Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market Report 2026

Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market Report 2026
Global Outlook – By Treatment Type (Complement C5 Inhibitors, Anti-Complement Component 5a Receptor (C5aR) Monoclonal Antibodies, Alternative Pathway Inhibitors, Hematopoietic Stem Cell Transplantation, Other Therapies), By Route Of Administration (Intravenous, Subcutaneous, Oral), By Market Availability (Approved, In Pipeline, Investigational), By End-Use (Hospitals, Specialty Clinics, Homecare Settings, Other End-Users ) - Market Size, Trends, And Global Forecast 2026-2035
Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market Overview
• Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment market size has reached to $6.07 billion in 2025 • Expected to grow to $11.32 billion in 2030 at a compound annual growth rate (CAGR) of 13.2% • Growth Driver: The Rising Incidence Of Hematologic Disorders Accelerating Growth In The Market Due To Increasing Cases And Enhanced Diagnostic Capabilities • Market Trend: Advancements In Hemoglobinuria Management With The Approval Of The First Oral Monotherapy For Improved Patient Care • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market?
Paroxysmal nocturnal hemoglobinuria (PNH) treatment refers to medical therapies to manage PNH, a rare blood disorder characterized by hemolysis (destruction of red blood cells), blood clots, and bone marrow dysfunction. Paroxysmal nocturnal hemoglobinuria (PNH) is a rare, acquired hematologic disorder characterized by the destruction of red blood cells due to complement-mediated hemolysis, bone marrow dysfunction leading to cytopenias, and an increased risk of thrombosis. Paroxysmal nocturnal hemoglobinuria (PNH) management focuses on inhibiting complement activation to prevent hemolysis, addressing bone marrow failure, and mitigating thrombotic complications to improve patient outcomes and quality of life. The main treatment types for paroxysmal nocturnal hemoglobinuria (PNH) treatment are complement C5 inhibitors, anti-complement component 5a receptor (C5aR) monoclonal antibodies, alternative pathway inhibitors, hematopoietic stem cell transplantation, and others. Complement C5 inhibitors are a class of targeted therapies that block the cleavage of complement component 5 (C5) in the immune system's complement cascade, preventing the formation of membrane attack complexes and thereby reducing complement-mediated hemolysis and inflammation in conditions such as Paroxysmal Nocturnal Hemoglobinuria (PNH) and other complement-driven diseases. These are administered through routes of administration including intravenous, subcutaneous, and oral. The drugs used are having market availability as approved, in the pipeline, and investigational for the end uses such as hospitals, specialty clinics, homecare settings, and others.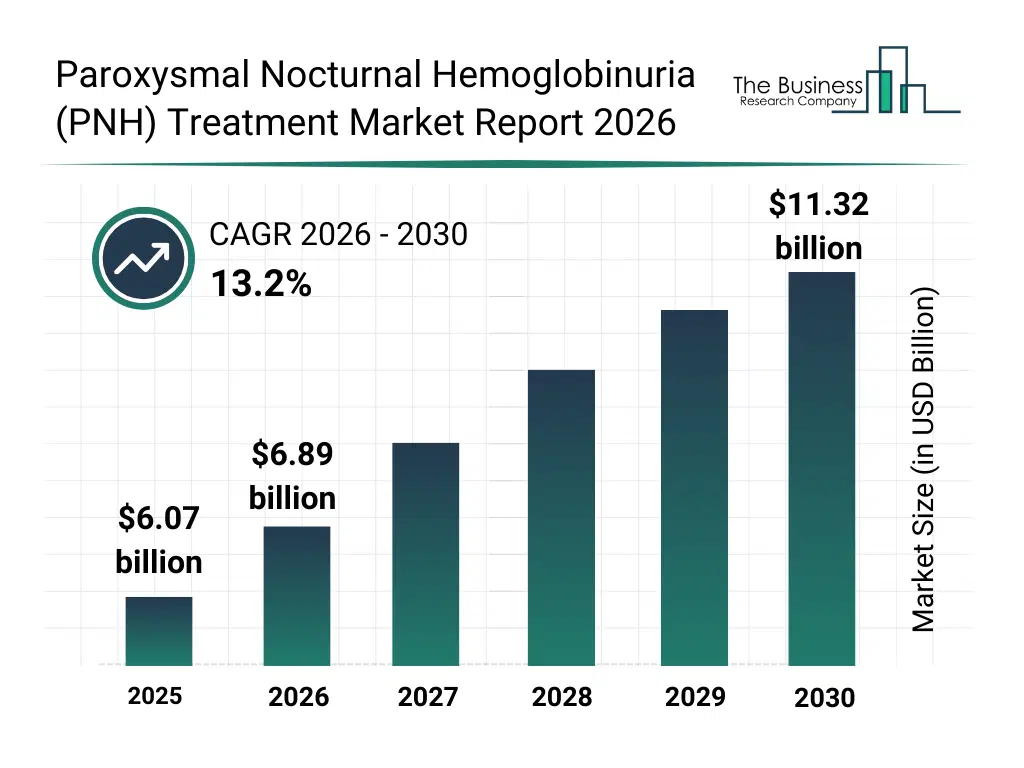
What Is The Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market Size and Share 2026?
The paroxysmal nocturnal hemoglobinuria (pnh) treatment market size has grown rapidly in recent years. It will grow from $6.07 billion in 2025 to $6.89 billion in 2026 at a compound annual growth rate (CAGR) of 13.4%. The growth in the historic period can be attributed to limited availability of effective targeted therapies, reliance on supportive care and transfusions, delayed diagnosis due to disease rarity, high risk of life-threatening thrombosis, lack of standardized long-term treatment protocols.What Is The Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market Growth Forecast?
The paroxysmal nocturnal hemoglobinuria (pnh) treatment market size is expected to see rapid growth in the next few years. It will grow to $11.32 billion in 2030 at a compound annual growth rate (CAGR) of 13.2%. The growth in the forecast period can be attributed to development of next-generation complement inhibitors, expanding clinical pipeline for oral and subcutaneous therapies, growing focus on orphan disease drug innovation, increasing diagnosis rates through advanced testing, improved reimbursement coverage for rare hematologic disorders. Major trends in the forecast period include rising adoption of complement c5 inhibitor therapies, increasing shift toward long-acting and subcutaneous treatments, growth of alternative pathway inhibitor development, expansion of combination and dual complement inhibition strategies, improved management of thrombotic and bone marrow complications.Global Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market Segmentation
1) By Treatment Type: Complement C5 Inhibitors, Anti-Complement Component 5a Receptor (C5aR) Monoclonal Antibodies, Alternative Pathway Inhibitors, Hematopoietic Stem Cell Transplantation, Other Therapies 2) By Route Of Administration: Intravenous, Subcutaneous, Oral 3) By Market Availability: Approved, In Pipeline, Investigational 4) By End-Use: Hospitals, Specialty Clinics, Homecare Settings, Other End-Users Subsegments: 1) By Complement: C5 InhibitorsEculizumab (Soliris), Ravulizumab (Ultomiris), Emerging C5 Inhibitors, Biosimilars Of C5 Inhibitors, 2) By Anti-Complement Component 5a Receptor Monoclonal Antibodies: Avacopan (Tavneos), Other Investigational C5aR mAbs, Monotherapy vs Combination Therapy, Clinical Trial Phase (Phase I, II, III) 3) By Alternative Pathway Inhibitors: C3 Inhibitors, Factor B Inhibitors, Factor D Inhibitors, Targeting Complement Amplification, Combination Therapies And Dual Inhibition 4) By Hematopoietic Stem Cell Transplantation (HSCT): Autologous HSCT vs. Allogeneic HSCT, Pre-Transplant Conditioning Therapies, Post-Transplant Immunosuppressive Therapies 5) By Other Therapies: Immunosuppressive Therapy, Supportive Care, Anticoagulation Therapy, Gene TherapyWhat Is The Driver Of The Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market?
The increasing incidence of hematologic disorders is expected to propel the growth of the paroxysmal nocturnal hemoglobinuria (PNH) treatment market going forward. Hematologic disorders refer to a broad category of medical conditions affecting the blood and blood-forming organs, including abnormalities in red blood cells, white blood cells, platelets, bone marrow, and the coagulation system, which can lead to anemia, clotting disorders, or malignancies. The increasing incidence of hematologic disorders is driven by factors such as aging populations and genetic predispositions, with genetic mutations playing a critical role as they can trigger abnormal blood cell production, leading to various hematologic conditions. Paroxysmal nocturnal hemoglobinuria (PNH) treatment positively manages hematologic disorders by offering targeted therapies that mitigate complement-mediated hemolysis, reduce thrombotic complications, and improve patient survival outcomes. For instance, in July 2024, according to Cancer Research UK, a UK-based charity organization, the average number of new leukemia (a hematologic disorder) cases in the UK is expected to increase from approximately 12,400 in 2023-2025 to around 14,500 by 2038-2040. Therefore, the increasing prevalence of hematologic disorders drives the growth of the paroxysmal nocturnal hemoglobinuria (PNH) treatment industry.Key Players In The Global Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market
Major companies operating in the paroxysmal nocturnal hemoglobinuria (pnh) treatment market are F. Hoffmann-La Roche Ltd, Novartis AG, Mayo Clinic, Cleveland Clinic Foundation, Johns Hopkins Medicine, Memorial Sloan Kettering Cancer Center, Stanford Health Care, University of California San Francisco (UCSF) Health, Emory Healthcare, Cedars-Sinai Medical Center, Duke University Health System, Mount Sinai Health System, Dana-Farber Cancer Institute, Swedish Orphan Biovitrum, Alnylam Pharmaceuticals, BioCryst Pharmaceuticals, Apellis Pharmaceuticals, Omeros Corporation, Takeda Pharmaceutical Company, Sanofi S A, Pfizer Inc, Akari Therapeutics Plc, NovelMed Therapeutics, Ionis Pharmaceuticals, Johnson & Johnson, Biogen Inc.Global Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market Trends and Insights
Major companies operating in the paroxysmal nocturnal hemoglobinuria (PNH) treatment market are focusing on developing innovative solutions, such as oral factor B inhibitors, to introduce a novel treatment option that enhances therapeutic effectiveness and improves patient convenience in managing PNH. An oral Factor B inhibitor is a targeted small-molecule drug that selectively inhibits Factor B in the alternative complement pathway, aiming to regulate immune system overactivation and reduce inflammation in complement-mediated diseases. For instance, in December 2023, Novartis AG, a Switzerland-based pharmaceutical corporation, announced the food and drug administration (FDA) approval of Fabhalta (iptacopan) as the first oral monotherapy for the treatment of paroxysmal nocturnal hemoglobinuria (PNH). This novel Factor B inhibitor provides a more comprehensive approach to controlling complement-mediated hemolysis by targeting the alternative complement pathway at an earlier stage, resulting in improved hemoglobin levels and a reduced need for blood transfusions. The approval of Fabhalta marks a groundbreaking advancement in PNH treatment by offering enhanced efficacy, better patient compliance, and an alternative to intravenous therapies, thereby transforming the treatment landscape for this rare hematologic disorder.What Are Latest Mergers And Acquisitions In The Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market?
In December 2024, The Max Foundation, a US-based nonprofit organization, announced a collaboration with Novartis AG to enhance global health equity by expanding access to innovative treatments for underserved populations. This deal aims to broaden access to advanced treatment options for paroxysmal nocturnal hemoglobinuria (PNH) patients in low-resource countries by leveraging strategic partnerships, strengthening healthcare infrastructure, providing essential medications at no cost, and fostering medical expertise through education and capacity-building initiatives, ultimately ensuring equitable healthcare access and improving patient outcomes worldwide. Novartis AG is a Switzerland-based pharmaceutical corporation that developed oral monotherapy to manage PNH.Regional Outlook
North America was the largest region in the paroxysmal nocturnal hemoglobinuria (PNH) treatment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market?
The paroxysmal nocturnal hemoglobinuria (PNH) treatment market includes revenues earned by entities by providing services such as diagnostic testing services, genetic counseling, patient support programs, blood transfusion services, iron supplementation therapy, clinical trial services, and telemedicine consultations. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market Report 2026?
The paroxysmal nocturnal hemoglobinuria (pnh) treatment market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the paroxysmal nocturnal hemoglobinuria (pnh) treatment industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $6.89 billion |
| Revenue Forecast In 2035 | $11.32 billion |
| Growth Rate | CAGR of 13.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Treatment Type, Route Of Administration, Market Availability, End-Use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd, Novartis AG, Mayo Clinic, Cleveland Clinic Foundation, Johns Hopkins Medicine, Memorial Sloan Kettering Cancer Center, Stanford Health Care, University of California San Francisco (UCSF) Health, Emory Healthcare, Cedars-Sinai Medical Center, Duke University Health System, Mount Sinai Health System, Dana-Farber Cancer Institute, Swedish Orphan Biovitrum, Alnylam Pharmaceuticals, BioCryst Pharmaceuticals, Apellis Pharmaceuticals, Omeros Corporation, Takeda Pharmaceutical Company, Sanofi S A, Pfizer Inc, Akari Therapeutics Plc, NovelMed Therapeutics, Ionis Pharmaceuticals, Johnson & Johnson, Biogen Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment market was valued at $6.07 billion in 2025, increased to $6.89 billion in 2026, and is projected to reach $11.32 billion by 2030.
The global Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment market is expected to grow at a CAGR of 13.2% from 2026 to 2035 to reach $11.32 billion by 2035.
Some Key Players in the Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment market Include, F. Hoffmann-La Roche Ltd, Novartis AG, Mayo Clinic, Cleveland Clinic Foundation, Johns Hopkins Medicine, Memorial Sloan Kettering Cancer Center, Stanford Health Care, University of California San Francisco (UCSF) Health, Emory Healthcare, Cedars-Sinai Medical Center, Duke University Health System, Mount Sinai Health System, Dana-Farber Cancer Institute, Swedish Orphan Biovitrum, Alnylam Pharmaceuticals, BioCryst Pharmaceuticals, Apellis Pharmaceuticals, Omeros Corporation, Takeda Pharmaceutical Company, Sanofi S A, Pfizer Inc, Akari Therapeutics Plc, NovelMed Therapeutics, Ionis Pharmaceuticals, Johnson & Johnson, Biogen Inc. .
Major trend in this market includes: Advancements In Hemoglobinuria Management With The Approval Of The First Oral Monotherapy For Improved Patient Care. For further insights on this market.
Request for SampleNorth America was the largest region in the paroxysmal nocturnal hemoglobinuria (PNH) treatment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the paroxysmal nocturnal hemoglobinuria (pnh) treatment market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
