
Point Of Care Infectious Disease Testing Market Report 2026
Global Outlook – By Product (Kits And Reagents, Instruments), By Technology (Lateral Flow Immunoassay, Agglutination Test, Flow-through Test Or Immunoconcentration Assay, Molecular Diagnostics, Other Technologies), By Disease (Human Immunodeficiency Virus Point-of-Care Testing, Clostridium Difficile Point-of-Care Testing, Hepatitis B Virus Point-of-Care Testing, Pneumonia or Streptococcus Associated Infections, Respiratory Syncytial Virus Point-of-Care Testing, Human Papillomavirus Point-of-Care Testing, Influenza Or Flu Point-of-Care Testing, Other Diseases), By End Use (Hospitals, Diagnostic Centers, Research and Academic Institutes, Homecare Settings, Other End Users) - Market Size, Trends, And Global Forecast 2026-2035
Point Of Care Infectious Disease Testing Market Overview
• Point Of Care Infectious Disease Testing market size has reached to $4.78 billion in 2025 • Expected to grow to $9.02 billion in 2030 at a compound annual growth rate (CAGR) of 13.5% • Growth Driver: The Rising Contagious Disease Is Fueling The Market Growth Due To The Increasing Need For Rapid Diagnosis And Containment • Market Trend: The Role Of Proprietary Microfluidic Technology In Diagnostic Advancements • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Point Of Care Infectious Disease Testing Market?
Point of care (POC) infectious disease testing refers to diagnostic tests that are performed at or near the site of patient care, such as hospitals, clinics, doctors’ offices, or even at home. These tests are designed to detect infectious diseases quickly and accurately, providing immediate results to facilitate prompt treatment decisions. POC tests are typically user-friendly, require minimal equipment, and offer results within minutes to hours, making them ideal for situations where timely diagnosis is crucial. The main product types in point of care infectious disease testing are kits and reagents, and instruments. Kits and reagents are pre-prepared components used in point-of-care diagnostics to quickly detect infectious agents through reactions with patient samples. Technologies used include lateral flow immunoassay, agglutination tests, flow-through tests or immunoconcentration assays, and molecular diagnostics, among others, while the diseases tested for include human immunodeficiency virus (HIV), clostridium difficile, hepatitis B virus, pneumonia or streptococcus-associated infections, respiratory syncytial virus, human papillomavirus, influenza, and others. Point-of-care testing is used by various end-users, including hospitals, diagnostic centers, research and academic institutes, homecare settings, and others.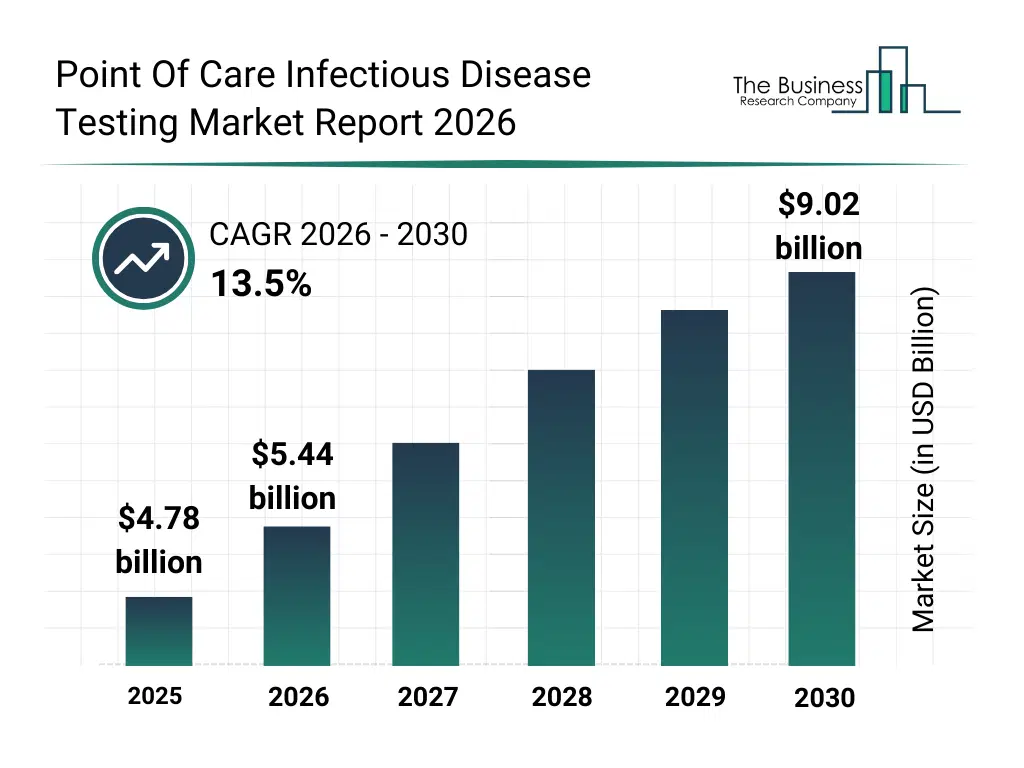
What Is The Point Of Care Infectious Disease Testing Market Size and Share 2026?
The point of care infectious disease testing market size has grown rapidly in recent years. It will grow from $4.78 billion in 2025 to $5.44 billion in 2026 at a compound annual growth rate (CAGR) of 13.7%. The growth in the historic period can be attributed to rising incidence of infectious diseases, increased demand for rapid diagnostics, expansion of decentralized healthcare delivery, availability of lateral flow testing technologies, growing use of rapid screening in hospitals.What Is The Point Of Care Infectious Disease Testing Market Growth Forecast?
The point of care infectious disease testing market size is expected to see rapid growth in the next few years. It will grow to $9.02 billion in 2030 at a compound annual growth rate (CAGR) of 13.5%. The growth in the forecast period can be attributed to growing adoption of self-testing solutions, expansion of molecular poc diagnostics, rising investments in digital health integration, increasing focus on outbreak preparedness, growing demand for low-cost rapid tests. Major trends in the forecast period include increasing adoption of rapid molecular poc tests, rising demand for home-based infectious disease testing, growing use of multiplex diagnostic kits, expansion of portable diagnostic instruments, enhanced focus on early disease detection.Global Point Of Care Infectious Disease Testing Market Segmentation
1) By Product: Kits And Reagents, Instruments 2) By Technology: Lateral Flow Immunoassay, Agglutination Test, Flow-through Test Or Immunoconcentration Assay, Molecular Diagnostics, Other Technologies 3) By Disease: Human Immunodeficiency Virus Point-of-Care Testing, Clostridium Difficile Point-of-Care Testing, Hepatitis B Virus Point-of-Care Testing, Pneumonia or Streptococcus Associated Infections, Respiratory Syncytial Virus Point-of-Care Testing, Human Papillomavirus Point-of-Care Testing, Influenza Or Flu Point-of-Care Testing, Other Diseases 4) By End Use: Hospitals, Diagnostic Centers, Research and Academic Institutes, Homecare Settings, Other End Users Subsegments: 1) By Kits And Reagents: Test Kits, Reagents 2) By Instruments: Portable Devices, Automated AnalyzersWhat Is The Driver Of The Point Of Care Infectious Disease Testing Market?
The rising contagious disease is expected to propel the growth of the point of care infectious disease testing market going forward. A contagious disease refers to an illness that can be spread from one person to another through direct contact, airborne droplets, or other means of transmission. The rising incidence of contagious diseases is primarily due to increasing human mobility, as greater movement of people across regions and countries facilitates faster and wider transmission of infectious agents. Point of care infectious disease testing accelerates the detection and diagnosis of contagious diseases, enabling timely treatment and reducing the risk of transmission within communities. For instance, in June 2024, according to the UK Health Security Agency, a UK-based government agency, between week 40 of 2022 and week 15 of 2023, 108 NHS trusts in England recorded 1,681 critical care admissions due to confirmed influenza. In contrast, the previous flu season had only 182 admissions across 114 trusts, which increased to 316 by week 39 of 2022. Therefore, the rising contagious disease is driving the growth of point of care infectious disease testing industry.Key Players In The Global Point Of Care Infectious Disease Testing Market
Major companies operating in the point of care infectious disease testing market are Cardinal Health, F Hoffmann La Roche Ltd, Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthineers AG, Becton Dickinson and Company, bioMérieux Société Anonyme, QuidelOrtho Corporation, Bio Rad Laboratories Inc., DiaSorin Società per Azioni, Danaher Corporation, Cue Health Inc., Nova Biomedical Corporation, Seegene Incorporated, Biosynex Société Anonyme, Trivitron Healthcare Private Limited, EKF Diagnostics Holdings plc, Trinity Biotech Public Limited Company, bioLytical Laboratories Inc., Cepheid Inc., LumiraDx UK Ltd., Meridian Bioscience Inc., GenBody Inc.Global Point Of Care Infectious Disease Testing Market Trends and Insights
Major companies operating in the point-of-care infectious disease testing market are focusing on integrating innovative technologies such as proprietary microfluidic technology to enhance test accuracy, reduce time-to-results, and improve patient outcomes. Proprietary microfluidic technology refers to specialized systems that manipulate small volumes of fluids at the microscale, enabling rapid, precise, and cost-effective analysis, which helps point of care (POC) infectious disease testing by allowing on-site, fast pathogen detection with integrated sample collection, analysis, and result output, enhancing diagnostic speed and accessibility without the need for centralized laboratory testing. For instance, in June 2023, Sysmex Corporation, a Japan-based diagnostics company, launched the PA-100 AST System in Europe, marking the world’s first point-of-care testing solution for directly detecting antimicrobial susceptibility. Utilizing advanced microfluidic technology, the system enables rapid identification of bacterial infections and evaluation of antimicrobial effectiveness, significantly reducing processing time compared to conventional methods. It comprises a compact, desktop-sized analyzer and single-use sample cartridges, making it well-suited for near-patient settings such as primary care clinics. Designed to facilitate faster and more accurate antimicrobial therapy decisions, the system contributes to the global effort to combat antimicrobial resistance by improving diagnostic efficiency.What Are Latest Mergers And Acquisitions In The Point Of Care Infectious Disease Testing Market?
In July 2024, F. Hoffmann-La Roche AG, a Switzerland-based pharmaceutical company, acquired LumiraDx’s point of care technology for $295 million. With this acquisition, Roche aims to expand global access to decentralized diagnostic testing by integrating LumiraDx’s versatile point-of-care platform into its diagnostics portfolio, enabling timely and actionable results across primary care and underserved healthcare settings. LumiraDx is a US-based medical equipment manufacturing company that specializes in providing point of care infectious disease testing.Regional Outlook
North America was the largest region in the point of care infectious disease testing market in 2025. Asia-Pacific is expected to be the Fastest-Growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Point Of Care Infectious Disease Testing Market?
The point-of-care infectious disease testing market consists of revenues earned by entities by providing services such as rapid diagnostic testing, disease screening, and onsite sample analysis for infectious conditions. The market value includes the value of related goods sold by the service provider or included within the service offering. The point-of-care infectious disease testing market also includes sales of handheld analyzers, lateral flow assays, molecular testing devices, and related consumables used for the immediate detection of infectious diseases. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Point Of Care Infectious Disease Testing Market Report 2026?
The point of care infectious disease testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the point of care infectious disease testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Point Of Care Infectious Disease Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $5.44 billion |
| Revenue Forecast In 2035 | $9.02 billion |
| Growth Rate | CAGR of 13.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Technology, Disease, End Use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Cardinal Health, F Hoffmann La Roche Ltd, Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthineers AG, Becton Dickinson and Company, bioMérieux Société Anonyme, QuidelOrtho Corporation, Bio Rad Laboratories Inc., DiaSorin Società per Azioni, Danaher Corporation, Cue Health Inc., Nova Biomedical Corporation, Seegene Incorporated, Biosynex Société Anonyme, Trivitron Healthcare Private Limited, EKF Diagnostics Holdings plc, Trinity Biotech Public Limited Company, bioLytical Laboratories Inc., Cepheid Inc., LumiraDx UK Ltd., Meridian Bioscience Inc., GenBody Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
