
Pharmaceutical Quality Control Market Report 2026
Global Outlook – By Product (Consumables, Services, Instruments), By Deployment Type (Cloud-Based And Web-Based, On-Premises), By Analysis Type (Sterility Testing, Membrane Filtration, Direct Inoculation, Bio Burden Testing, Aerobic Count Testing, Anaerobic Count Testing, Spore Count Testing, Fungi or Mold Count Testing, Endotoxin Testing, Other Analysis Types), By End-user (Pharmaceutical And Biotechnology Companies, Contract Development and Manufacturing Organization Or Contract Research Organization, Research Laboratories) – Market Size, Trends, Strategies, and Forecast to 2035
Pharmaceutical Quality Control Market Overview
• Pharmaceutical Quality Control market size has reached to $9.08 billion in 2025 • Expected to grow to $14.59 billion in 2030 at a compound annual growth rate (CAGR) of 9.9% • Growth Driver: The Rising Demand For Biosimilars Drives The Growth Of The Market Due To Patent Expirations And The Need For Rigorous Quality Control • Market Trend: Artificial Intelligence-Powered Software Revolutionizes Accuracy And Compliance In Drug Manufacturing • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Pharmaceutical Quality Control Market?
Pharmaceutical quality control is a systematic process that ensures the identity, strength, purity, and consistency of pharmaceutical products. It involves rigorous testing of raw materials, in-process samples, and finished products to meet established standards. The purpose is to guarantee the safety and efficacy of medicines before they reach consumers. The main products of pharmaceutical quality control are consumables, services, and instruments. Pharmaceutical quality control consumables refer to the essential materials and supplies, such as culture media, reagents, assay kits, filters, and pipette tips, that are used during laboratory testing and analysis to ensure the safety, quality, and compliance of pharmaceutical products. These can be deployed through varied deployment models such as cloud-based, web-based, and on-premises methods. The analysis types involved encompass sterility testing, membrane filtration, direct inoculation, bioburden testing, aerobic count testing, anaerobic count testing, spore count testing, fungi or mold count testing, endotoxin testing, and others, and caters to various end users, including pharmaceutical and biotechnology companies, contract development and manufacturing organizations or contract research organizations, and research laboratories.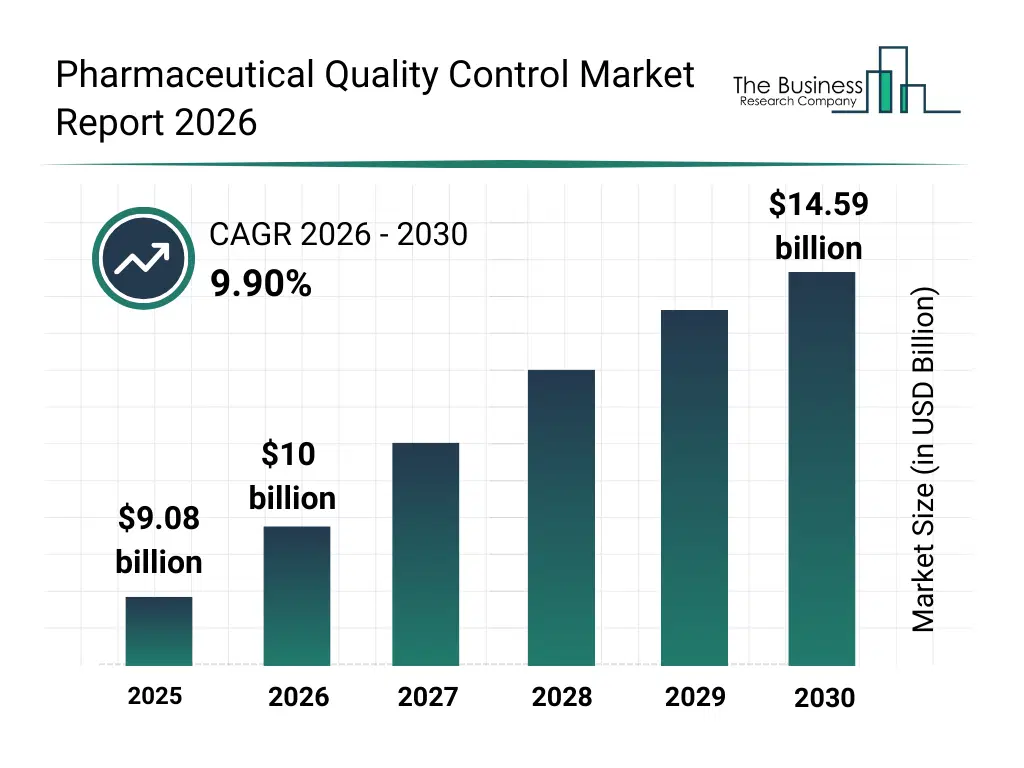
What Is The Pharmaceutical Quality Control Market Size and Share 2026?
The pharmaceutical quality control market size has grown rapidly in recent years. It will grow from $9.08 billion in 2025 to $10 billion in 2026 at a compound annual growth rate (CAGR) of 10.2%. The growth in the historic period can be attributed to limited access to advanced qc instruments, reliance on manual testing methods, growing pharmaceutical manufacturing, increasing regulatory compliance requirements, expansion of microbiological testing capabilities.What Is The Pharmaceutical Quality Control Market Growth Forecast?
The pharmaceutical quality control market size is expected to see strong growth in the next few years. It will grow to $14.59 billion in 2030 at a compound annual growth rate (CAGR) of 9.9%. The growth in the forecast period can be attributed to integration of cloud-based and ai-enabled qc solutions, rising adoption of automated analytical instruments, increasing investment in validation and qualification services, growing focus on sterility assurance and stability testing, expansion of contract research and development partnerships. Major trends in the forecast period include adoption of advanced chromatography and spectroscopy instruments, increasing demand for microbiological and sterility testing services, expansion of stability testing and validation services, rising focus on endotoxin and bio burden analysis, growing requirement for on-premises and cloud-based quality control solutions.Global Pharmaceutical Quality Control Market Segmentation
1) By Product: Consumables, Services, Instruments 2) By Deployment Type: Cloud-Based And Web-Based, On-Premises 3) By Analysis Type: Sterility Testing, Membrane Filtration, Direct Inoculation, Bio Burden Testing, Aerobic Count Testing, Anaerobic Count Testing, Spore Count Testing, Fungi or Mold Count Testing, Endotoxin Testing, Other Analysis Types 4) By End-user: Pharmaceutical And Biotechnology Companies, Contract Development and Manufacturing Organization Or Contract Research Organization, Research Laboratories Subsegments: 1) By Consumables: Culture Media, Reagents, Assay Kits, Filters And Membranes, Pipette Tips 2) By Services: Microbiological Testing Services, Analytical Testing Services, Stability Testing Services, Sterility Assurance Services, Validation And Qualification Services 3) By Instruments: Chromatography Systems, Spectroscopy Instruments, Particle Size Analyzers, Potential Of Hydrogen Meters And Electrical Conductivity Meters, Autoclaves And IncubatorsWhat Is The Driver Of The Pharmaceutical Quality Control Market?
The rising demand for biosimilars is expected to propel the growth of the pharmaceutical quality control market going forward. Biosimilars refer to biologic medical products that are highly similar to an already approved reference biologic, with no clinically meaningful differences in terms of safety, purity, and potency. The rising demand for biosimilars is primarily due to the expiration of patents on original biologic drugs, enabling more affordable alternatives to become available in the market. Pharmaceutical quality control ensures the safety, purity, and consistency of biosimilars by rigorously testing them against reference biologics, thereby supporting regulatory compliance and market approval. For instance, in January 2023, Cardinal Health Inc., a US-based healthcare company, reported that as of January 2022, there were 33 FDA-approved biosimilar products in the US, with 21 available on the market. By 2023, this figure had risen to 40 FDA-approved biosimilars, with 25 commercially available. Therefore, the rising demand for biosimilars is driving the growth of the pharmaceutical quality control industry.Key Players In The Global Pharmaceutical Quality Control Market
Major companies operating in the pharmaceutical quality control market are Thermo Fisher Scientific Inc., Abbott Laboratories, Danaher Corporation, Merck KGaA, Becton Dickinson and Company, SGS S.A., Eurofins Scientific SE, WuXi AppTec Co. Ltd., Charles River Laboratories International Inc., PerkinElmer Inc., Mettler-Toledo International Inc., bioMérieux SA, Shimadzu Scientific Instruments Inc., Waters Corporation, Randox Laboratories Ltd., SOLVIAS AG, Esco Micro Pte. Ltd, REMI Elektrotechnik Limited, Lucideon Limited, BRAM-COR S.p.A.Global Pharmaceutical Quality Control Market Trends and Insights
Major companies operating in the pharmaceutical quality control market are focusing on technological advancements such as intelligent software platforms to enhance testing accuracy and regulatory compliance. Intelligent software platforms in pharmaceutical quality control are advanced digital systems that use automation, data analytics, and artificial intelligence (AI) to improve the accuracy, efficiency, and compliance of testing processes. For instance, in June 2025, Dycine Pharmaceuticals Ltd., an India-based pharmaceutical company, launched an artificial intelligence-driven quality control platform designed to enhance manufacturing standards and ensure product integrity. This platform utilizes advanced artificial intelligence algorithms to detect anomalies, monitor real-time data, and streamline compliance processes throughout the pharmaceutical production line. It offers key features such as predictive analytics, automated reporting, and seamless integration with existing manufacturing systems, helping to reduce errors, improve efficiency, and maintain consistent quality across production batches.What Are Latest Mergers And Acquisitions In The Pharmaceutical Quality Control Market?
In October 2024, the United States Pharmacopeial Convention, a US-based independent scientific organization, acquired Stratix Labs for an undisclosed amount. With this acquisition, USP aims to expand its microbiological solutions to reduce microbial contamination risks and address industry gaps in contamination control strategies, thereby ensuring safer and higher-quality pharmaceutical products reach patients. Stratix Labs Corporation is a US-based company that is a provider of advanced microbiological testing solutions.Regional Insights
North America was the largest region in the pharmaceutical quality control market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Pharmaceutical Quality Control Market?
The pharmaceutical quality control market consists of revenues earned by entities by providing services such as analytical testing services, microbiological testing services, stability testing services, sterility assurance services, and validation and qualification services. The market value includes the value of related goods sold by the service provider or included within the service offering. The pharmaceutical quality control market also includes sales of reagents and assay kits, filters and membranes, sterility testing systems, and chromatography columns and consumables. Values in this market are ‘factory gate’ values; that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Pharmaceutical Quality Control Market Report 2026?
The pharmaceutical quality control market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the pharmaceutical quality control industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Pharmaceutical Quality Control Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $10 billion |
| Revenue Forecast In 2035 | $14.59 billion |
| Growth Rate | CAGR of 10.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Deployment Type, Analysis Type, End-user |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Abbott Laboratories, Danaher Corporation, Merck KGaA, Becton Dickinson and Company, SGS S.A., Eurofins Scientific SE, WuXi AppTec Co. Ltd., Charles River Laboratories International Inc., PerkinElmer Inc., Mettler-Toledo International Inc., bioMérieux SA, Shimadzu Scientific Instruments Inc., Waters Corporation, Randox Laboratories Ltd., SOLVIAS AG, Esco Micro Pte. Ltd, REMI Elektrotechnik Limited, Lucideon Limited, BRAM-COR S.p.A. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
