
Pharmaceutical Stability And Storage Services Market Report 2026
Global Outlook – By Service (Stability, Storage), By Product Type (Tablets, Capsules, Injectable Solutions, Oral Suspensions, Combination Products, Other Product Types), By Molecule (Small Molecule, Large Molecule), By Mode (In-House, Outsourcing), By End User (Biopharmaceutical Companies, Contract Manufacturing Organizations, Contract Research Organizations, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Pharmaceutical Stability And Storage Services Market Overview
• Pharmaceutical Stability And Storage Services market size has reached to $2.81 billion in 2025 • Expected to grow to $3.78 billion in 2030 at a compound annual growth rate (CAGR) of 6.1% • Growth Driver: Rise In Novel Drug Approvals Driving The Growth Of The Market Due To Increasing Demand For Regulatory Compliance And Shelf-Life Management • Market Trend: Advancements In Cryogenic Storage Enhancing Long-Term Preservation Of Biological Materials • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Pharmaceutical Stability And Storage Services Market?
Pharmaceutical stability and storage services refer to specialized services to ensure that pharmaceutical products (such as drugs, medications, and vaccines) maintain their quality, efficacy, and safety over time. These services include controlled environments for storing drugs, conducting stability studies to monitor chemical, physical, and microbiological changes, and ensuring compliance with regulatory standards. The main pharmaceutical stability and storage services include stability and storage. Stability services assess how pharmaceutical products maintain quality, potency, and safety under different environmental conditions over time. It is used for various products such as tablets, capsules, injectable solutions, oral suspensions, combination products, and others, and it can be both a small and a large molecule. It is provided through in-house and outsourcing modes and is used by biopharmaceutical companies, contract manufacturing organizations, contract research organizations, and others.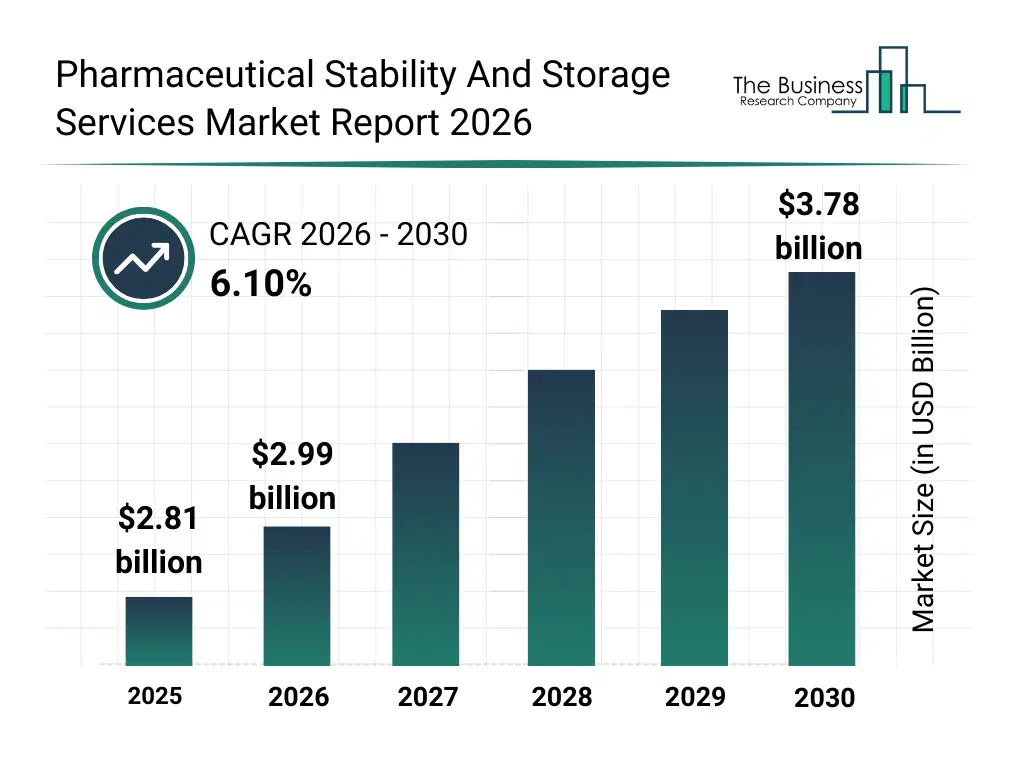
What Is The Pharmaceutical Stability And Storage Services Market Size and Share 2026?
The pharmaceutical stability and storage services market size has grown strongly in recent years. It will grow from $2.81 billion in 2025 to $2.99 billion in 2026 at a compound annual growth rate (CAGR) of 6.1%. The growth in the historic period can be attributed to strict pharmaceutical regulatory requirements, growth in global drug production, expansion of biologics manufacturing, need for shelf-life validation, increasing clinical trial volumes.What Is The Pharmaceutical Stability And Storage Services Market Growth Forecast?
The pharmaceutical stability and storage services market size is expected to see strong growth in the next few years. It will grow to $3.78 billion in 2030 at a compound annual growth rate (CAGR) of 6.1%. The growth in the forecast period can be attributed to growth in biologics and cell therapies, increasing pharmaceutical outsourcing, expansion of global cold chain infrastructure, rising vaccine production, stronger regulatory scrutiny on drug stability. Major trends in the forecast period include rising demand for regulatory-compliant storage, increasing outsourcing of stability testing services, growing need for cold chain and cryogenic storage, expansion of stability services for biologics, higher focus on long-term drug quality assurance.Global Pharmaceutical Stability And Storage Services Market Segmentation
1) By Service: Stability, Storage 2) By Product Type: Tablets, Capsules, Injectable Solutions, Oral Suspensions, Combination Products, Other Product Types 3) By Molecule: Small Molecule, Large Molecule 4) By Mode: In-House, Outsourcing 5) By End User: Biopharmaceutical Companies, Contract Manufacturing Organizations, Contract Research Organizations, Other End Users Subsegments: 1) By Stability Services: Long-Term Stability Studies, Accelerated Stability Studies, Intermediate Stability Studies, Photostability Testing, Forced Degradation Studies 2) By Storage Services: Ambient Storage, Refrigerated Storage (2°C to 8°C), Frozen Storage (-20°C to -80°C), Cryogenic Storage (-150°C to -196°C), Controlled Drug Substance And Product StorageWhat Is The Driver Of The Pharmaceutical Stability And Storage Services Market?
The increasing number of novel drug approvals is expected to propel the growth of the pharmaceutical stability and storage services market going forward. A novel drug refers to a pharmaceutical compound that is newly developed and has never been previously approved for use or marketed. The increasing number of novel drug approvals is due to advancements in biotechnology, enabling the discovery of new compounds and improved regulatory processes that facilitate faster, more efficient drug development. The rise in novel drug approvals drives demand for pharmaceutical stability and storage services, ensuring optimal conditions, compliance, and extended shelf-life management. For instance, in October 2023, according to the U.S. Food and Drug Administration (FDA), a US-based federal agency responsible for protecting public health, in 2022, the FDA approved 37 novel drugs, followed by 50 in 2024, advancing treatments across cancer, rare diseases, neurology, infections, and conditions like schizophrenia, pain, and Alzheimer's. Therefore, the increasing number of novel drug approvals drive the growth of the pharmaceutical stability and storage services industry.Key Players In The Global Pharmaceutical Stability And Storage Services Market
Major companies operating in the pharmaceutical stability and storage services market are SGS S.A., Eurofins Scientific SE, Intertek Group plc, Catalent Inc., Almac Group, Alcami Corporation, Reading Scientific Services Ltd., Charles River Laboratories, Element Materials Technology Inc., Precision Stability Storage, Roylance Stability Storage Limited, BioLife Solutions Inc., Alloga UK Ltd., Auriga Research Private Limited, Q Laboratories Inc., ALS Limited, Lucideon Limited, Cencora Inc., PCI Pharma Services, DHL Life Sciences & HealthcareGlobal Pharmaceutical Stability And Storage Services Market Trends and Insights
Major companies operating in the pharmaceutical stability and storage services market are focusing on developing technologically advanced solutions, such as cryogenic storage suites, to enhance the preservation of temperature-sensitive pharmaceutical products. Cryogenic storage suites are specialized storage facilities that maintain extremely low temperatures, typically below -150°C, to preserve temperature-sensitive biological and pharmaceutical materials such as cell therapies, vaccines, and biologics. For instance, in December 2024, Astoriom, a UK-based stability and biospecimen storage industry, launched a cryogenic storage suite at its Tramore facility in County Waterford, Ireland, marking a significant expansion in its capabilities for biological sample storage. This state-of-the-art suite is designed to meet the increasing demand for safe, long-term preservation of biological materials, ensuring their viability over extended periods. This storage features precise temperature control down to -196°C, vast storage capacity, ISO 9001 certification for regulatory compliance, and advanced security measures, including access controls and monitoring systems.What Are Latest Mergers And Acquisitions In The Pharmaceutical Stability And Storage Services Market?
In January 2024, Alcami Corporation, a US-based contract development and manufacturing organization (CDMO), acquired Pacific Pharmaceutical Services, Inc. for an undisclosed amount. With this acquisition, Alcami aims to expand its pharma storage and services footprint, enhance bi-coastal service coverage, and strengthen its ability to support pre-clinical and clinical material management with improved security and multi-location resilience. Pacific Pharmaceutical Services, Inc. is a US-based pharmaceutical clinical distribution services company that offers pharmaceutical stability and storage services, including various cGMP-compliant storage conditions from controlled room temperature to cryo temperatures.Regional Insights
North America was the largest region in the pharmaceutical stability and storage services market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Pharmaceutical Stability And Storage Services Market?
The pharmaceutical stability and storage services market consists of revenues earned by entities by providing services such as photostability testing, cold chain management, disaster recovery and backup storage. The market value includes the value of related goods sold by the service provider or included within the service offering. The pharmaceutical stability and storage services market also includes sales of stability chambers, cryogenic storage systems, cold storage units and automated sample management systems. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Pharmaceutical Stability And Storage Services Market Report 2026?
The pharmaceutical stability and storage services market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the pharmaceutical stability and storage services industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Pharmaceutical Stability And Storage Services Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.99 billion |
| Revenue Forecast In 2035 | $3.78 billion |
| Growth Rate | CAGR of 6.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Service, Product Type, Molecule, Mode, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | SGS S.A., Eurofins Scientific SE, Intertek Group plc, Catalent Inc., Almac Group, Alcami Corporation, Reading Scientific Services Ltd., Charles River Laboratories, Element Materials Technology Inc., Precision Stability Storage, Roylance Stability Storage Limited, BioLife Solutions Inc., Alloga UK Ltd., Auriga Research Private Limited, Q Laboratories Inc., ALS Limited, Lucideon Limited, Cencora Inc., PCI Pharma Services, DHL Life Sciences & Healthcare |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
