
Pharmaceutical Sterility Testing Market Report 2026
Global Outlook – By Sample (Sterile Drugs, Medical Devices, Biologics and Therapeutics), By Product Type (Instruments, Kits and Reagents, Services), By Service Type (In- house, Outsourcing), By Test Type (Sterility Testing, Bioburden Testing, Bacterial Endotoxin Testing), By End- User (Compounding Pharmacies, Medical Devices Companies, Pharmaceutical Companies) – Market Size, Trends, Strategies, and Forecast to 2035
Pharmaceutical Sterility Testing Market Overview
• Pharmaceutical Sterility Testing market size has reached to $1.68 billion in 2025 • Expected to grow to $4.91 billion in 2030 at a compound annual growth rate (CAGR) of 24.1% • Growth Driver: Rise In Biologics Sales Are Anticipated To Fuel The Growth Of The Pharmaceutical Sterility Testing Market • Market Trend: Advancements In Rapid Biological Indicator Sterility Testing For Enhanced Pharmaceutical Compliance And Efficiency • North America was the largest region in 2025 and Middle East is the fastest growing region.What Is Covered Under Pharmaceutical Sterility Testing Market?
Pharmaceutical sterility testing refers to a test that is intended to show if biological parenteral made for human use contain extraneous, viable contaminating microorganisms or not. All pharmaceutical items used on patients by medical professionals must be of the highest quality and extremely safe. Otherwise, it could harm the patients. Testing for sterility assures that the offered product is suitably sterile. The main types of pharmaceutical sterility testing are in-house and outsourced. The various types of tests include sterility testing, bioburden testing, and bacterial endotoxin testing, which are used to test sterile drugs, medical devices, biologics, and therapeutics. An in vitro assay for detecting bacterial endotoxins is known as a bacterial endotoxin test. The bacterial endotoxin test detects bacterial endotoxins by lysing blood cells from horseshoe crabs. The different products used are instruments, kits reagents, and services.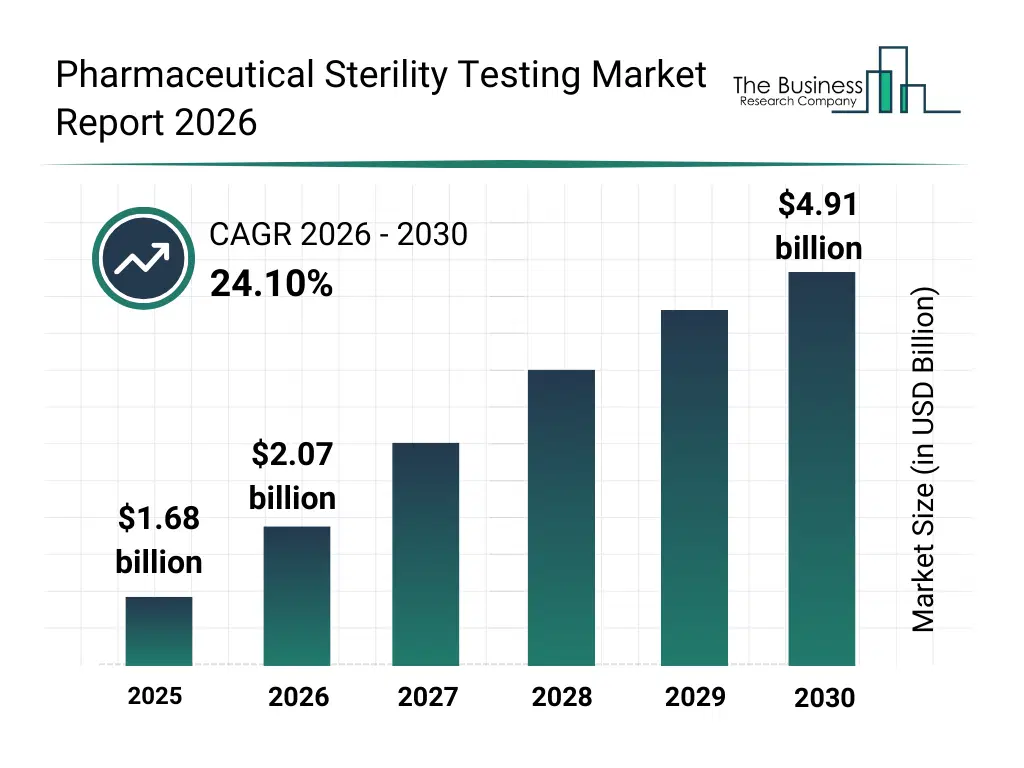
What Is The Pharmaceutical Sterility Testing Market Size and Share 2026?
The pharmaceutical sterility testing market size has grown exponentially in recent years. It will grow from $1.68 billion in 2025 to $2.07 billion in 2026 at a compound annual growth rate (CAGR) of 23.7%. The growth in the historic period can be attributed to increasing need for sterile pharmaceutical products, limited availability of advanced testing laboratories, growing pharmaceutical production and biologics pipelines, reliance on conventional sterility testing methods, rising awareness of quality control standards.What Is The Pharmaceutical Sterility Testing Market Growth Forecast?
The pharmaceutical sterility testing market size is expected to see exponential growth in the next few years. It will grow to $4.91 billion in 2030 at a compound annual growth rate (CAGR) of 24.1%. The growth in the forecast period can be attributed to advancements in automated sterility testing technologies, expansion of outsourcing testing services, integration of ai and digital monitoring tools, increasing adoption of rapid microbiological methods, growing regulatory mandates for biologics and gene therapies. Major trends in the forecast period include rising demand for in-house and outsourced sterility testing services, growing adoption of bioburden and endotoxin testing in pharmaceutical manufacturing, increasing use of advanced instruments, kits, and reagents for sterility analysis, expansion of testing services for biologics, vaccines, gene, and cell therapies, enhanced focus on regulatory compliance and quality assurance in sterility testing.Global Pharmaceutical Sterility Testing Market Segmentation
1) By Sample: Sterile Drugs, Medical Devices, Biologics and Therapeutics 2) By Product Type: Instruments, Kits and Reagents, Services 3) By Service Type: In- house, Outsourcing 4) By Test Type: Sterility Testing, Bioburden Testing, Bacterial Endotoxin Testing 5) By End- User: Compounding Pharmacies, Medical Devices Companies, Pharmaceutical Companies Subsegments: 1) By Sterile Drugs: Injectable Drugs, Infusion Solutions, Ophthalmic Solutions 2) By Medical Devices: Surgical Instruments, Implants, Catheters 3) By Biologics And Therapeutics: Vaccines, Gene Therapies, Cell TherapiesWhat Is The Driver Of The Pharmaceutical Sterility Testing Market?
The rise in biologics sales is expected to propel the pharmaceutical sterility testing market. Biologics, or biological products refers to medicines derived from living organisms, including a variety of products such as vaccines, blood and blood components, allergenics, somatic cells, gene therapies, tissues, and recombinant therapeutic proteins. Pharmaceutical sterility testing provides the necessary quality control to confirm that biologics are free from microbial contamination before they are administered to patients. For instance, in October 2024, according to the Generics and Biosimilars Initiative, a Belgium-based organization, that provides high-quality, scientific information on generic and biosimilar medicines, beyond the nine leading biologics, two additional complex drugs recorded significant sales in 2023. Novo Nordisk’s Ozempic (semaglutide) and Eli Lilly’s Trulicity (dulaglutide), both prescribed for type 2 diabetes, achieved sales of US$14 billion and US$7.2 billion, respectively. Therefore, the rise in biologics sales is driving the pharmaceutical sterility testing industry.Key Players In The Global Pharmaceutical Sterility Testing Market
Major companies operating in the pharmaceutical sterility testing market are Pacific Biolabs Inc., STERIS Corporation, Boston Analytical Inc., Gibraltar Laboratories Inc., Sartorius AG, Solvias AG, SGS S.A., Toxikon Inc., Pace Analytical Services LLC, Charles River Laboratories International Inc., Nelson Laboratories Inc., Rapid Micro Biosystems Inc., bioMérieux Inc., Merck KGaA, WuXi AppTec, Sotera Health LLC, Dynalabs LLC, Infinity Laboratories, Eurofins Scientific SE, Avomeen Analytical Services, Microbac Laboratories Inc., Analytical Lab Group LLC, North American Science Associates Inc., Medical Device Testing Services Inc., Microtest Laboratories Inc., BioScreen Testing Services Inc., Microchem Laboratory Inc., Bactolac Pharmaceutical Inc., Micro Quality Labs Inc., BioPharma Services Inc., Microbiology Research Associates, BioScience Laboratories Inc.Global Pharmaceutical Sterility Testing Market Trends and Insights
Major companies operating in the pharmaceutical sterility testing market are focusing on developing innovative solutions such as Advanced biological indicators to accelerate sterility confirmation, improve testing accuracy, and enhance overall manufacturing safety. Advanced biological indicators are specialized tools containing highly resistant microorganisms that rapidly and accurately verify the effectiveness of sterilization processes in pharmaceutical manufacturing. For instance, in September 2025, Nelson Labs, a US-based contract testing laboratory, launched the RapidCert Biological Indicator (BI) sterility testing platform. This new system significantly shortens the time required to confirm sterility, enhancing efficiency in pharmaceutical manufacturing and supporting faster product release while maintaining rigorous quality standards.What Are Latest Mergers And Acquisitions In The Pharmaceutical Sterility Testing Market?
In September 2024, Eurofins Scientific SE, a Luxembourg -based global leader in bio-analytical testing, acquired Infinity Laboratories, LLC, for an undisclosed amount. With this acquisition, Eurofins aims to expand its laboratory services capabilities, enhance testing portfolio offerings, strengthen regional presence in the US, and accelerate service delivery to clients. Infinity Laboratories, LLC, is a US-based contract laboratory specializing in pharmaceutical sterility testing.Regional Insights
North America was the largest region in the pharmaceutical sterility testing market in 2025. The Middle East is expected to be the fastest-growing region in the global pharmaceutical sterility testing market share during the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Pharmaceutical Sterility Testing Market?
The pharmaceutical sterility testing market includes revenues earned by entities by membrane filtration, direct transfer (product immersion), and product flush. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Pharmaceutical Sterility Testing Market Report 2026?
The pharmaceutical sterility testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the pharmaceutical sterility testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Pharmaceutical Sterility Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.07 billion |
| Revenue Forecast In 2035 | $4.91 billion |
| Growth Rate | CAGR of 23.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Sample, Product Type, Service Type, Test Type, End- User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Pacific Biolabs Inc., STERIS Corporation, Boston Analytical Inc., Gibraltar Laboratories Inc., Sartorius AG, Solvias AG, SGS S.A., Toxikon Inc., Pace Analytical Services LLC, Charles River Laboratories International Inc., Nelson Laboratories Inc., Rapid Micro Biosystems Inc., bioMérieux Inc., Merck KGaA, WuXi AppTec, Sotera Health LLC, Dynalabs LLC, Infinity Laboratories, Eurofins Scientific SE, Avomeen Analytical Services, Microbac Laboratories Inc., Analytical Lab Group LLC, North American Science Associates Inc., Medical Device Testing Services Inc., Microtest Laboratories Inc., BioScreen Testing Services Inc., Microchem Laboratory Inc., Bactolac Pharmaceutical Inc., Micro Quality Labs Inc., BioPharma Services Inc., Microbiology Research Associates, BioScience Laboratories Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
