
Point-of-Care Diagnostics Devices And Equipment Market Report 2026
Global Outlook – By Type (Infectious disease testing kits, Cardio metabolic monitoring kits, Cholesterol testing kits, Pregnancy and fertility tests kits, Tumor Or cancer markers, Urinalysis testing kits, Hematology testing kits, Drugs of abuse testing kits, Fecal occult testing kits, Other Types), By Prescription Mode (Prescription-based Testing Devices, Over-The-Counter (OTC) Testing Devices), By End User (Professional diagnostic centers, Hospitals/critical care centers, Outpatient healthcare Setting, Ambulatory care settings, Research laboratories, Home) – Market Size, Trends, Strategies, and Forecast to 2035
Point-of-Care Diagnostics Devices And Equipment Market Overview
• Point-of-Care Diagnostics Devices And Equipment market size has reached to $64.11 billion in 2025 • Expected to grow to $118.72 billion in 2030 at a compound annual growth rate (CAGR) of 13.5% • Growth Driver: Increasing Adoption Of Mobile Health Technologies Boost The Point-Of-Care Diagnostics Devices Market • Market Trend: Innovative Point-Of-Care Diagnostic Device Cippoint Revolutionizes Healthcare Accessibility • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Point-of-Care Diagnostics Devices And Equipment Market?
Point-of-care diagnostics devices are designed to aid doctors in performing medical diagnostic testing at or near the point of care, for immediate knowledge on diseases or conditions. These devices are used to test glucose and cholesterol levels, do electrolyte and enzyme analysis, test for drugs of abuse and for infectious diseases, and for pregnancy testing. The main types of point-of-care diagnostic devices and equipment are infectious disease testing kits, cardiometabolic monitoring kits, cholesterol testing kits, pregnancy and fertility tests kits, tumor/cancer markers, urinalysis testing kits, hematology testing kits, drugs of abuse testing kits, fecal occult testing kits, and others. Pregnancy and fertility test kits are point-of-care diagnostic devices used to test pregnancy and fertility prior to seeing a doctor. The various prescription modes include prescription-based testing devices, and over-the-counter (OTC) testing devices. These are used by end-users such as professional diagnostic centers, hospitals/critical care centres, outpatient healthcare settings, ambulatory care settings, research laboratories, and homes.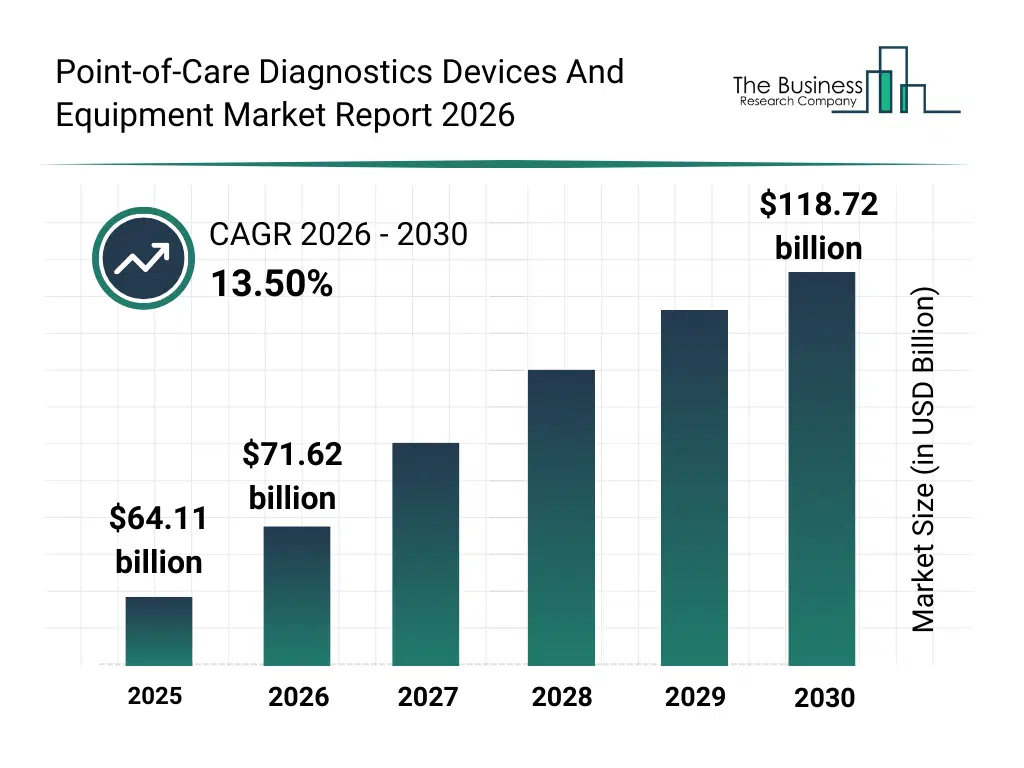
What Is The Point-of-Care Diagnostics Devices And Equipment Market Size and Share 2026?
The point-of-care diagnostics devices and equipment market size has grown rapidly in recent years. It will grow from $64.11 billion in 2025 to $71.62 billion in 2026 at a compound annual growth rate (CAGR) of 11.7%. The growth in the historic period can be attributed to growing burden of chronic diseases, increasing demand for rapid clinical decision-making, expansion of outpatient healthcare services, rising adoption of portable diagnostic devices, growth of preventive healthcare practices.What Is The Point-of-Care Diagnostics Devices And Equipment Market Growth Forecast?
The point-of-care diagnostics devices and equipment market size is expected to see rapid growth in the next few years. It will grow to $118.72 billion in 2030 at a compound annual growth rate (CAGR) of 13.5%. The growth in the forecast period can be attributed to increasing demand for home healthcare diagnostics, rising adoption of digital health ecosystems, expansion of infectious disease screening programs, growing integration of ai-based diagnostic support, increasing focus on remote patient monitoring. Major trends in the forecast period include increasing adoption of rapid diagnostic testing devices, rising demand for decentralized healthcare diagnostics, growing integration of connected point-of-care platforms, expansion of home-based testing solutions, enhanced focus on user-friendly device design.Global Point-of-Care Diagnostics Devices And Equipment Market Segmentation
1) By Type: Infectious disease testing kits, Cardio metabolic monitoring kits, Cholesterol testing kits, Pregnancy and fertility tests kits, Tumor Or cancer markers, Urinalysis testing kits, Hematology testing kits, Drugs of abuse testing kits, Fecal occult testing kits, Other Types 2) By Prescription Mode: Prescription-based Testing Devices, Over-The-Counter (OTC) Testing Devices 3) By End User: Professional diagnostic centers, Hospitals/critical care centers, Outpatient healthcare Setting, Ambulatory care settings, Research laboratories, Home Subsegments: 1) By Infectious Disease Testing Kits: HIV Testing Kits, Hepatitis Testing Kits, Malaria Testing Kits, Influenza Testing Kits, COVID-19 Testing Kits, Other Infectious Disease Testing Kits 2) By Cardio Metabolic Monitoring Kits: Blood Glucose Testing Kits, Blood Pressure Monitoring Kits, Lipid Testing Kits, Other Cardio Metabolic Monitoring Kits 3) By Cholesterol Testing Kits: Total Cholesterol Testing Kits, HDL Cholesterol Testing Kits, LDL Cholesterol Testing Kits, Triglyceride Testing Kits 4) By Pregnancy and Fertility Test Kits: Pregnancy Test Kits, Ovulation Test Kits, Fertility Monitoring Kits 5) By Tumor Or Cancer Markers: Prostate-Specific Antigen (PSA) Test Kits, Human Epidermal Growth Factor Receptor 2 (HER2) Test Kits, CA-125 Test Kits, Alpha-Fetoprotein (AFP) Test Kits, Other Cancer Marker Test Kits 6) By Urinalysis Testing Kits: Protein Testing Kits, Glucose Testing Kits, pH Testing Kits, Specific Gravity Testing Kits, Nitrite Testing Kits, Other Urinalysis Testing Kits 7) By Hematology Testing Kits: Complete Blood Count (CBC) Kits, Hemoglobin Testing Kits, Hematocrit Testing Kits, Platelet Count Kits, Other Hematology Testing Kits 8) By Drugs of Abuse Testing Kits: Marijuana Testing Kits, Cocaine Testing Kits, Opioid Testing Kits, Amphetamine Testing Kits, Other Drugs of Abuse Testing Kits 9) By Fecal Occult Testing Kits: Stool Blood Test Kits, Occult Blood Screening Kits, Other Fecal Occult Testing Kits 10) By Other Types: Glucose Monitoring Kits, Temperature Monitoring Kits, Oxygen Saturation Monitoring Kits, Other Specialty Point-of-Care KitsWhat Is The Driver Of The Point-of-Care Diagnostics Devices And Equipment Market?
The increasing adoption of mobile health technologies is expected to propel the growth of the point-of-care diagnostics devices market going forward. Mobile health refers to the use of mobile devices, such as smartphones and tablets, as well as other wireless technology such as wearable devices, for the delivery of healthcare services, information, and monitoring of health-related data. Point-of-care diagnostics (POC) devices play a crucial role in mobile health (mHealth) by enabling rapid and convenient testing and data collection at or near the patient's location. These devices are designed to provide immediate results for various medical tests and are often integrated with mobile apps or connected to smartphones for data management and transmission. For instance, in February2025, according to PharmiWeb, about 23% of healthcare encounters are expected to happen virtually in 2025. Therefore, the increasing adoption of mobile health technologies driving the growth of the point-of-care diagnostics devices market.Key Players In The Global Point-of-Care Diagnostics Devices And Equipment Market
Major companies operating in the point-of-care diagnostics devices and equipment market are Abbott Laboratories, F. Hoffmann-La Roche AG, Siemens Healthineers, Danaher, Beckman Coulter, Becton Dickinson and Company, Qiagen N.V., Johnson & Johnson, Alere, BioMerieux, Chembio Diagnostic Systems, Quidel Corporation, EKF Diagnostics, Trinity Biotech Plc, OraSure Technologies, Sysmex Corporation, Radiometer Medical ApS, Response Biomedical Corporation, Bio-Rad Laboratories, Illumina, Thermo Fisher Scientific Inc., LabCorp, Luminex Corporation, LifeScan, Nova, Sekisui Diagnostics, Hemocue AB, Akers Biosciences,Global Point-of-Care Diagnostics Devices And Equipment Market Trends and Insights
Major companies operating in the point-of-care diagnostic devices and equipment market are developing innovative products such as Cippoint to improve access to healthcare and reduce the time to diagnosis. Cippoint is a point-of-care testing device for multiple health conditions. It is a state-of-the-art device that offers a wide range of testing parameters, including cardiac markers, diabetes, infectious diseases, fertility, thyroid function, inflammation, metabolic markers, and coagulation markers. For instance, in January 2023, Cipla Limited, an India-based pharmaceutical company, launched a point-of-care testing device for multiple health conditions called Cippoint. It is a state-of-the-art device that offers a wide range of testing parameters. Cippoint is an immunofluorescence-based quantitative analyzer that allows healthcare professionals to get test results in 3 to 15 minutes. This makes it possible to make clinical decisions more quickly and efficiently. Moreover, it is easy to use and can be used in a variety of settings, including hospitals, clinics, and mobile vans.What Are Latest Mergers And Acquisitions In The Point-of-Care Diagnostics Devices And Equipment Market?
In December 2023, Roche, a Switzerland-based provider of in vitro diagnostics, pharmaceuticals, and healthcare solutions, acquired select point-of-care (POC) technology assets from LumiraDx for US$ 295 million. With this acquisition, Roche aims to integrate a versatile, multi-assay point-of-care testing platform into its diagnostics portfolio to expand decentralized testing capabilities in primary-care and low-resource settings. LumiraDx is a UK-based company that offers point of care diagnostics.Regional Outlook
North America was the largest region in the point-of-care diagnostic devices and equipment market in 2025. Asia-Pacific was the second-largest region in the global point-of-care diagnostics devices and equipment market. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are China, India, Japan, Australia, Indonesia, South Korea, Bangladesh, Thailand, Vietnam, Malaysia, Singapore, Philippines, Hong Kong, Taiwan, New Zealand, UK, Germany, France, Italy, Spain, Austria, Belgium, Denmark, Finland, Ireland, Netherlands, Norway, Portugal, Sweden, Switzerland, Russia, Czech Republic, Poland, Romania, Ukraine, USA, Canada, Mexico, Brazil, Chile, Argentina, Colombia, Peru, Saudi Arabia, Israel, Iran, Turkey, UAE, Egypt, Nigeria, South Africa.What Defines the Point-of-Care Diagnostics Devices And Equipment Market?
The point-of-care diagnostics market consists of sales of instruments such as pregnancy and fertility testing kits, cholesterol test strips, drug abuse testing kits, and others that are used in point-of-care diagnostics. Values in this market are factory gate values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Point-of-Care Diagnostics Devices And Equipment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $71.62 billion |
| Revenue Forecast In 2035 | $118.72 billion |
| Growth Rate | CAGR of 11.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Prescription Mode, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Abbott Laboratories, F. Hoffmann-La Roche AG, Siemens Healthineers, Danaher, Beckman Coulter, Becton Dickinson and Company, Qiagen N.V., Johnson & Johnson, Alere, BioMerieux, Chembio Diagnostic Systems, Quidel Corporation, EKF Diagnostics, Trinity Biotech Plc, OraSure Technologies, Sysmex Corporation, Radiometer Medical ApS, Response Biomedical Corporation, Bio-Rad Laboratories, Illumina, Thermo Fisher Scientific Inc., LabCorp, Luminex Corporation, LifeScan, Nova, Sekisui Diagnostics, Hemocue AB, Akers Biosciences, |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
