
Pharmaceutical Contract Development And Manufacturing Organization (CMO) Market Report 2026
Global Outlook – By Type (Contract Manufacturing Services, Contract Research Services), By Research Phase (Preclinical, Phase I, Phase II, Phase III, Phase IV), By End User (Big Pharmaceutical Companies, Generic Pharmaceutical Companies, Small And Medium-Sized Pharmaceutical Companies) – Market Size, Trends, Strategies, and Forecast to 2035
Pharmaceutical Contract Development And Manufacturing Organization (CMO) Market Overview
• Pharmaceutical Contract Development And Manufacturing Organization (CMO) market size has reached to $54.73 billion in 2025 • Expected to grow to $90.76 billion in 2030 at a compound annual growth rate (CAGR) of 10.6% • Growth Driver: Increasing Patent Expirations Are Anticipated To Fuel The Growth Of The Pharmaceutical Contract Development And Manufacturing Market • Market Trend: Pharmaceutical Contract Development And Manufacturing Firms Introduce Innovative Devpack Services To Enhance Industry Capabilities • North America was the largest region in 2025 and Middle East is the fastest growing region.What Is Covered Under Pharmaceutical Contract Development And Manufacturing Organization (CMO) Market?
Pharmaceutical contract development and manufacturing refers to a business that offers services for drug research and manufacturing within the pharmaceutical sector. Using CDMOs as a partner, pharmaceutical companies can outsource medication development and production. Full-service drug development and manufacturing outsourcing companies (CDMOs) can handle every stage of the process and work with clients who want to outsource specific steps in their workflow. Everything is based on the requirements of each client. The main types of pharmaceutical contract development and manufacturing organizations (CMO) are contract manufacturing services and contract research services. Contract manufacturing services refer to the outsourcing of manufacturing processes or the production of goods to a third-party company, known as a contract manufacturer. The various research phases include preclinical, phase I, phase II, phase III, and phase IV. The different end-use sectors include big pharmaceutical companies, generic pharmaceutical companies, and small & medium-sized pharmaceutical companies.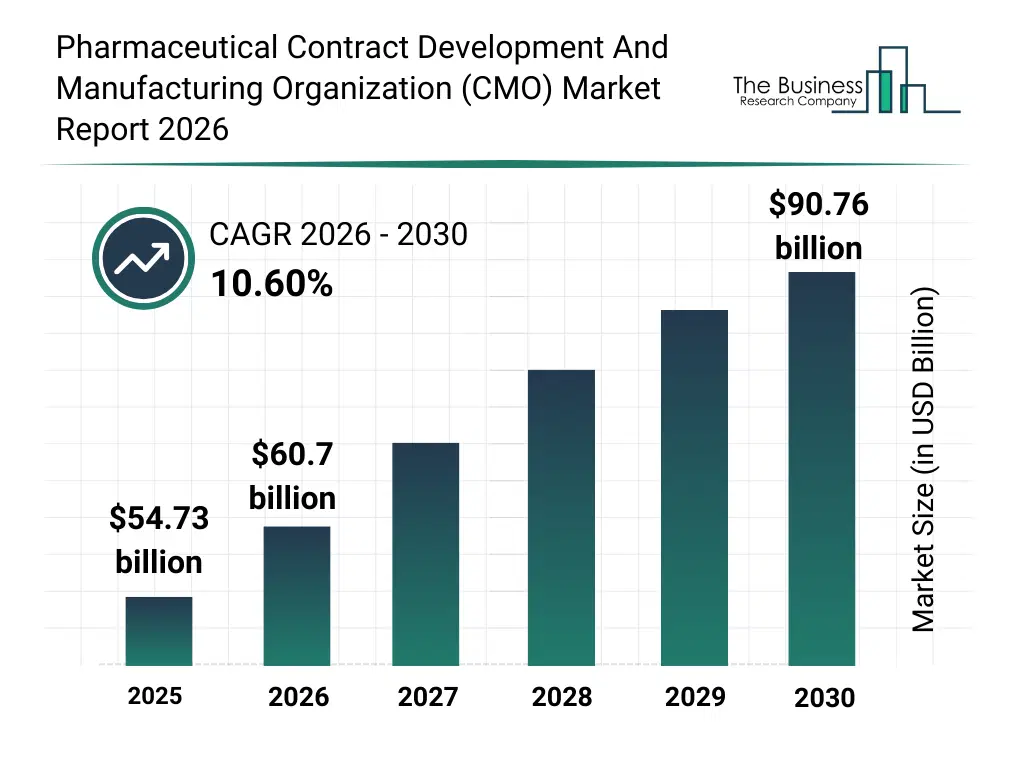
What Is The Pharmaceutical Contract Development And Manufacturing Organization (CMO) Market Size and Share 2026?
The pharmaceutical contract development and manufacturing organization (cmo) market size has grown rapidly in recent years. It will grow from $54.73 billion in 2025 to $60.7 billion in 2026 at a compound annual growth rate (CAGR) of 10.9%. The growth in the historic period can be attributed to increasing r&d expenditures by pharmaceutical companies, rising prevalence of chronic diseases, growth in generic drug demand, technological advancements in drug manufacturing, increasing focus on preclinical and clinical research.What Is The Pharmaceutical Contract Development And Manufacturing Organization (CMO) Market Growth Forecast?
The pharmaceutical contract development and manufacturing organization (cmo) market size is expected to see rapid growth in the next few years. It will grow to $90.76 billion in 2030 at a compound annual growth rate (CAGR) of 10.6%. The growth in the forecast period can be attributed to rising adoption of personalized medicine, growing demand for biologics and complex therapeutics, expansion of cdmo capabilities in emerging markets, integration of digital technologies in drug development, increasing strategic partnerships and mergers in pharmaceutical sector. Major trends in the forecast period include outsourcing of drug development and manufacturing, expansion of cdmo service portfolios, regulatory compliance and quality assurance focus, strategic partnerships between pharma companies and cdmos, adoption of advanced analytical and testing services.Global Pharmaceutical Contract Development And Manufacturing Organization (CMO) Market Segmentation
1) By Type: Contract Manufacturing Services, Contract Research Services 2) By Research Phase: Preclinical, Phase I, Phase II, Phase III, Phase IV 3) By End User: Big Pharmaceutical Companies, Generic Pharmaceutical Companies, Small And Medium-Sized Pharmaceutical Companies Subsegments: 1) By Contract Manufacturing Services: Active Pharmaceutical Ingredient (API) Manufacturing, Formulation Development, Finished Dosage Form Manufacturing, Packaging Services, Supply Chain Management 2) By Contract Research Services: Preclinical Research Services, Clinical Trial Management, Regulatory Affairs Consulting, Biostatistics And Data Management, Analytical And Testing ServicesWhat Is The Driver Of The Pharmaceutical Contract Development And Manufacturing Organization (CMO) Market?
The increasing patent expiration is expected to propel the pharmaceutical contract development and manufacturing market. Patent expiration refers to the end of the legally protected exclusive rights granted to an inventor or patent holder to make, sell, or use an invention for a specified period of time. Pharmaceutical contract development and manufacturing aid in the swift development and production of generic drugs for market entry upon patent expiration. For instance, in February 2024, according to the DrugTimes is a Chinese-based company focused on the pharmaceutical and biotech industry, by 2037, more than 25 products will lose patent protection, potentially resulting in a combined revenue decline exceeding $236.4 billion, based on 2024 sales. Therefore, the increasing patent expiration is driving the pharmaceutical contract development and manufacturing market.Key Players In The Global Pharmaceutical Contract Development And Manufacturing Organization (CMO) Market
Major companies operating in the pharmaceutical contract development and manufacturing organization (cmo) market are Lonza Group, Catalent Inc, WuXi AppTec Inc., Recipharm AB, Almac Group, Aenova Group, Baxter International Inc, SGS Life Science Services SA, Jubilant Pharmova Ltd, Dishman Pharmaceuticals, Kemwell Pvt. Ltd., Nipro Corp., CMIC Group, Sawai Pharmaceutical Co. Ltd, IDT Australia Limited, Aurigene Pharmaceutical Services Limited, Vetter Pharma International GMBH, Consort Medical PLC, Siegfried Holding AG, Evonik Industries, NextPharma, Royal DSM N.V, HAUPT Pharma AG, Famar, OTC-PharmNEUCA, Farmacol, Polska Grupa Farmaceutyczna, Polpharma, TZMO, Eurofarma Laboratórios S.A., Ache Laboratórios Farmacêuticos S.A., Blanver Farmoquímica e Farmacêutica S.A., Prati-Donaduzzi, União Química Farmacêutica Nacional S.A, Laboratorios Richmond S.A.C.I.F., Bago S.A., Elea Laboratories S.A.C.I. y F., Gador S.A., LIFEPharma, Neopharm, Gulf Pharmaceutical Industries JULPHA, NewBridge Pharmaceuticals Limited, Aspen Pharmacare Holdings Limited, Adcock Ingram Holdings Limited, Pharma-Q (Pty) Ltd, Vital Health Foods, EIPICO (Egyptian International Pharmaceutical Industries Company), Pharco PharmaceuticalsGlobal Pharmaceutical Contract Development And Manufacturing Organization (CMO) Market Trends and Insights
Major companies operating in the pharmaceutical contract development and manufacturing market are providing new services such as the DevPack to enhance their service portfolios and meet the evolving needs of the industry. The DevPack is a service optimized across multiple partners to create a standardized dataset of development process steps. For instance, in May 2023, ASM Research Chemicals, a Germany-based company providing contract research and manufacturing services launched DevPack, a new development pack service intended for active pharmaceutical ingredients (APIs) manufacturing. The DevPack service, optimized across ten core partners, offers a standardized dataset for development processes. It aids in streamlining API research, development, and scale-up. Ideal for CMOs and pharmaceutical firms aiming for quick early-stage manufacturing route development for APIs, the service also supports analytical development and regulatory submissions.What Are Latest Mergers And Acquisitions In The Pharmaceutical Contract Development And Manufacturing Organization (CMO) Market?
In December 2023, Ajinomoto Group, a Japan-based global food and biotechnology company, acquired Forge Biologics Inc., for an undisclosed amount. With this acquisition, Ajinomoto Group aims to expand its gene therapy contract development and manufacturing capabilities, strengthen its global biopharmaceutical manufacturing footprint, and enhance its portfolio in advanced therapeutics. Forge Biologics Inc. is a US-based gene therapy contract development and manufacturing organization that provides AAV-based gene therapy solutions.Regional Insights
North America was the largest region in the global pharmaceutical contract development and manufacturing market in 2025. The Middle East is expected to be the fastest-growing region in the global pharmaceutical contract development and manufacturing market during the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Pharmaceutical Contract Development And Manufacturing Organization (CMO) Market?
The pharmaceutical contract development and manufacturing market includes revenues earned by entities by providing drug development and manufacturing services. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Pharmaceutical Contract Development And Manufacturing Organization (CMO) Market Report 2026?
The pharmaceutical contract development and manufacturing organization (cmo) market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the pharmaceutical contract development and manufacturing organization (cmo) industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Pharmaceutical Contract Development And Manufacturing Organization (CMO) Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $60.7 billion |
| Revenue Forecast In 2035 | $90.76 billion |
| Growth Rate | CAGR of 10.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Research Phase, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Lonza Group, Catalent Inc, WuXi AppTec Inc., Recipharm AB, Almac Group, Aenova Group, Baxter International Inc, SGS Life Science Services SA, Jubilant Pharmova Ltd, Dishman Pharmaceuticals, Kemwell Pvt. Ltd., Nipro Corp., CMIC Group, Sawai Pharmaceutical Co. Ltd, IDT Australia Limited, Aurigene Pharmaceutical Services Limited, Vetter Pharma International GMBH, Consort Medical PLC, Siegfried Holding AG, Evonik Industries, NextPharma, Royal DSM N.V, HAUPT Pharma AG, Famar, OTC-PharmNEUCA, Farmacol, Polska Grupa Farmaceutyczna, Polpharma, TZMO, Eurofarma Laboratórios S.A., Ache Laboratórios Farmacêuticos S.A., Blanver Farmoquímica e Farmacêutica S.A., Prati-Donaduzzi, União Química Farmacêutica Nacional S.A, Laboratorios Richmond S.A.C.I.F., Bago S.A., Elea Laboratories S.A.C.I. y F., Gador S.A., LIFEPharma, Neopharm, Gulf Pharmaceutical Industries JULPHA, NewBridge Pharmaceuticals Limited, Aspen Pharmacare Holdings Limited, Adcock Ingram Holdings Limited, Pharma-Q (Pty) Ltd, Vital Health Foods, EIPICO (Egyptian International Pharmaceutical Industries Company), Pharco Pharmaceuticals |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
