
Pediatric And Neonatal Testing Kit Market Report 2026
Global Outlook – By Test Type (Dried Blood Spot, Hearing Screening, Critical Congenital Heart Defect (CCHD), Other Test Types), By Product (Assay Kits And Reagents, Instruments), By Technology (Tandem Mass Spectrometry, Pulse Oximetry, Enzyme-Based Assays, DNA Based Assays, Electrophoresis, Others Technologies), By End User (Hospitals, Diagnostic Centers, Maternity And Specialty Clinics, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Pediatric And Neonatal Testing Kit Market Overview
• Pediatric And Neonatal Testing Kit market size has reached to $4.18 billion in 2025 • Expected to grow to $6.59 billion in 2030 at a compound annual growth rate (CAGR) of 9.7% • Growth Driver: Rising Neonatal And Pediatric Demands Propel Growth In Pediatric And Neonatal Testing Kit Market • Market Trend: Innovations In Pediatric And Neonatal Screening Kits Enhance Early Detection And Treatment Of Rare Diseases • North America was the largest region in 2025.What Is Covered Under Pediatric And Neonatal Testing Kit Market?
A pediatric and neonatal testing kit refers to a collection of medical devices, instruments, and tools specifically designed and curated for assessing and diagnosing infants, newborns, and children. These kits are used by healthcare professionals, such as pediatricians, neonatologists, nurses, and other medical staff, to aid in diagnosing, monitoring, and treating infants and children. The main pediatric and neonatal testing kit test types are dried blood spot, hearing screening, critical congenital heart defect (CCHD), and others. A dried blood spot (DBS) test refers to a diagnostic method that involves collecting a small volume of blood from a person and allowing it to dry on a specialized filter paper. The product types are assay kits and reagents, and instruments that involve various technologies during the process named as tandem mass spectrometry, pulse oximetry, enzyme-based assays, DNA based assays, electrophoresis, and others. The end users are hospitals, diagnostic centers, maternity and specialty clinics, and others.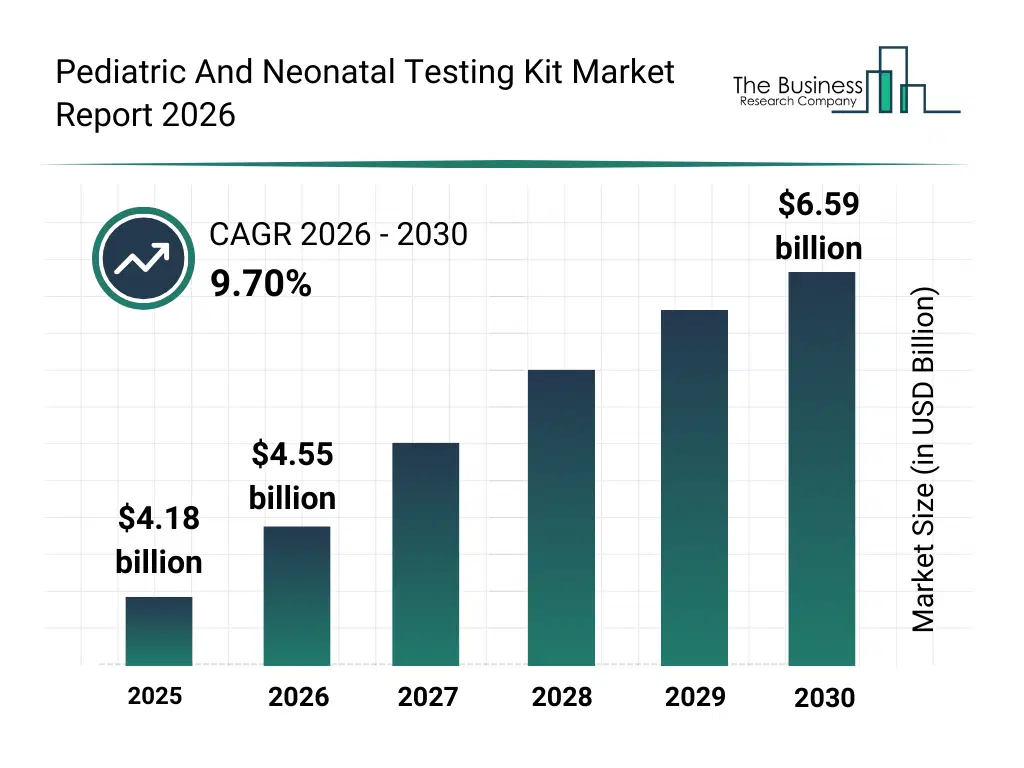
What Is The Pediatric And Neonatal Testing Kit Market Size and Share 2026?
The pediatric and neonatal testing kit market size has grown strongly in recent years. It will grow from $4.18 billion in 2025 to $4.55 billion in 2026 at a compound annual growth rate (CAGR) of 9.1%. The growth in the historic period can be attributed to expansion of neonatal screening initiatives, rising awareness of congenital disorders, improvements in diagnostic assay technologies, growth of pediatric healthcare infrastructure, increased hospital-based testing adoption.What Is The Pediatric And Neonatal Testing Kit Market Growth Forecast?
The pediatric and neonatal testing kit market size is expected to see strong growth in the next few years. It will grow to $6.59 billion in 2030 at a compound annual growth rate (CAGR) of 9.7%. The growth in the forecast period can be attributed to expansion of precision pediatric diagnostics, rising investments in newborn health programs, growing adoption of molecular diagnostic technologies, increasing demand for rapid and portable testing kits, integration of digital health monitoring. Major trends in the forecast period include increasing adoption of point-of-care pediatric testing kits, rising use of dried blood spot screening, growing integration of genetic and metabolic testing, expansion of newborn screening programs, enhanced focus on early disease detection.Global Pediatric And Neonatal Testing Kit Market Segmentation
1) By Test Type: Dried Blood Spot, Hearing Screening, Critical Congenital Heart Defect (CCHD), Other Test Types 2) By Product: Assay Kits And Reagents, Instruments 3) By Technology: Tandem Mass Spectrometry, Pulse Oximetry, Enzyme-Based Assays, DNA Based Assays, Electrophoresis, Others Technologies 4) By End User: Hospitals, Diagnostic Centers, Maternity And Specialty Clinics, Other End Users Subsegments: 1) By Dried Blood Spot: Newborn Screening Panels, Genetic Testing, Infectious Disease Screening 2) By Hearing Screening: Automated Auditory Brainstem Response (ABR) Tests, Otoacoustic Emissions (OAE) Tests, Portable Hearing Screening Devices 3) By Critical Congenital Heart Defect (CCHD): Pulse Oximetry Screening Kits, Echocardiography Screening Tools, Clinical Assessment Protocols 4) By Other Test Types: Blood Gas Analysis Kits, Urine Testing Kits, Infectious Disease Testing, Point-of-Care Testing DevicesWhat Is The Driver Of The Pediatric And Neonatal Testing Kit Market?
The increasing neonatal and pediatric population is driving the pediatric and neonatal testing kit market going forward. Neonates are newborn children, typically ranging from birth through the first 28 days of life and Pediatric patients are children and infants aged 21 or younger at the time of their diagnosis or treatment. The pediatric and neonatal testing kit market is indispensable for addressing the specific healthcare needs of the increasing neonatal and pediatric population. For instance, in October 2025, according to the Australian Bureau of Statistics, an Australia-based government agency, in 2024, a total of 292,318 births were registered, reflecting a 1.9% rise (an additional 5,320 births) compared to 2023, with 150,299 male and 142,019 female babies. Therefore, increasing the Neonatal and Pediatric Population drives the pediatric and neonatal testing kits industry.Key Players In The Global Pediatric And Neonatal Testing Kit Market
Major companies operating in the pediatric and neonatal testing kit market are F. Hoffmann-La Roche AG, Thermo Fisher Scientific Inc., Abbott Laboratories, Medtronic Plc, Siemens Healthineers, GE Healthcare GmbH, Eurofins Scientific SE, Trivitron Healthcare Pvt Ltd., Hologic Inc., PerkinElmer Inc., Quidel Corporation, Bio-Rad Laboratories Inc., QIAGEN NV, Natus Medical Incorporated, BioFire Diagnostics LLC, Luminex Corporation, Meridian Bioscience Inc., MP Biomedicals LLC, ELITech Group SAS, Baebies Inc., Labsystems Diagnostics Oy, Beckman Coulter Inc., JETA Molecular BV, ZenTech SA, Hangzhou Testsea Biotechnology Co. Ltd.Global Pediatric And Neonatal Testing Kit Market Trends and Insights
Major companies operating in the pediatric and neonatal testing market are focusing on developing advanced solutions, such as mass screening tests, to enhance early detection, reduce diagnostic delays, and improve treatment outcomes for newborns. Mass screening tests refer to diagnostic kits designed to identify serious conditions in newborns shortly after birth, enabling prompt medical intervention. For instance, in May 2024, Shimadzu Techno-Research, Inc., a Japan-based contract analysis company, launched a new contract service to conduct PCR testing for enhanced newborn mass screening. The service provides testing for severe rare diseases, including Severe Combined Immunodeficiency (SCID), B-cell Deficiency (BCD), and Spinal Muscular Atrophy (SMA), and is currently offered in 40 out of Japan’s 47 prefectures, with plans to expand nationwide through public funding. By partnering with hospitals and delivery facilities, the service aims to improve the health and welfare of newborns throughout Japan.What Are Latest Mergers And Acquisitions In The Pediatric And Neonatal Testing Kit Market?
In March 2025, LaCAR MDx Technologies, a Belgium-based molecular diagnostics company, acquired the Newborn Screening Division of Baebies, Inc. for an undisclosed amount. Through this acquisition, LaCAR MDx aims to expand its pediatric and neonatal testing capabilities by integrating Baebies’ expertise in newborn screening and enhancing its portfolio of diagnostic tests for early detection of congenital disorders. Baebies, Inc. is a US-based company that provides pediatric and neonatal testing kits, including screening for metabolic, endocrine, and genetic conditions.Regional Outlook
North America was the largest region in the pediatric and neonatal testing kit market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Pediatric And Neonatal Testing Kit Market?
The pediatric and neonatal testing kit market consists of sales of maternal blood screening, ultrasound tests, amniocentesis, and glucose screening. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Pediatric And Neonatal Testing Kit Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $4.55 billion |
| Revenue Forecast In 2035 | $6.59 billion |
| Growth Rate | CAGR of 9.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Test Type, Product, Technology, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche AG, Thermo Fisher Scientific Inc., Abbott Laboratories, Medtronic Plc, Siemens Healthineers, GE Healthcare GmbH, Eurofins Scientific SE, Trivitron Healthcare Pvt Ltd., Hologic Inc., PerkinElmer Inc., Quidel Corporation, Bio-Rad Laboratories Inc., QIAGEN NV, Natus Medical Incorporated, BioFire Diagnostics LLC, Luminex Corporation, Meridian Bioscience Inc., MP Biomedicals LLC, ELITech Group SAS, Baebies Inc., Labsystems Diagnostics Oy, Beckman Coulter Inc., JETA Molecular BV, ZenTech SA, Hangzhou Testsea Biotechnology Co. Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
