
Pharmaceutical Quality Control Release Testing Service Market Report 2026
Global Outlook – By Product Type (Dissolution Testing, Anti-Corrosion Testing, ICH Stability Storage and Testing, Other Product Types), By Service Type (Analytical Testing Services, Microbiological Testing Services, Stability Testing Services, Sterility Testing Services), By Technology (Gas Chromatography (GC), Mass Spectrometry, Spectroscopy Methods), By End Users (Pharmaceutical Manufacturers, Biotechnology Companies, Contract Development and Manufacturing Organizations (CDMOs), Contract Manufacturing Organizations (CMOs), Research and Academic Institutions) – Market Size, Trends, Strategies, and Forecast to 2035
Pharmaceutical Quality Control Release Testing Service Market Overview
• Pharmaceutical Quality Control Release Testing Service market size has reached to $7.55 billion in 2025 • Expected to grow to $11.01 billion in 2030 at a compound annual growth rate (CAGR) of 7.9% • Growth Driver: Expansion Of Cell Or Gene Therapy Pipelines Driving Market Growth Due To Rising Advanced Therapy Development Demand • Market Trend: Innovations In Rapid Microbiological Methods Enhance Efficiency and Reliability In Pharmaceutical Quality Control Release Testing • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Pharmaceutical Quality Control Release Testing Service Market?
Pharmaceutical quality control release testing service is a specialized service that ensures pharmaceutical products meet predefined quality, safety, and regulatory standards before they are released for commercial distribution. It involves systematic testing and analysis of raw materials, in-process samples, and finished drug products to verify identity, strength, purity, potency, and compliance with regulatory specifications. These services support manufacturers in maintaining product integrity, ensuring patient safety, and meeting global regulatory requirements across complex pharmaceutical production environments. The main product types of pharmaceutical quality control release testing service include dissolution testing, anti-corrosion testing, ICH stability storage and testing, and other product types. Dissolution testing refers to procedures that assess the rate at which active pharmaceutical ingredients (APIs) are released from dosage forms, ensuring proper efficacy and safety. These services are offered as service types including analytical testing services, microbiological testing services, stability testing services, and sterility testing services. They use technologies such as gas chromatography (GC), mass spectrometry (MS), and various spectroscopy methods, and are utilized by end users including pharmaceutical manufacturers, biotechnology companies, contract development and manufacturing organizations (CDMOs), contract manufacturing organizations (CMOs), and research and academic institutions.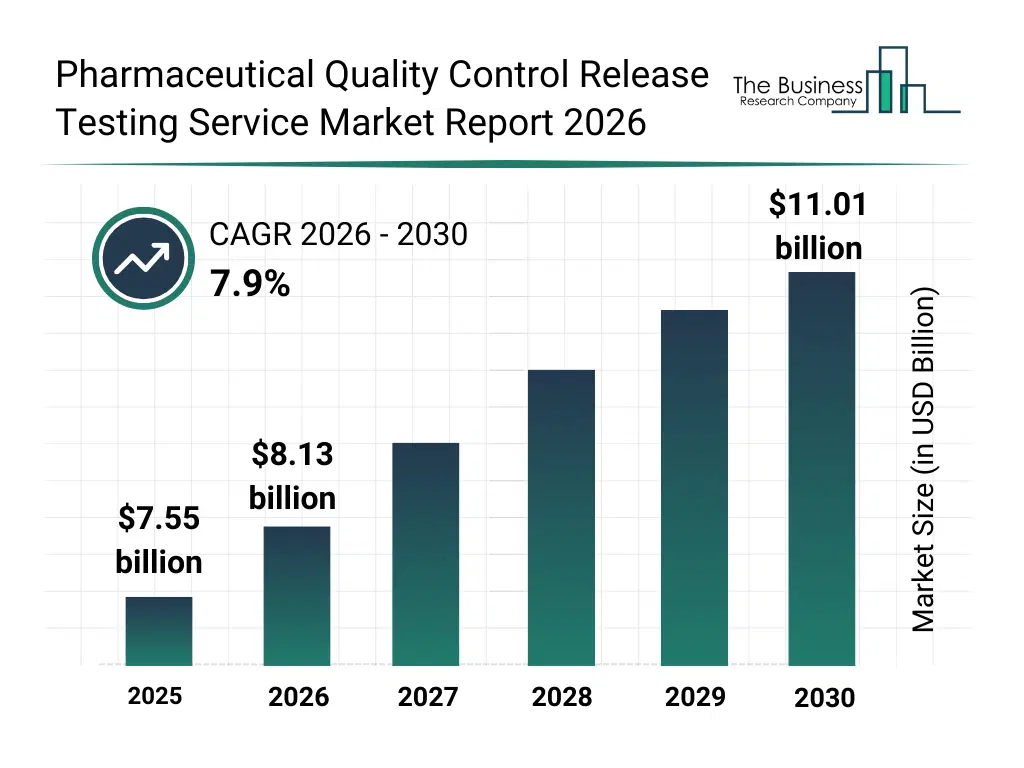
What Is The Pharmaceutical Quality Control Release Testing Service Market Size and Share 2026?
The pharmaceutical quality control release testing service market size has grown strongly in recent years. It will grow from $7.55 billion in 2025 to $8.13 billion in 2026 at a compound annual growth rate (CAGR) of 7.6%. The growth in the historic period can be attributed to stricter gmp regulations, rise in global drug production, growth in contract testing labs, increase in biologics testing needs, expansion of generic drug manufacturing.What Is The Pharmaceutical Quality Control Release Testing Service Market Growth Forecast?
The pharmaceutical quality control release testing service market size is expected to see strong growth in the next few years. It will grow to $11.01 billion in 2030 at a compound annual growth rate (CAGR) of 7.9%. The growth in the forecast period can be attributed to growth in complex biologic pipelines, rising regulatory audit frequency, expansion of outsourced qc models, demand for faster batch release cycles, increased multi region drug approvals. Major trends in the forecast period include automated batch release testing, digital laboratory quality systems, AI assisted analytical validation, connected stability testing labs, smart quality data platforms.Global Pharmaceutical Quality Control Release Testing Service Market Segmentation
1) By Product Type: Dissolution Testing; Anti-Corrosion Testing; ICH Stability Storage and Testing; Other Product Types 2) By Service Type: Analytical Testing Services; Microbiological Testing Services; Stability Testing Services; Sterility Testing Services 3) By Technology: Gas Chromatography (GC); Mass Spectrometry; Spectroscopy Methods 4) By End Users: Pharmaceutical Manufacturers; Biotechnology Companies; Contract Development and Manufacturing Organizations (CDMOs); Contract Manufacturing Organizations (CMOs); Research and Academic Institutions Subsegments: 1) By Dissolution Testing: Immediate Release Testing; Extended Release Testing; Delayed Release Testing; Sink Condition Evaluation 2) By Anti-Corrosion Testing: Packaging Material Corrosion Testing; Container Compatibility Assessment; Environmental Stress Corrosion Testing; Protective Coating Evaluation 3) By ICH Stability Storage and Testing: Long Term Stability Studies; Accelerated Stability Studies; Intermediate Stability Studies; Photostability Testing 4) By Other Product Types: Impurity Profiling; Residual Solvent Testing; Elemental Impurity Analysis; Container Closure Integrity TestingWhat Is The Driver Of The Pharmaceutical Quality Control Release Testing Service Market?
The expansion of cell or gene therapy pipelines is expected to propel the growth of the pharmaceutical quality control release testing service market going forward. The cell and gene therapy pipeline refers to the cellular and genetic therapeutic products under development and regulatory review for commercialization. The expansion is primarily driven by increasing investment in biotechnology research and development, as pharmaceutical companies and biotech firms allocate substantial resources toward innovative cellular and genetic treatments for previously untreatable conditions. The growing cell and gene therapy pipeline directly increases demand for specialized quality control release testing services to ensure these complex biological products meet stringent safety and efficacy standards before commercialization. For instance, in January 2025, according to the American Society of Gene and Cell Therapy, a US-based scientific association, the global gene therapy pipeline reached 2,117 programs in the fourth quarter of 2024, increasing from 2,041 programs in the third quarter of 2024. Therefore, the expansion of cell or gene therapy pipelines is driving the growth of the pharmaceutical quality control release testing service industry.Key Players In The Global Pharmaceutical Quality Control Release Testing Service Market
Major companies operating in the pharmaceutical quality control release testing service market are Labcorp Holdings Inc., SGS S.A., Eurofins Scientific SE, Selvita S.A., WuXi AppTec Co. Ltd., Intertek Group plc, Charles River Laboratories International Inc., Piramal Pharma Limited, Siegfried Holding AG, Almac Group Limited, NSF International, ClinChoice Inc., BioAgilytix Inc., Microbac Laboratories Inc., ARL Bio Pharma Inc., Quality Chemical Laboratories LLC, Neopharm Labs Inc., Butterworth Laboratories Limited, Cambridge Polymer Group Inc., Honeyman Group Limited, and Element Materials Technology Group Limited.Global Pharmaceutical Quality Control Release Testing Service Market Trends and Insights
Major companies operating in the pharmaceutical quality control release testing services market are focusing on developing advanced solutions, such as microbiological-based quality control testing, to ensure product sterility, regulatory compliance, and timely batch release. Microbiological-based quality control testing uses microbiological techniques to monitor and measure microbial presence in pharmaceutical products and production settings, which helps confirm sterility, maintain product safety, and meet regulatory requirements before batch release. For instance, in March 2025, Nelson Laboratories, a US-based microbiological testing company, introduced its Rapid Sterility Testing, a microbiology-based quality control testing offering to modernize sterility assurance processes. It is designed to support faster batch release of sterile pharmaceutical products. The solution shortens traditional fourteen-day incubation timelines to approximately six days and incorporates automated, quantitative data analytics to enhance result precision, reduce manual intervention, and improve overall testing reliability.What Are Latest Mergers And Acquisitions In The Pharmaceutical Quality Control Release Testing Service Market?
In January 2025, Cormica, a UK-based medical device and pharmaceutical testing services company, acquired Focus Laboratories for an undisclosed amount. With this acquisition, Cormica aimed to expand its pharmaceutical microbiological testing capabilities, strengthen quality control and batch release services, and broaden its presence in the U.S. laboratory testing market by integrating Focus Laboratories’ expertise in rapid microbiology and regulatory consulting. Focus Laboratories is a US-based microbiological testing company that specializes in providing microbiological testing services and rapid detection solutions for the pharmaceutical and biotechnology sectors.Regional Insights
North America was the largest region in the pharmaceutical quality control release testing service market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Pharmaceutical Quality Control Release Testing Service Market?
The pharmaceutical quality control release testing service market includes revenues earned by entities by providing services, such as analytical testing, stability testing, microbiological testing, method development and validation, impurity profiling, formulation testing, batch release testing, regulatory compliance support, stability storage services, and pharmacopeial testing. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Pharmaceutical Quality Control Release Testing Service Market Report 2026?
The pharmaceutical quality control release testing service market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the pharmaceutical quality control release testing service industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Pharmaceutical Quality Control Release Testing Service Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $8.13 billion |
| Revenue Forecast In 2035 | $11.01 billion |
| Growth Rate | CAGR of 7.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Service Type, Technology, End Users |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Labcorp Holdings Inc., SGS S.A., Eurofins Scientific SE, Selvita S.A., WuXi AppTec Co. Ltd., Intertek Group plc, Charles River Laboratories International Inc., Piramal Pharma Limited, Siegfried Holding AG, Almac Group Limited, NSF International, ClinChoice Inc., BioAgilytix Inc., Microbac Laboratories Inc., ARL Bio Pharma Inc., Quality Chemical Laboratories LLC, Neopharm Labs Inc., Butterworth Laboratories Limited, Cambridge Polymer Group Inc., Honeyman Group Limited, and Element Materials Technology Group Limited. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
