
Pharmaceutical Safety Assessment Market Report 2026
Global Outlook – By Type (In-Vitro Safety Assessment, In-Vivo Safety Assessment, Computational Safety Assessment), By Drug Type (Biological Drugs, Chemical Drugs), By Phase (Preclinical, Clinical (Phase I, II, III), Post-Market Surveillance (Phase IV)), By End User (Pharmaceutical Companies, Biotechnology Companies, Medical Device Companies, Academic And Research Institutions, Contract Research Organizations (CROs)) – Market Size, Trends, Strategies, and Forecast to 2035
Pharmaceutical Safety Assessment Market Overview
• Pharmaceutical Safety Assessment market size has reached to $11.32 billion in 2025 • Expected to grow to $16.97 billion in 2030 at a compound annual growth rate (CAGR) of 8.5% • Growth Driver: Expansion Of Clinical Trials Is Fueling Market Growth Due To Increasing Demand For Drug Safety Assessment • Market Trend: AI-Driven Platforms Enhancing Accuracy And Efficiency In Toxicity Prediction • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Pharmaceutical Safety Assessment Market?
Pharmaceutical safety assessment is the systematic evaluation of drugs and pharmaceutical products to ensure their safety and minimize potential risks to patients. It involves analyzing preclinical and clinical data, monitoring adverse effects, and ensuring compliance with regulatory standards. This process helps in making informed decisions regarding the approval, labeling, and safe use of medications. The main types in the pharmaceutical safety assessment market are in-vitro safety assessment, in-vivo safety assessment, and computational safety assessment. In-vitro safety assessment refers to the evaluation of the safety and toxicity of drugs, chemicals, or other substances using controlled laboratory tests performed outside of a living organism. The various types of drugs include biological drugs and chemical drugs. It spans multiple phases, such as preclinical, clinical (phase I, II, III), and post-market surveillance (phase IV). Key end users include pharmaceutical companies, biotechnology firms, medical device manufacturers, academic and research institutions, and contract research organizations (CROs).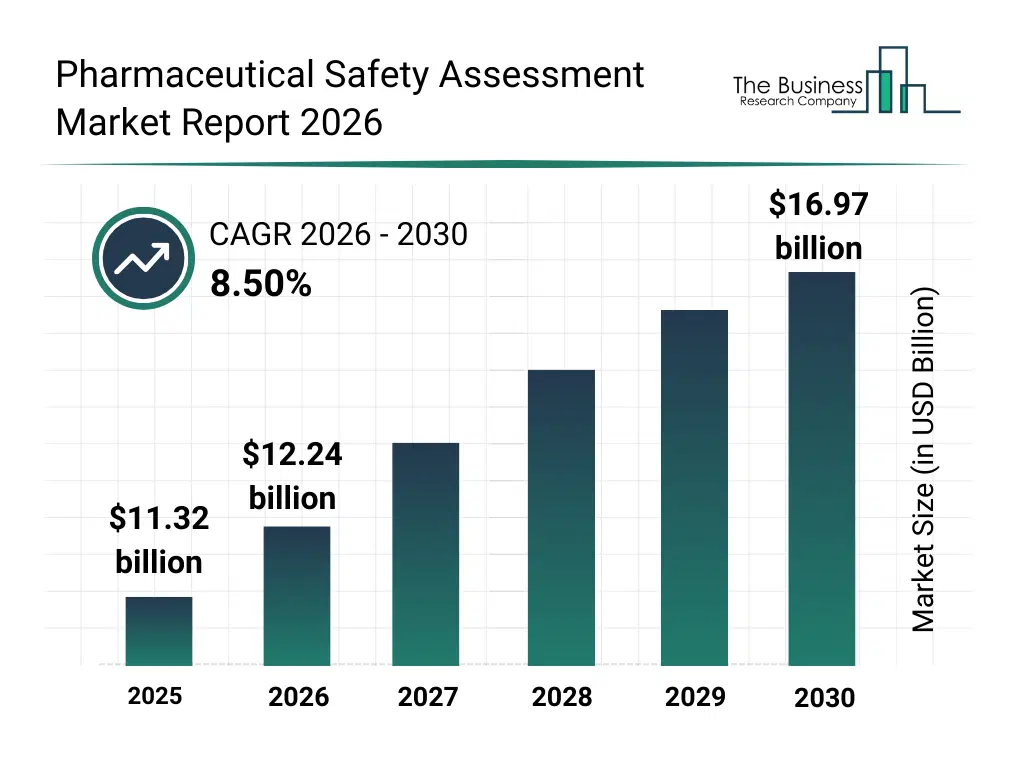
What Is The Pharmaceutical Safety Assessment Market Size and Share 2026?
The pharmaceutical safety assessment market size has grown strongly in recent years. It will grow from $11.32 billion in 2025 to $12.24 billion in 2026 at a compound annual growth rate (CAGR) of 8.2%. The growth in the historic period can be attributed to expansion of pharmaceutical R&D pipelines, increasing regulatory scrutiny on drug safety, growth in preclinical and clinical testing volumes, wider adoption of in-vitro safety assessment methods, rising use of CRO-led safety studies.What Is The Pharmaceutical Safety Assessment Market Growth Forecast?
The pharmaceutical safety assessment market size is expected to see strong growth in the next few years. It will grow to $16.97 billion in 2030 at a compound annual growth rate (CAGR) of 8.5%. The growth in the forecast period can be attributed to increasing reliance on computational safety assessment, expansion of personalized medicine development, rising demand for faster regulatory approvals, growing integration of ai in drug safety workflows, increased post-market surveillance requirements. Major trends in the forecast period include increasing use of ai-based toxicity prediction tools, rising adoption of in-silico safety assessment models, growing integration of real-time pharmacovigilance systems, expansion of automated preclinical testing platforms, enhanced focus on regulatory-compliant safety analytics.Global Pharmaceutical Safety Assessment Market Segmentation
1) By Type: In-Vitro Safety Assessment, In-Vivo Safety Assessment, Computational Safety Assessment 2) By Drug Type: Biological Drugs, Chemical Drugs 3) By Phase: Preclinical, Clinical (Phase I, II, III), Post-Market Surveillance (Phase IV) 4) By End User: Pharmaceutical Companies, Biotechnology Companies, Medical Device Companies, Academic And Research Institutions, Contract Research Organizations (CROs) Subsegments: 1) By In-Vitro Safety Assessment: Cell-Based Assays, Organotypic Models, Tissue Culture Models, High-Throughput Screening 2) By In-Vivo Safety Assessment: Rodent Studies, Non-Rodent Studies, Toxicokinetic Studies, Carcinogenicity Studies 3) By Computational Safety Assessment: Quantitative Structure-Activity Relationship (QSAR) Modeling, Physiologically Based Pharmacokinetic (PBPK) Modeling, Toxicity Prediction Software, In Silico Simulation StudiesWhat Is The Driver Of The Pharmaceutical Safety Assessment Market?
The expansion of clinical trials is expected to propel the growth of the pharmaceutical safety assessment market going forward. Clinical trials refer to research studies conducted in humans to evaluate the safety, effectiveness, and side effects of new medical treatments, drugs, or interventions. Clinical trials are rising due to the growing need for new and effective treatments, as researchers and healthcare providers aim to develop therapies that can better prevent, manage, or cure diseases. Clinical trials support pharmaceutical safety assessment by systematically evaluating a drug’s effects on humans through carefully controlled studies, monitoring for adverse reactions, determining safe dosage ranges, and providing essential data that regulatory authorities use to ensure that medications are both effective and safe for public use. For instance, in September 2025, according to the National Library of Medicine (NLM), a US-based biomedical library, as of September 26, 2025, a total of 554,798 studies were registered on ClinicalTrials.gov. Therefore, the expansion of clinical trials is driving the growth of the pharmaceutical safety assessment industry.Key Players In The Global Pharmaceutical Safety Assessment Market
Major companies operating in the pharmaceutical safety assessment market are IQVIA Holding Inc., LabCorp (formerly Covance), Thermo Fisher Scientific (PPD), Charles River Laboratories, ICON plc, Parexel International Corp., Syneos Health, Eurofins Scientific SE, Wuxi Biologics Inc., SGS SA, Medpace Holdings Inc., Fujifilm Wako (Fujifilm Corp.), Pharmaron Beijing Co. Ltd., Inotiv (incl. Envigo), Evotec (Cyprotex), Intertek Group, Altasciences Inc., Aragen Life Sciences Ltd., CTI Clinical Trial and Consulting Inc., ChemDiv Inc., Frontage Laboratories, Medicilon Inc., Biotoxtech Co. Ltd., BioIVT, Accelera Srl.Global Pharmaceutical Safety Assessment Market Trends and Insights
Major companies in the pharmaceutical safety assessment market are focusing on developing AI-powered decision support tools to enhance the accuracy and efficiency of toxicity prediction and safety profiling. An AI-powered decision support tool is a software system that uses artificial intelligence to analyze data and provide insights that support faster and more accurate decision-making. For instance, in April 2024, Charles River Laboratories, a US-based life sciences company, in partnership with Deciphex, an Ireland-based company enhances diagnostic precision and speed through AI-driven quality assurance, launched the Patholytix Foresight platform, an AI-powered decision support tool, designed to transform toxicologic pathology in pharmaceutical safety assessment. The platform uses artificial intelligence to quickly analyze complex preclinical pathology data, helping identify potential safety risks earlier and reducing subjectivity in results. It offers unique features such as an AI algorithm that detects tissue abnormalities, a cloud-based system for global collaboration, and a scalable design that cuts down slide review time while improving data accuracy and consistency.What Are Latest Mergers And Acquisitions In The Pharmaceutical Safety Assessment Market?
In January 2025, Groupe ProductLife S.A.S., a France-based life sciences services company, acquired Stragen Services SAS for an undisclosed amount. With this acquisition, ProductLife Group aims to enhance its pharmacovigilance and clinical safety capabilities, expand its European market presence, and provide more comprehensive regulatory and safety support to pharmaceutical and biotech clients. Stragen Services SAS is a France-based company that specializes in pharmaceutical safety assessment, also known as pharmacovigilance.Regional Insights
North America was the largest region in the pharmaceutical safety assessment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Pharmaceutical Safety Assessment Market?
The pharmaceutical safety assessment market includes revenues earned by entities by providing services such as post-marketing surveillance, safety pharmacology, regulatory consulting and compliance, bioanalytics and biomarker services, and in silico and computational toxicology. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Pharmaceutical Safety Assessment Market Report 2026?
The pharmaceutical safety assessment market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the pharmaceutical safety assessment industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Pharmaceutical Safety Assessment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $12.24 billion |
| Revenue Forecast In 2035 | $16.97 billion |
| Growth Rate | CAGR of 8.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Drug Type, Phase, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | IQVIA Holding Inc., LabCorp (formerly Covance), Thermo Fisher Scientific (PPD), Charles River Laboratories, ICON plc, Parexel International Corp., Syneos Health, Eurofins Scientific SE, Wuxi Biologics Inc., SGS SA, Medpace Holdings Inc., Fujifilm Wako (Fujifilm Corp.), Pharmaron Beijing Co. Ltd., Inotiv (incl. Envigo), Evotec (Cyprotex), Intertek Group, Altasciences Inc., Aragen Life Sciences Ltd., CTI Clinical Trial and Consulting Inc., ChemDiv Inc., Frontage Laboratories, Medicilon Inc., Biotoxtech Co. Ltd., BioIVT, Accelera Srl. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
