
Pediatric Clinical Trials Market Report 2026
Global Outlook – By Phase (Phase I, Phase II, Phase III, Phase IV), By Study Design (Treatment Studies, Observational Studies), By Therapeutic Areas (Infectious Diseases, Oncology, Autoimmune Or Inflammatory Diseases, Respiratory Disorders, Mental Health Disorders, Other Therapeutic Areas ) - Market Size, Trends, And Global Forecast 2026-2035
Pediatric Clinical Trials Market Overview
• Pediatric Clinical Trials market size has reached to $18.38 billion in 2025 • Expected to grow to $26.67 billion in 2030 at a compound annual growth rate (CAGR) of 7.8% • Growth Driver: Rising Pediatric Cancer Cases Propel Growth In Pediatric Cancer Trials Market • Market Trend: Strategic Collaborations In Pediatric Clinical Trials • North America was the largest region in 2025.What Is Covered Under Pediatric Clinical Trials Market?
Pediatric clinical trials are trials that investigate the effectiveness, safety, and dosage of pharmaceuticals, medical devices, and other therapies in children. It is essential to develop age-specific, scientifically proven treatments and procedures to identify and improve the best medical therapy possible. The main pediatric clinical trial phase types are phase I, phase II, phase III, and phase IV. Phase I trials are primarily focused on determining the safety and dosing range of new medicine in around 20-100 healthy volunteers. The various study designs are treatment studies, and observational studies used in various therapeutic areas that include infectious diseases, oncology, autoimmune or inflammatory diseases, respiratory disorders, mental health disorders, and others.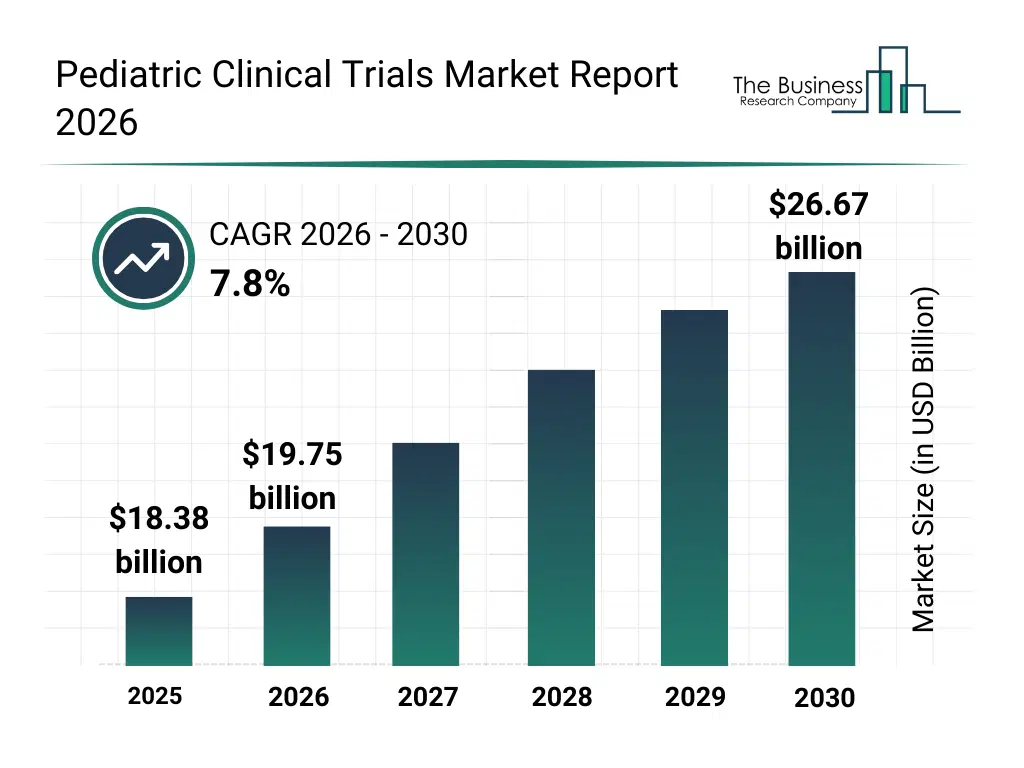
What Is The Pediatric Clinical Trials Market Size and Share 2026?
The pediatric clinical trials market size has grown strongly in recent years. It will grow from $18.38 billion in 2025 to $19.75 billion in 2026 at a compound annual growth rate (CAGR) of 7.4%. The growth in the historic period can be attributed to limited pediatric-specific drug research, reliance on adult trial data, ethical and regulatory challenges, low patient recruitment rates, minimal use of advanced analytics in trial design.What Is The Pediatric Clinical Trials Market Growth Forecast?
The pediatric clinical trials market size is expected to see strong growth in the next few years. It will grow to $26.67 billion in 2030 at a compound annual growth rate (CAGR) of 7.8%. The growth in the forecast period can be attributed to growing demand for pediatric-specific therapies, increasing adoption of digital and ai tools, rising regulatory incentives for pediatric studies, growing collaboration between pharma and research institutes, increasing focus on patient-centric and personalized clinical trials. Major trends in the forecast period include increasing adoption of precision medicine in pediatric clinical trials, rising use of ai and digital tools for trial design and data analysis, growing integration of real-world evidence in pediatric studies, increasing investment in pediatric drug development, rising focus on personalized and age-specific therapeutics.Global Pediatric Clinical Trials Market Segmentation
1) By Phase: Phase I, Phase II, Phase III, Phase IV 2) By Study Design: Treatment Studies, Observational Studies 3) By Therapeutic Areas: Infectious Diseases, Oncology, Autoimmune Or Inflammatory Diseases, Respiratory Disorders, Mental Health Disorders, Other Therapeutic Areas Subsegments: 1) By Phase I: First-in-Pediatrics Trials, Dose Escalation Studies, Safety And Tolerability Assessments 2) By Phase II: Efficacy Studies, Dose Optimization Trials, Pharmacokinetic Studies 3) By Phase III: Large-Scale Efficacy Trials, Comparative Studies, Long-Term Safety Studies 4) By Phase IV: Post-Marketing Surveillance, Long-Term Safety Monitoring, Effectiveness Studies In Real-World SettingsWhat Is The Driver Of The Pediatric Clinical Trials Market?
The rise in pediatric cancer cases is expected to propel the pediatric clinical trials market going forward. Pediatric cancer refers to cancers that develop in children and adolescents under the age of 14. Pediatric cancers are extremely uncommon, and their growth and dissemination patterns may be distinct from those of adult tumors. Pediatric clinical trials continue to play a significant role in the advancement of prognosis and treatment. For instance, in January 2023, according to the American Cancer Society, a US-based voluntary health organization, it is estimated that about 9,910 children in the US under the age of 15 will be diagnosed with cancer and about 1,040 children under the age of 15 are expected to die from cancer in 2023. Therefore, the growing healthcare expenditure is driving the pediatric clinical trials industry.Key Players In The Global Pediatric Clinical Trials Market
Major companies operating in the pediatric clinical trials market are Medpac Inc., Pharmaceutical Product Development Inc., ICON plc, Syneos Health Inc., QPS Holdings LLC, Pfizer Inc., IQVIA Inc., Premier Research, Labcorp - Laboratory Corporation of America Holdings, The Emmes Company LLC, Synteract Inc., Charles River Laboratories International Inc., Covance Inc., Bristol-Myers Squibb Company, GlaxoSmithKline plc, BioNTech SE, Moderna Inc., Parexel International Corporation, PRA Health Sciences, Wuxi AppTec, Merck & Co. Inc., AstraZeneca plc, Sanofi S.A., Johnson & Johnson, Eli Lilly and Company, Takeda Pharmaceutical Company Limited, C.H. Boehringer Sohn AG & Co. KG, AbbVie Inc., Teva Pharmaceutical Industries Ltd., Oracle Health sciencesGlobal Pediatric Clinical Trials Market Trends and Insights
Major companies operating in pediatric clinical trials are focused on collaboration and partnerships to provide reliable services to customers. A strategic partnership refers to a structured affiliation between two or more commercial enterprises, typically established via one or more business agreements or contracts. For instance, in September 2023, Strados Labs, a US-based provider of smart biosensors for respiratory monitoring, partnered with Ann & Robert H. Lurie Children's Hospital, a US-based pediatric acute care children's hospital, to conduct a clinical trial on RESP Biosensor. The RESP Biosensor is a wearable device that uses acoustic sensors to monitor lung sounds for asthma symptoms such as coughing and wheezing. The goal of the clinical trial is to evaluate the effectiveness of the RESP Biosensor in accurately detecting and monitoring asthma exacerbations in children.What Are Latest Mergers And Acquisitions In The Pediatric Clinical Trials Market?
In March 2024, PCM Trials, a US-based provider of mobile research nurse visits for decentralized clinical trials, acquired EmVenio Research for an undisclosed amount. Through this acquisition, PCM Trials will offer a more patient-centric and decentralized model for clinical trials, improving participant recruitment and retention, particularly among underrepresented minorities. EmVenio is a US-based company that enhances access to pediatric clinical trials, particularly for diverse and underserved populations.Regional Outlook
North America was the largest region in the pediatric clinical trials market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Pediatric Clinical Trials Market?
The pediatric clinical trials market includes revenues earned by entities through clinical research services, consulting, outsourcing services, and medical communications. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Pediatric Clinical Trials Market Report 2026?
The pediatric clinical trials market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the pediatric clinical trials industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Pediatric Clinical Trials Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $19.75 billion |
| Revenue Forecast In 2035 | $26.67 billion |
| Growth Rate | CAGR of 7.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Phase, Study Design, Therapeutic Areas |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Medpac Inc., Pharmaceutical Product Development Inc., ICON plc, Syneos Health Inc., QPS Holdings LLC, Pfizer Inc., IQVIA Inc., Premier Research, Labcorp - Laboratory Corporation of America Holdings, The Emmes Company LLC, Synteract Inc., Charles River Laboratories International Inc., Covance Inc., Bristol-Myers Squibb Company, GlaxoSmithKline plc, BioNTech SE, Moderna Inc., Parexel International Corporation, PRA Health Sciences, Wuxi AppTec, Merck & Co. Inc., AstraZeneca plc, Sanofi S.A., Johnson & Johnson, Eli Lilly and Company, Takeda Pharmaceutical Company Limited, C.H. Boehringer Sohn AG & Co. KG, AbbVie Inc., Teva Pharmaceutical Industries Ltd., Oracle Health sciences |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
