
Plaque Modification Devices Market Report 2026
Global Outlook – By Type (Atherectomy Devices, Thrombectomy Devices, Chronic Total Occlusion Devices, Embolic Protection Devices), By Application (Coronary Artery Disease, Peripheral Artery Disease, Neurovascular Diseases), By End User (Hospitals, Ambulatory Surgical Centers, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Plaque Modification Devices Market Overview
• Plaque Modification Devices market size has reached to $1.49 billion in 2025 • Expected to grow to $2.23 billion in 2030 at a compound annual growth rate (CAGR) of 8.4% • Growth Driver: Rising Minimally Invasive Procedures Driving Market Growth • Market Trend: Advancements In Plaque Modification Devices Enhanced Market Growth • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Plaque Modification Devices Market?
Plaque modification devices refer to medical tools and technologies designed to treat atherosclerotic plaque buildup in blood vessels. These devices are used in minimally invasive procedures to restore blood flow by removing, modifying, or managing plaque within the arteries, particularly in cases of cardiovascular or peripheral artery diseases. The main types of plaque modification devices are atherectomy devices, thrombectomy devices, chronic total occlusion devices, and embolic protection devices. Atherectomy devices refer to specialized medical tools used to remove or reduce atherosclerotic plaque buildup within blood vessels, thereby improving blood flow. It is applicable for coronary artery disease, peripheral artery disease, and neurovascular diseases and caters to various end users such as hospitals, ambulatory surgical centers, and others.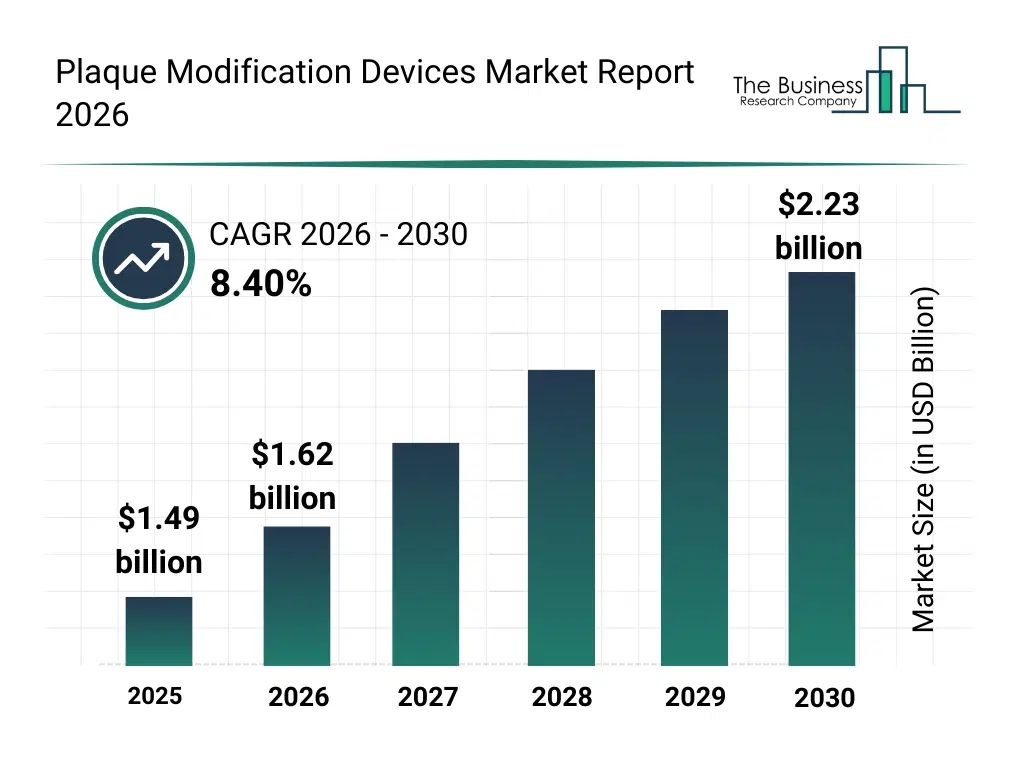
What Is The Plaque Modification Devices Market Size and Share 2026?
The plaque modification devices market size has grown strongly in recent years. It will grow from $1.49 billion in 2025 to $1.62 billion in 2026 at a compound annual growth rate (CAGR) of 8.5%. The growth in the historic period can be attributed to increasing atherosclerosis prevalence, growth in angioplasty procedures, aging population, rise in peripheral artery disease, advancement in interventional cardiology.What Is The Plaque Modification Devices Market Growth Forecast?
The plaque modification devices market size is expected to see strong growth in the next few years. It will grow to $2.23 billion in 2030 at a compound annual growth rate (CAGR) of 8.4%. The growth in the forecast period can be attributed to AI-guided vascular imaging, expansion of complex vascular interventions, demand for safer procedures, growth in outpatient interventions, innovation in plaque removal devices. Major trends in the forecast period include minimally invasive plaque removal, precision atherectomy technologies, improved embolic protection, advanced catheter-based systems, reduced procedure time devices.Global Plaque Modification Devices Market Segmentation
1) By Type: Atherectomy Devices, Thrombectomy Devices, Chronic Total Occlusion Devices, Embolic Protection Devices 2) By Application: Coronary Artery Disease, Peripheral Artery Disease, Neurovascular Diseases 3) By End User: Hospitals, Ambulatory Surgical Centers, Other End Users Subsegments: 1) By Atherectomy Devices: Rotational Atherectomy Devices, Directional Atherectomy Devices, Laser Atherectomy Devices, Orbital Atherectomy Devices 2) By Thrombectomy Devices: Mechanical Thrombectomy Devices, Aspiration Thrombectomy Devices, Manual Thrombectomy Devices, Hybrid Thrombectomy Devices 3) By Chronic Total Occlusion (CTO) Devices: CTO Crossing Guidewires, CTO Balloons, CTO Catheters, CTO Support Systems 4) By Embolic Protection Devices: Distal Protection Devices, Proximal Protection Devices, Filter-Based Embolic Protection Devices, Balloon-Based Embolic Protection DevicesWhat Is The Driver Of The Plaque Modification Devices Market?
A rising number of minimally invasive procedures is expected to propel the growth of the plaque modification devices market going forward. Minimally invasive procedures refer to medical or surgical techniques that involve smaller incisions, minimal tissue disruption, and reduced physical trauma compared to traditional open surgery. The rise in minimally invasive procedures is driven by advancements in technology, such as high-definition imaging and robotic systems, which improve precision, and increasing patient demand for faster recovery and reduced scarring. Plaque modification devices enhance the safety and efficiency of minimally invasive procedures by enabling precise removal of arterial blockages and expanding the scope of treatment for patients with complex vascular conditions. For instance, in January 2024, Intuitive Surgical Inc., a US-based manufacturer of robotic products, reported that they had placed 415 da Vinci surgical systems (robotic surgical systems that use a minimally invasive surgical approach) in the fourth quarter of 2023, representing a 12% increase compared to 2022. Therefore, the rising number of minimally invasive procedures is driving the growth of the plaque modification devices industry.Key Players In The Global Plaque Modification Devices Market
Major companies operating in the plaque modification devices market are Cardinal Health, Johnson & Johnson, Abbott Laboratories, Siemens Healthineers, Philips Healthcare, Becton Dickinson, Stryker Corporation, Boston Scientific, Terumo Corporation, Edward Lifesciences Corporation, Teleflex Incorporated, Merit Medical Systems Inc., MicroPort Scientific Corporation, Asahi Intecc Co. Ltd., AngioDynamics, Cardiovascular Systems Inc. (CSI), ACIST Medical Systems Inc., Endologix Inc., Avinger Inc., SoundBite Medical Solutions Inc.Global Plaque Modification Devices Market Trends and Insights
Major companies operating in the plaque modification devices market are focusing on developing advanced solutions, such as intravascular image-guided, catheter-based systems, to gain a competitive edge in the market. Intravascular image-guided refers to medical procedures or interventions that use imaging technology to guide instruments or devices inside blood vessels. For instance, in August 2024, Avinger, a US-based medical device company, launched the Pantheris LV image-guided directional atherectomy system, designed specifically for treating peripheral artery disease (PAD) in larger blood vessels such as the superficial femoral and popliteal arteries. It offers unique features such as higher-speed plaque excision for efficient removal of challenging occlusive tissue, a proprietary jog mechanism for enhanced plaque apposition, and an advanced guidewire management system to streamline user operation.What Are Latest Mergers And Acquisitions In The Plaque Modification Devices Market?
In May 2024, Johnson & Johnson, a US-based pharmaceutical industry company, acquired Shockwave Medical Inc. for an undisclosed amount. Through this acquisition, Johnson & Johnson aims to enhance its cardiovascular portfolio by integrating Shockwave's intravascular lithotripsy technology to treat calcified arterial plaque, with expectations of generating at least $1 billion in annual sales and addressing unmet needs in coronary and peripheral artery diseases. Shockwave Medical Inc. is a US-based medical company that develops and commercializes products to treat cardiovascular disease, including plaque modification devices.Regional Outlook
North America was the largest region in the plaque modification devices market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Plaque Modification Devices Market?
The plaque modification devices market consists of sales of atherectomy devices, scoring balloons, cutting balloons, plaque excision systems, and rotational ablation devices. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Plaque Modification Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.62 billion |
| Revenue Forecast In 2035 | $2.23 billion |
| Growth Rate | CAGR of 8.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Cardinal Health, Johnson & Johnson, Abbott Laboratories, Siemens Healthineers, Philips Healthcare, Becton Dickinson, Stryker Corporation, Boston Scientific, Terumo Corporation, Edward Lifesciences Corporation, Teleflex Incorporated, Merit Medical Systems Inc., MicroPort Scientific Corporation, Asahi Intecc Co. Ltd., AngioDynamics, Cardiovascular Systems Inc. (CSI), ACIST Medical Systems Inc., Endologix Inc., Avinger Inc., SoundBite Medical Solutions Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
