
Pharmaceutical Rapid Microbiology Testing Market Report 2026
Global Outlook – By Product (Instruments, Reagents And Kits, Accessories), By Technology Type (Polymerase Chain Reaction (PCR), Adenosine Triphosphate (ATP) Bioluminescence, Flow Cytometry, Enzyme-Linked Immunosorbent Assay (ELISA) Chromatography, Biosensors And Bioassays, Impedance Microbiology, Other Technology Types), By Microorganism Type Tested (Bacteria, Fungi (Yeast And Mold), Viruses, Mycoplasma, Endotoxins), By Application (Raw Material Testing, In-Process Testing, Microbial Limit Testing, Bioburden Assessment, Process Water Testing, Environment Monitoring, Sterility Testing), By End-User (Pharmaceutical And Biotechnology Companies, Contract Manufacturing Organizations (CMOs), Hospitals And Diagnostic Laboratories, Research And Academic Institutes) – Market Size, Trends, Strategies, and Forecast to 2035
Pharmaceutical Rapid Microbiology Testing Market Overview
• Pharmaceutical Rapid Microbiology Testing market size has reached to $1.19 billion in 2025 • Expected to grow to $2.65 billion in 2030 at a compound annual growth rate (CAGR) of 17.3% • Growth Driver: Increasing Emphasis On Personalized Medicine Driving Growth In The Market Due To Increasing Demand For Targeted And Safe Therapies • Market Trend: Advancements In Rapid Microbiology Testing For Faster, Compliant Product Release • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Pharmaceutical Rapid Microbiology Testing Market?
Pharmaceutical rapid microbiology testing refers to innovative methods that detect and quantify microorganisms in pharmaceutical products and processes much faster than traditional culture-based techniques, often delivering results in hours instead of days. These rapid tests enhance quality control by enabling quicker decision-making, minimizing product release delays, and reducing the risk of microbial contamination. The main product types of pharmaceutical rapid microbiology testing are instruments, reagents and kits, and accessories. Instruments are specialized laboratory tools that enable accurate, efficient, and standardized microbiological testing for pharmaceutical product safety and quality. It utilizes various technologies, such as polymerase chain reaction (PCR), adenosine triphosphate (ATP) bioluminescence, flow cytometry, enzyme-linked immunosorbent assay (ELISA), chromatography, biosensors and bioassays, impedance microbiology, and others and is used to test several microorganism types, including bacteria, fungi (yeast and mold), viruses, mycoplasma, and endotoxins. It is applied in various applications such as raw material testing, in-process testing, microbial limit testing, bioburden assessment, process water testing, environmental monitoring, and sterility testing and is primarily utilized by several end-users, including pharmaceutical and biotechnology companies, contract manufacturing organizations (CMOs), hospitals and diagnostic laboratories, and research and academic institutions.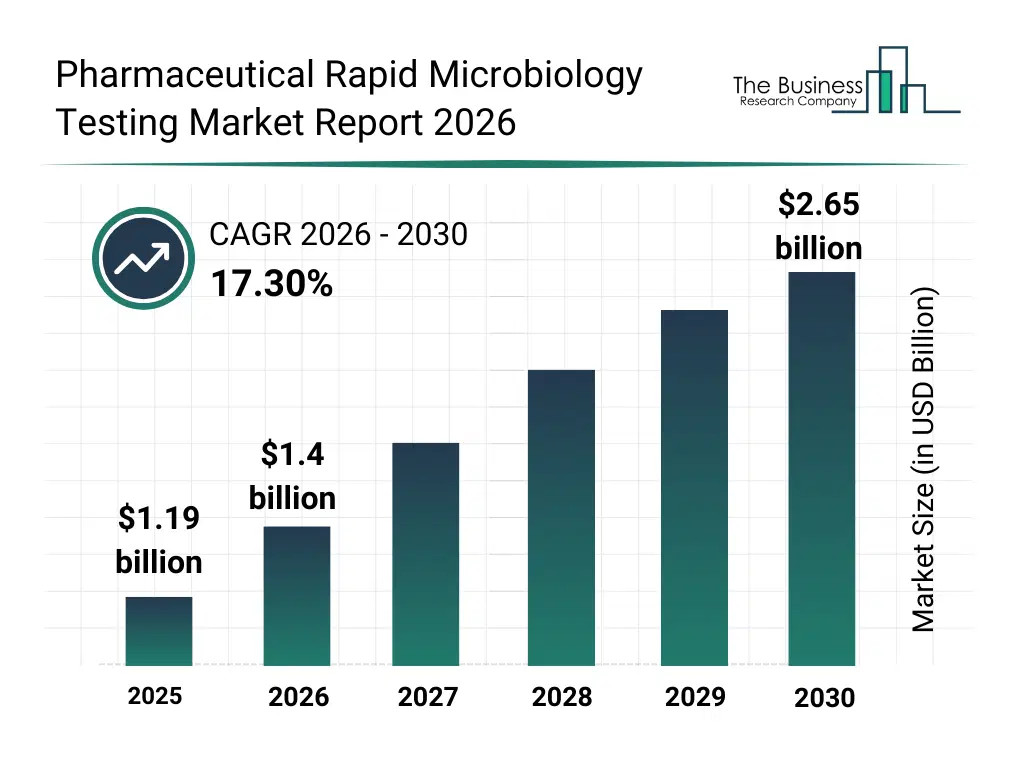
What Is The Pharmaceutical Rapid Microbiology Testing Market Size and Share 2026?
The pharmaceutical rapid microbiology testing market size has grown rapidly in recent years. It will grow from $1.19 billion in 2025 to $1.4 billion in 2026 at a compound annual growth rate (CAGR) of 17.6%. The growth in the historic period can be attributed to reliance on traditional culture-based microbial testing, increasing regulatory requirements for faster product release, growing pharmaceutical manufacturing activities, limited automation in microbiology labs, rising awareness of contamination risks.What Is The Pharmaceutical Rapid Microbiology Testing Market Growth Forecast?
The pharmaceutical rapid microbiology testing market size is expected to see rapid growth in the next few years. It will grow to $2.65 billion in 2030 at a compound annual growth rate (CAGR) of 17.3%. The growth in the forecast period can be attributed to development of automated and high-throughput testing systems, integration of digital data management and cloud-based analytics, rising adoption of rapid testing in biopharmaceutical processes, expansion of cmos and contract testing labs, increasing investments in sterility and quality control innovations. Major trends in the forecast period include adoption of rapid pcr and atp-based microbiology testing, increased use of flow cytometry and elisa for microbial detection, expansion of in-process and bioburden monitoring applications, growing demand for automated microbial detection systems, integration of rapid microbiology testing in quality control and sterility assurance.Global Pharmaceutical Rapid Microbiology Testing Market Segmentation
1) By Product: Instruments, Reagents And Kits, Accessories 2) By Technology Type: Polymerase Chain Reaction (PCR), Adenosine Triphosphate (ATP) Bioluminescence, Flow Cytometry, Enzyme-Linked Immunosorbent Assay (ELISA) Chromatography, Biosensors And Bioassays, Impedance Microbiology, Other Technology Types 3) By Microorganism Type Tested: Bacteria, Fungi (Yeast And Mold), Viruses, Mycoplasma, Endotoxins 4) By Application: Raw Material Testing, In-Process Testing, Microbial Limit Testing, Bioburden Assessment, Process Water Testing, Environment Monitoring, Sterility Testing 5) By End-User: Pharmaceutical And Biotechnology Companies, Contract Manufacturing Organizations (CMOs), Hospitals And Diagnostic Laboratories, Research And Academic Institutes Subsegments: 1) By Instruments: Adenosine Triphosphate (ATP) Bioluminometer, Flow Cytometer, Polymerase Chain Reaction (PCR) Thermocycler Or Real-Time Polymerase Chain Reaction (PCR) System, Automated Microbial Detection Systems, Rapid Immunoassay Analyzers 2) By Reagents And Kits: Adenosine Triphosphate (ATP) Detection Kits, Polymerase Chain Reaction (PCR) Reagents, Enzyme-Linked Immunosorbent Assay (ELISA) Kits, Microbial Culture Media Kits, Fluorescent Staining Kits 3) By Accessories: Microplates Or Test Tubes, Pipettes And Tips, Filters And Membranes, Sample Collection SwabsWhat Is The Driver Of The Pharmaceutical Rapid Microbiology Testing Market?
The increasing emphasis on personalized medicine is expected to propel the growth of the pharmaceutical rapid microbiology testing market going forward. Personalized medicine is a medical approach that tailors healthcare decisions and treatments to an individual's unique genetic makeup, lifestyle, and environmental factors. Personalized medicine is growing due to rising demand for more effective and targeted treatments that reduce trial-and-error in therapy selection. Pharmaceutical rapid microbiology testing supports personalized medicine by enabling faster detection of microbial contaminants, ensuring the safety and sterility of customized therapies tailored to individual patients. For instance, in February 2024, according to the Personalized Medicine Coalition, a US-based non-profit organization, in 2023, the FDA approved 16 new personalized treatments for rare disease patients, up from six in 2022. Therefore, the increasing emphasis on personalized medicine is driving the growth of the pharmaceutical rapid microbiology testing industry.Key Players In The Global Pharmaceutical Rapid Microbiology Testing Market
Major companies operating in the pharmaceutical rapid microbiology testing market are Merck KGaA, Becton Dickinson and Company, SGS SA, Eurofins Scientific SE, Charles River Laboratories International Inc, ALS Limited, WESSLING GmbH, Nelson Labs LLC, Labor LS SE & Co. KG, GBA Pharma GmbH, ANACON LABORATORIES PRIVATE LIMITED, Vimta Labs Limited, Pacific Biolabs Inc., Wickham Laboratories, Microchem Laboratory Inc., Infinity Laboratories Private Limited, Ghaziabad Testing Laboratories Private Limited, Medistri SA, PREWEL LABS PRIVATE LIMITED, TUV SUD SOUTH ASIA PRIVATE LIMITEDGlobal Pharmaceutical Rapid Microbiology Testing Market Trends and Insights
Major companies operating in the pharmaceutical rapid microbiology testing market are focusing on developing advanced advancements, such as adenosine triphosphate (ATP) bioluminescence, to support faster product release in compliance with regulatory standards. Adenosine triphosphate (ATP) bioluminescence is a rapid testing method that detects living microorganisms by measuring ATP levels using a luciferase-driven light reaction, with light intensity indicating microbial contamination in pharmaceuticals, medical devices, and other materials. For instance, in March 2025, Nelson Labs LLC, a US-based health company, launched a rapid-method sterility testing service. This innovative pharmaceutical rapid microbiology testing approach features accelerated detection times, automated result analysis, and high-throughput capabilities. Rapid sterility testing is highly versatile, supporting a wide range of pharmaceutical and medical device products by significantly reducing testing times while maintaining compliance with regulatory standards.What Are Latest Mergers And Acquisitions In The Pharmaceutical Rapid Microbiology Testing Market?
In January 2025, Cormica Ltd., a UK-based company that offers medical device and pharmaceutical testing services, acquired Focus Laboratories Inc. for an undisclosed amount. Through this acquisition, Cormica aims to strengthen its position in the laboratory testing sector and expand its service offerings with advanced, high-quality testing solutions for pharmaceutical and healthcare clients. Focus Laboratories Inc. is a US-based pharmaceutical company that offers rapid microbiology testing for pharmaceuticals.Regional Insights
North America was the largest region in the pharmaceutical rapid microbiology testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Pharmaceutical Rapid Microbiology Testing Market?
The pharmaceutical rapid microbiology testing market consists of revenues earned by entities providing services such as microbial contamination detection, identification and enumeration, and sterility testing. The market value includes the value of related goods sold by the service provider or included within the service offering. The pharmaceutical rapid microbiology testing market also includes sales of rapid sterility testing kits, endotoxin detection kits, bioburden testing systems, environmental monitoring tools and molecular testing kits. Values in this market are ‘factory gate’ values; that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Pharmaceutical Rapid Microbiology Testing Market Report 2026?
The pharmaceutical rapid microbiology testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the pharmaceutical rapid microbiology testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Pharmaceutical Rapid Microbiology Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.4 billion |
| Revenue Forecast In 2035 | $2.65 billion |
| Growth Rate | CAGR of 17.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Technology Type, Microorganism Type Tested, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Merck KGaA, Becton Dickinson and Company, SGS SA, Eurofins Scientific SE, Charles River Laboratories International Inc, ALS Limited, WESSLING GmbH, Nelson Labs LLC, Labor LS SE & Co. KG, GBA Pharma GmbH, ANACON LABORATORIES PRIVATE LIMITED, Vimta Labs Limited, Pacific Biolabs Inc., Wickham Laboratories, Microchem Laboratory Inc., Infinity Laboratories Private Limited, Ghaziabad Testing Laboratories Private Limited, Medistri SA, PREWEL LABS PRIVATE LIMITED, TUV SUD SOUTH ASIA PRIVATE LIMITED |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
