
Pharmaceutical Analytical Testing Market Report 2026
Global Outlook – By Service Type (Bioanalytical Testing, Method Development And Validation, Stability Testing, Drug Substances Testing, Microbial Testing, Physical Characterization, Other Service Types), By Technique Used (Chromatography, Spectroscopy, Mass Spectrometry, Electrophoresis, Microscopy), By Application (Quality Control, Research And Development, Regulatory Compliance, Drug Approval And Registration, Post-Marketing Surveillance), By End User (Pharmaceutical Companies, Contract Research Organizations (CROs), Academic Institutions, Regulatory Bodies, Testing Laboratories) - Market Size, Trends, And Global Forecast 2026-2035
Pharmaceutical Analytical Testing Market Overview
• Pharmaceutical Analytical Testing market size has reached to $8.21 billion in 2025 • Expected to grow to $12.46 billion in 2030 at a compound annual growth rate (CAGR) of 8.7% • Growth Driver: Surge In Drug Approvals And Trials Fueling The Growth Of The Market Due To Advancements In Precision Medicine • Market Trend: Digital Reference Materials Platform Transforming Pharmaceutical Analytical Testing • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Pharmaceutical Analytical Testing Market?
Pharmaceutical analytical testing refers to the comprehensive examination of pharmaceutical products using various scientific methods to assess their physical, chemical, biological, and microbiological characteristics, ensuring they meet required quality, safety, and efficacy standards. This testing is a vital component throughout the drug development and manufacturing process, as it verifies the identity, purity, potency, and stability of drugs in compliance with regulatory guidelines. The main types of services in pharmaceutical analytical testing are bioanalytical testing, method development and validation, stability testing, drug substance testing, microbial testing, physical characterization, and others. Bioanalytical testing is the process of measuring drugs, metabolites, and biomarkers in biological samples such as blood or urine. It used various techniques, such as chromatography, spectroscopy, mass spectrometry, electrophoresis, and microscopy, for various applications, including quality control, research and development, regulatory compliance, drug approval and registration, and post-marketing surveillance. It serves end users such as pharmaceutical companies, contract research organizations (CROs), academic institutions, regulatory bodies, and testing laboratories.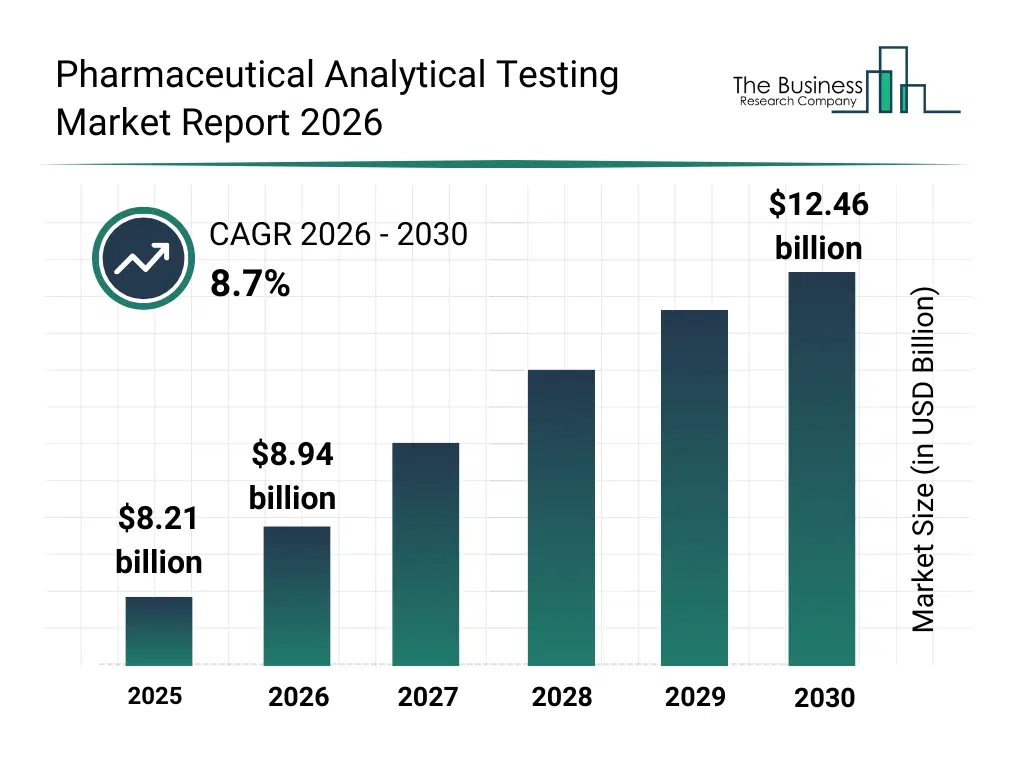
What Is The Pharmaceutical Analytical Testing Market Size and Share 2026?
The pharmaceutical analytical testing market size has grown strongly in recent years. It will grow from $8.21 billion in 2025 to $8.94 billion in 2026 at a compound annual growth rate (CAGR) of 8.9%. The growth in the historic period can be attributed to expansion of pharmaceutical manufacturing activities, increasing regulatory scrutiny on drug quality, growth in generic drug development, rising investments in R&D pipelines, expansion of global testing laboratory networks.What Is The Pharmaceutical Analytical Testing Market Growth Forecast?
The pharmaceutical analytical testing market size is expected to see strong growth in the next few years. It will grow to $12.46 billion in 2030 at a compound annual growth rate (CAGR) of 8.7%. The growth in the forecast period can be attributed to increasing demand for complex biologics testing, rising adoption of automated analytical platforms, expansion of personalized medicine development, growing focus on faster regulatory approvals, increasing reliance on contract testing organizations. Major trends in the forecast period include increasing adoption of advanced analytical testing techniques, rising demand for outsourced testing services, growing focus on regulatory-compliant testing protocols, expansion of stability and bioanalytical testing, enhanced integration of automation in testing laboratories.Global Pharmaceutical Analytical Testing Market Segmentation
1) By Service Type: Bioanalytical Testing, Method Development And Validation, Stability Testing, Drug Substances Testing, Microbial Testing, Physical Characterization, Other Service Types 2) By Technique Used: Chromatography, Spectroscopy, Mass Spectrometry, Electrophoresis, Microscopy 3) By Application: Quality Control, Research And Development, Regulatory Compliance, Drug Approval And Registration, Post-Marketing Surveillance 4) By End User: Pharmaceutical Companies, Contract Research Organizations (CROs), Academic Institutions, Regulatory Bodies, Testing Laboratories Subsegments: 1) By Bioanalytical Testing: Pharmacokinetics Testing, Biomarker Testing, Immunogenicity Testing, Toxicokinetics Testing 2) By Method Development And Validation: Analytical Method Development, Analytical Method Validation, Method Transfer 3) By Stability Testing: Long-Term Stability Testing, Accelerated Stability Testing, Stress Stability Testing, Photostability Testing 4) By Drug Substances Testing: Raw Material Testing, Intermediate Testing, Final Drug Substance Testing 5) By Microbial Testing: Bioburden Testing, Sterility Testing, Endotoxin Testing, Microbial Limit Testing 6) By Physical Characterization: Particle Size Analysis, Polymorphism Analysis, Rheology Testing, Thermal Analysis 7) By Other Service Types: Impurity Testing, Elemental Impurity Testing, Extractables And Leachables Testing, Packaging TestingWhat Is The Driver Of The Pharmaceutical Analytical Testing Market?
The rising drug approvals and trials are expected to propel the growth of the pharmaceutical analytical testing market going forward. Drug approvals and trials refer to the regulated process of testing new medications for safety and effectiveness through clinical trials, followed by official authorization from health authorities for public use. The rising drug approvals and trials are due to the advancement of precision medicine, as it allows for the development of highly targeted therapies tailored to individual genetic profiles, increasing the likelihood of efficacy and regulatory success. Pharmaceutical analytical testing is essential for drug approvals and clinical trials as it ensures the safety, efficacy, and quality of drug products by providing validated data on stability, impurities, bioavailability, and pharmacokinetics, supporting regulatory compliance and informed decision-making. For instance, in December 2024, according to the Association of the British Pharmaceutical Industry, a UK-based pharmaceutical industry trade body, the total number of industry-sponsored clinical trials initiated in the United Kingdom increased from 411 in 2022 to 426 in 2023, while Phase III trial initiations grew by 16.5 percent over the same period, rising from 182 to 212. Therefore, the rising drug approvals and trials are driving the growth of the pharmaceutical analytical testing industry.Key Players In The Global Pharmaceutical Analytical Testing Market
Major companies operating in the pharmaceutical analytical testing market are Thermo Fisher Scientific Inc., Merck KGaA, IQVIA Holdings Inc., Laboratory Corporation of America Holdings, ICON Public Limited Company, Eurofins Scientific SE, Pharmaceutical Product Development LLC, WuXi AppTec Co. Ltd., Syneos Health Inc., Steris plc, Catalent Inc., Charles River Laboratories International Inc., Intertek Group plc, SGS Société Générale de Surveillance SA, Almac Group Limited, Pace Analytical Services LLC, Neotron S.p.A., Toxikon Corporation, Element Materials Technology Group Limited, Boston Analytical Inc., ALS Limited, Frontage Laboratories Inc.Global Pharmaceutical Analytical Testing Market Trends and Insights
Major companies operating in the pharmaceutical analytical testing market are focusing on introducing innovative solutions, such as digital reference materials platforms, to enhance the accuracy, traceability, and efficiency of laboratory testing processes, thereby ensuring regulatory compliance and improving overall data integrity. A digital reference materials platform refers to an online or software-based system that provides authenticated, standardized digital datasets or models used for calibrating, validating, and verifying analytical testing processes in pharmaceutical laboratories. For instance, in November 2023, Merck Group, a Germany-based pharmaceutical company, launched ChemisTwin, the first-ever digital reference materials platform designed for analytical testing that automates the analysis of sample purity, compound identification, and degradation using calibrated, algorithm-based digital references. Featuring digital signatures for over 1,500 reference materials, ChemisTwin streamlines and accelerates workflows in pharmaceutical quality control, quality assurance, and research and development by providing traceable digital records linked to physical reference materials. This innovative platform reduces manual tasks such as weighing and documentation, enhances sampling rates, and improves detection of sample inconsistencies, thereby ensuring safer and more reliable products.What Are Latest Mergers And Acquisitions In The Pharmaceutical Analytical Testing Market?
In April 2025, Eureka Analytical Services Pvt Ltd., an India-based company that provides testing, inspection, and auditing services, acquired Teena Biolabs Pvt Ltd. for an undisclosed amount. With this acquisition, Eureka aims to significantly expand Eureka Analytical Services' range of pharmaceutical analysis services in India, serve the market more holistically, and position both Eureka and the GBA Group as leading addresses in laboratory analysis in the country. Teena Biolabs Pvt Ltd. is an India-based contract testing laboratory that specializes in providing analytical testing services, particularly for the pharmaceutical and biotech industries, with a focus on quality and regulatory compliance.Regional Outlook
North America was the largest region in the pharmaceutical analytical testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Pharmaceutical Analytical Testing Market?
The pharmaceutical analytical testing market includes revenues earned by entities by providing services, such as method development and validation, stability testing, quality control testing, raw material testing, impurity profiling, and elemental analysis. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Pharmaceutical Analytical Testing Market Report 2026?
The pharmaceutical analytical testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the pharmaceutical analytical testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Pharmaceutical Analytical Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $8.94 billion |
| Revenue Forecast In 2035 | $12.46 billion |
| Growth Rate | CAGR of 8.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Service Type, Technique Used, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Merck KGaA, IQVIA Holdings Inc., Laboratory Corporation of America Holdings, ICON Public Limited Company, Eurofins Scientific SE, Pharmaceutical Product Development LLC, WuXi AppTec Co. Ltd., Syneos Health Inc., Steris plc, Catalent Inc., Charles River Laboratories International Inc., Intertek Group plc, SGS Société Générale de Surveillance SA, Almac Group Limited, Pace Analytical Services LLC, Neotron S.p.A., Toxikon Corporation, Element Materials Technology Group Limited, Boston Analytical Inc., ALS Limited, Frontage Laboratories Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
