWhat Is The Driver Of The Orthopedic Regenerative Surgical Products Market?
The rising prevalence of orthopedic diseases across the globe is likely to contribute to the growth of the orthopedic regenerative surgical products market. The orthopedic regenerative surgical products contribute to the prevention of orthopedic problems such as fractures, sprains, and strains, as well as lifelong conditions associated with ongoing functional limitations and disability. For instance, in June 2024, according to a report published by the Australian Bureau of Statistics, an Australia-based statistical agency, musculoskeletal conditions accounted for 12.8% of total disease burden (DALY), 23.1% of non-fatal burden (YLD), and 0.8% of fatal burden (YLL) in 2023. Hence, the rising prevalence of orthopedic diseases drives the market for orthopedic regenerative surgical products.Key Players In The Global Orthopedic Regenerative Surgical Products Market
Major companies operating in the orthopedic regenerative surgical products market are Baxter International Inc., Zimmer Biomet Holdings Inc., Stryker Corporation, Smith&Nephew plc, Allosource, Anika Therapeutics Inc., Vericel Corporation, Amniox Medical Inc., MIMEDX Group Inc., Aptissen S.A, Arthrex Inc., Bioventus LLC, Conmed Corporation, DePuy Synthes Inc., Exactech Inc., Globus Medical Inc., Integra LifeSciences Corporation, Johnson & Johnson Medical Devices group, Kuros Biosciences AG, Medtronic plc, NuVasive Inc., Orthofix Medical Inc., Osiris Therapeutics Inc., RTI Surgical Inc., Terumo BCT Inc., Tissue Regenix Group plc, Wright Medical Group N.V., Xtant Medical Holdings Inc., Camber Spine Technologies, Lima Corporate, Life Spine Inc., CollPlant Biotechnologies Ltd.Global Orthopedic Regenerative Surgical Products Market Trends and Insights
Major companies operating in the orthopedic regenerative surgical products market are focusing on innovative products such as non-permanent ossiofiber compression staple to drive revenues in their market. The non-permanent ossiofiber compression staple is a medical device used in orthopedic and musculoskeletal surgeries for fracture fixation and bone fusion which is designed to provide temporary stability and compression to promote the healing and fusion of bones. For instance, in February 2023, Ossio Inc. a US-based company that specializes in creating innovative products for orthopedic and musculoskeletal applications launched non-permanent ossiofiber compression staple. Ossiofiber combines mechanical strength and spontaneous bone regeneration, there is an alternative to traditional resorbable and allograft implants and permanent metal hardware. By using Ossiofiber Compression Staples, the company is able to offer solutions for a variety of midfoot and hindfoot procedures, such as flatfoot corrective procedures, Lapidus fusions, and midfoot fusions.What Are Latest Mergers And Acquisitions In The Orthopedic Regenerative Surgical Products Market?
In October 2023, Xtant Medical Holdings Inc., a US-based medical technology company, acquired the nanOss production operations from RTI Surgical Inc. for an undisclosed amount. With this acquisition, Xtant aimed to bolster its manufacturing capabilities and capacity for synthetic bone grafts, gain full control over the nanOss product line to revive and scale it, and improve cash flows by internalizing production. RTI Surgical Inc.is a US-based ortho biologics company that specialized in producing and distributing surgical implants, allografts, and synthetic biomaterials, including the nanOss synthetic bone-graft products.Regional Outlook
North America was the largest region in the orthopedic regenerative surgical products market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Orthopedic Regenerative Surgical Products Market?
The orthopedic regenerative surgical products market consists of sales of allograft wedges, allowrap DS, BIO⁴, DBM, hydroset, imbibe, nerve repair, and prochondrix CR.Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Orthopedic Regenerative Surgical Products Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $5.32 billion |
| Revenue Forecast In 2035 | $7.39 billion |
| Growth Rate | CAGR of 9.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Baxter International Inc., Zimmer Biomet Holdings Inc., Stryker Corporation, Smith&Nephew plc, Allosource, Anika Therapeutics Inc., Vericel Corporation, Amniox Medical Inc., MIMEDX Group Inc., Aptissen S.A, Arthrex Inc., Bioventus LLC, Conmed Corporation, DePuy Synthes Inc., Exactech Inc., Globus Medical Inc., Integra LifeSciences Corporation, Johnson & Johnson Medical Devices group, Kuros Biosciences AG, Medtronic plc, NuVasive Inc., Orthofix Medical Inc., Osiris Therapeutics Inc., RTI Surgical Inc., Terumo BCT Inc., Tissue Regenix Group plc, Wright Medical Group N.V., Xtant Medical Holdings Inc., Camber Spine Technologies, Lima Corporate, Life Spine Inc., CollPlant Biotechnologies Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
