
Pharmacovigilance And Drug Safety Software Market Report 2026
Global Outlook – By Software Type (Adverse Event Reporting Software, Drug Safety Audits Software, Issue Tracking Software, Fully Integrated Software), By Delivery Mode (On-premise, Cloud-based), By End User (Pharmaceutical And Biotechnology Companies, Contract Research Organizations, Business Process Outsourcing Firms, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Pharmacovigilance And Drug Safety Software Market Overview
• Pharmacovigilance And Drug Safety Software market size has reached to $2.53 billion in 2025 • Expected to grow to $4.6 billion in 2030 at a compound annual growth rate (CAGR) of 12.6% • Growth Driver: Increasing Adoption Of Personalized Medicine Drives Growth In The Pharmacovigilance And Drug Safety Software Market • Market Trend: Cloud-Based Data Lake Platforms Revolutionizing Pharmacovigilance And Drug Safety Software Market • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Pharmacovigilance And Drug Safety Software Market?
Pharmacovigilance and drug safety software refers to a suite of digital tools designed to monitor, analyze, and report adverse drug reactions (ADRs) and other safety-related information associated with pharmaceuticals and medical products. This software plays a critical role in ensuring patient safety and regulatory compliance throughout the lifecycle of a drug, from development to post-marketing surveillance. The main software types of pharmacovigilance and drug safety software are adverse event reporting software, drug safety audit software, issue tracking software, and fully integrated software. Adverse event reporting software is designed to track, document, and manage adverse events related to pharmaceutical products or medical devices. The delivery modes are on-premise and cloud-based and used by various end users, such as pharmaceutical and biotechnology companies, contract research organizations, business process outsourcing firms, and others.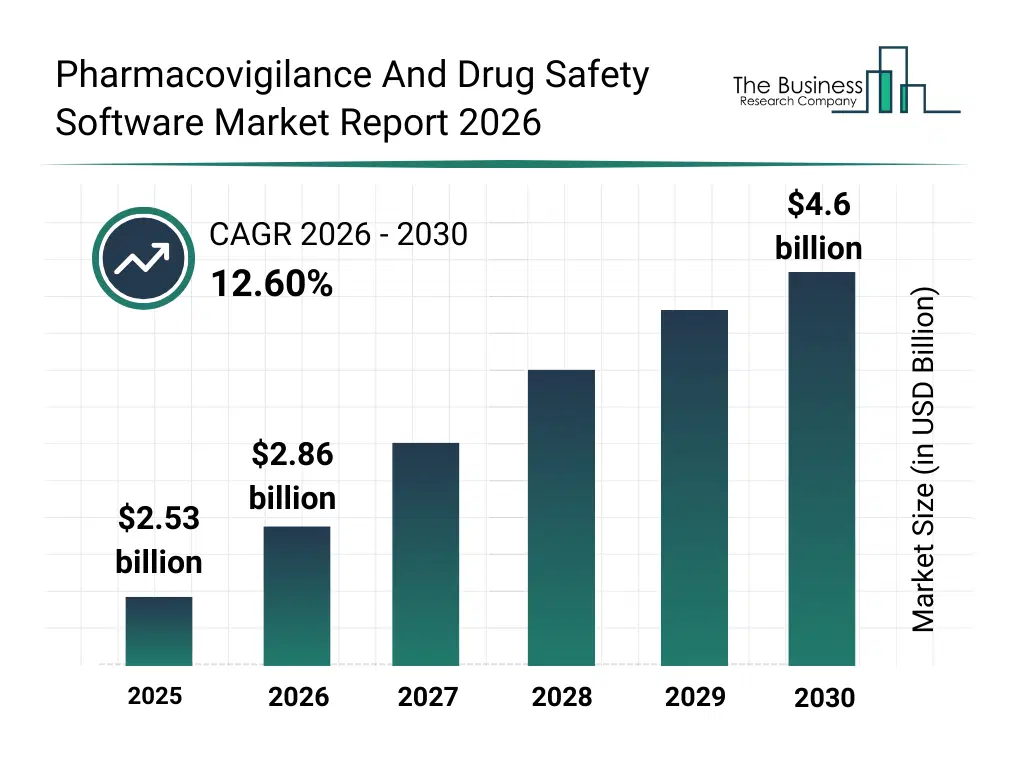
What Is The Pharmacovigilance And Drug Safety Software Market Size and Share 2026?
The pharmacovigilance and drug safety software market size has grown rapidly in recent years. It will grow from $2.53 billion in 2025 to $2.86 billion in 2026 at a compound annual growth rate (CAGR) of 12.8%. The growth in the historic period can be attributed to increasing adverse drug reaction reporting, globalization of clinical trials, stringent drug safety regulations, growth in pharmaceutical production, rising post-marketing surveillance requirements.What Is The Pharmacovigilance And Drug Safety Software Market Growth Forecast?
The pharmacovigilance and drug safety software market size is expected to see rapid growth in the next few years. It will grow to $4.6 billion in 2030 at a compound annual growth rate (CAGR) of 12.6%. The growth in the forecast period can be attributed to expansion of ai-driven signal detection, growth in biologics and specialty drugs, rising regulatory scrutiny, increasing outsourcing of pharmacovigilance services, demand for real-time safety analytics. Major trends in the forecast period include growing adoption of cloud-based pharmacovigilance platforms, increasing focus on real-world evidence monitoring, rising automation in adverse event reporting, expansion of integrated drug safety systems, higher demand for regulatory compliance solutions.Global Pharmacovigilance And Drug Safety Software Market Segmentation
1) By Software Type: Adverse Event Reporting Software, Drug Safety Audits Software, Issue Tracking Software, Fully Integrated Software 2) By Delivery Mode: On-premise, Cloud-based 3) By End User: Pharmaceutical And Biotechnology Companies, Contract Research Organizations, Business Process Outsourcing Firms, Other End Users Subsegments: 1) By Adverse Event Reporting Software: Spontaneous Reporting Systems, EHR Integration Solutions, Mobile Reporting Applications 2) By Drug Safety Audits Software: Audit Management Solutions, Compliance Tracking Tools, Reporting And Analytics Modules 3) By Issue Tracking Software: Incident Reporting Systems, Workflow Management Tools, Collaboration Platforms 4) By Fully Integrated Software: Pharmacovigilance Platforms, Regulatory Compliance Systems, Data Analytics And Visualization ToolsWhat Is The Driver Of The Pharmacovigilance And Drug Safety Software Market?
The growing demand for personalized medicine is expected to propel the growth of the pharmacovigilance and drug safety software market going forward. Personalized medicine, also known as precision medicine, is a healthcare approach that tailors treatment decisions to an individual’s genetic, biological, and lifestyle characteristics. The demand for personalized medicine is rising due to improved treatment outcomes, enhanced prevention and early detection strategies, and greater cost-effectiveness. The pharmacovigilance and drug safety software market supports personalized medicine by integrating pharmacogenetics, real-time data analytics, and post-marketing surveillance to optimize patient safety, monitor individualized treatments, and strengthen risk-management practices. For instance, in February 2024, according to the Personalized Medicine Coalition (PMC), a US-based non-profit organization, the FDA approved 16 novel personalized therapies for patients with rare diseases in 2023, compared to 6 approvals in 2022. Therefore, the growing demand for personalized medicine is driving the growth of the pharmacovigilance and drug safety software industry.Key Players In The Global Pharmacovigilance And Drug Safety Software Market
Major companies operating in the pharmacovigilance and drug safety software market are IQVIA, Oracle Corporation, ArisGlobal, RXLogix, Anju Software Inc., EXTEDO GmbH, Sarjen Systems Pvt Ltd, Ennov Group, AB Cube, Clinevo Technologies, Accenture plc, International Business Machines Corporation, Capgemini SE, Cognizant Technology Solutions, Wipro Limited, Genpact, Indegene Limited, PharmaLex, Deloitte Touche Tohmatsu Limited, ParexelGlobal Pharmacovigilance And Drug Safety Software Market Trends and Insights
Major companies operating in the pharmacovigilance and drug safety software market are focusing on developing innovative technologies such as cloud-based data lake platforms to strengthen adverse event monitoring, improve regulatory compliance, and enhance overall patient safety across clinical trials and post-market surveillance. A cloud-based data lake platform refers to a scalable centralized system capable of storing and analyzing large volumes of structured and unstructured safety data in real time, enabling advanced analytics, AI/ML modeling, and streamlined case processing within pharmacovigilance workflows. For instance, in December 2023, Thermo Fisher Scientific Inc., a US-based biotechnology company, launched CorEvidence, a proprietary cloud-based data lake platform designed to optimize pharmacovigilance processes by improving safety data management and case processing efficiency, particularly supporting post-authorization safety studies within clinical research registries.What Are Latest Mergers And Acquisitions In The Pharmacovigilance And Drug Safety Software Market?
In April 2024, Qinecsa, a UK-based company, acquired Insife ApS for an undisclosed amount. This acquisition strengthens Qinecsa’s end-to-end pharmacovigilance technology capabilities by integrating Insife’s advanced safety-software solutions into its digital PV ecosystem. It further enhances Qinecsa’s position in the global pharmacovigilance market by expanding its technology portfolio and supporting its strategy to become a leading provider of digital PV solutions. Insife ApS is a Denmark-based provider of pharmacovigilance software.Regional Insights
North America was the largest region in the pharmacovigilance and drug safety software market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Pharmacovigilance And Drug Safety Software Market?
The pharmacovigilance and drug safety software market includes revenues earned by entities by signal detection and management tools, risk assessment, and management software. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Pharmacovigilance And Drug Safety Software Market Report 2026?
The pharmacovigilance and drug safety software market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the pharmacovigilance and drug safety software industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Pharmacovigilance And Drug Safety Software Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.86 billion |
| Revenue Forecast In 2035 | $4.6 billion |
| Growth Rate | CAGR of 12.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Software Type, Delivery Mode, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | IQVIA, Oracle Corporation, ArisGlobal, RXLogix, Anju Software Inc., EXTEDO GmbH, Sarjen Systems Pvt Ltd, Ennov Group, AB Cube, Clinevo Technologies, Accenture plc, International Business Machines Corporation, Capgemini SE, Cognizant Technology Solutions, Wipro Limited, Genpact, Indegene Limited, PharmaLex, Deloitte Touche Tohmatsu Limited, Parexel |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
