Point-Of-Care International Normalized Ratio (INR) Testing Market Report 2026

Point-Of-Care International Normalized Ratio (INR) Testing Market Report 2026
Global Outlook – By Product Type (Devices, Consumables, Software), By Technology (Electrochemical, Optical, Others), By Distribution Channel (Direct Sales, Retail Pharmacies, Online Or E-Commerce, Others), By Application (Hospitals, Clinics, Home Care, Diagnostic Centers, Others), By End-User (Healthcare Professionals, Patients) – Market Size, Trends, Strategies, and Forecast to 2035
Point-Of-Care International Normalized Ratio (INR) Testing Market Overview
• Point-Of-Care International Normalized Ratio (INR) Testing market size has reached to $1.58 billion in 2025 • Expected to grow to $2.56 billion in 2030 at a compound annual growth rate (CAGR) of 10.1% • Growth Driver: Rising Prevalence Of Cardiovascular Diseases Fueling The Growth Of The Market Due To The Need For Rapid Coagulation Monitoring And Timely Anticoagulation Management • Market Trend: Developing Compact Patient Self-Monitoring INR Test Meters To Expand Decentralized Testing And Improve Cost-Efficient Anticoagulation Management • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Point-Of-Care International Normalized Ratio (INR) Testing Market?
Point-of-care International Normalized Ratio (INR) testing refers to the immediate measurement of the international normalized ratio using portable devices at or near the patient’s location. It allows rapid assessment of blood clotting status, enabling timely adjustments to anticoagulant therapy. This method enhances patient safety by providing quick, accurate results without the need for conventional laboratory testing. The main product types of point-of-care international normalized ratio testing solutions are devices, consumables, and software. Devices are portable diagnostic instruments used to rapidly measure coagulation status at or near the patient site to support anticoagulation management. These solutions are based on electrochemical and optical technologies and others, distributed through direct sales, retail pharmacies, online or e-commerce channels, and others, used across hospitals, clinics, home care, diagnostic centers, and others, among end users including healthcare professionals and patients.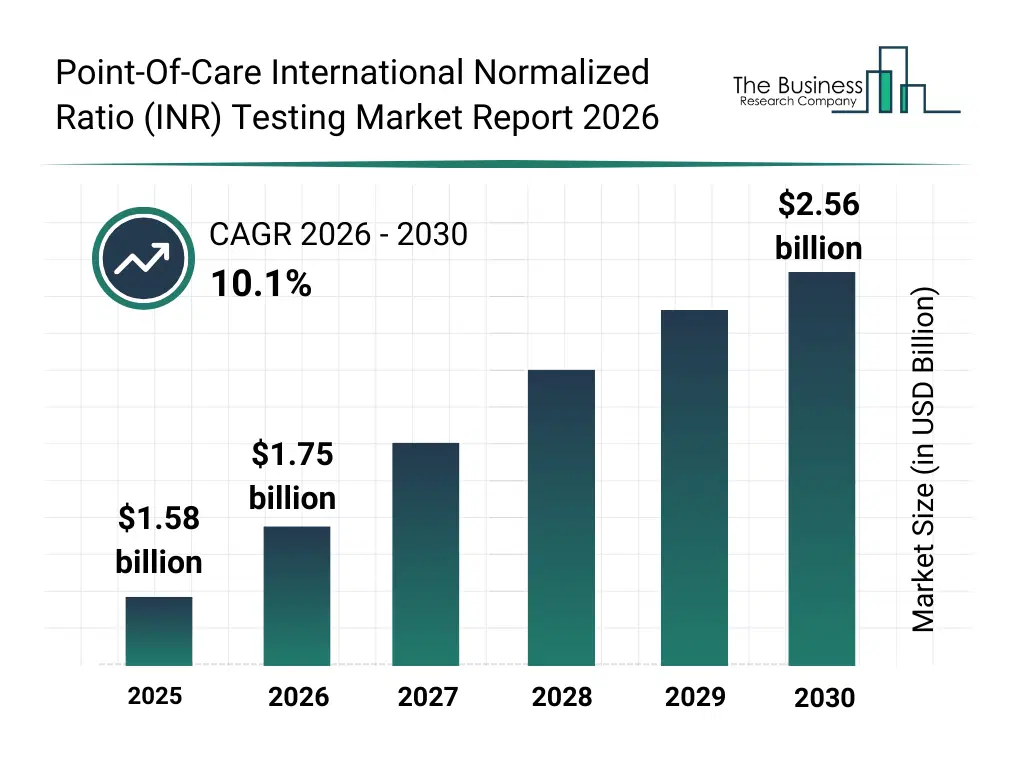
What Is The Point-Of-Care International Normalized Ratio (INR) Testing Market Size and Share 2026?
The point-of-care inr testing market size has grown rapidly in recent years. It will grow from $1.58 billion in 2025 to $1.75 billion in 2026 at a compound annual growth rate (CAGR) of 10.4%. The growth in the historic period can be attributed to increasing prevalence of cardiovascular diseases, growing use of anticoagulant therapy, rising incidence of atrial fibrillation, expansion of outpatient and homecare settings, increasing need for rapid coagulation testing.What Is The Point-Of-Care International Normalized Ratio (INR) Testing Market Growth Forecast?
The point-of-care inr testing market size is expected to see rapid growth in the next few years. It will grow to $2.56 billion in 2030 at a compound annual growth rate (CAGR) of 10.1%. The growth in the forecast period can be attributed to increasing demand for home-based monitoring, growing geriatric population on long-term anticoagulation, rising focus on personalized anticoagulation management, expansion of decentralized healthcare models, increasing adoption in primary care clinics. Major trends in the forecast period include technological advancements in portable coagulation analyzers, innovations in biosensor and microfluidic technologies, developments in wireless connectivity and data integration, research and development in high-accuracy test strips, advancements in smartphone-enabled inr testing.Global Point-Of-Care International Normalized Ratio (INR) Testing Market Segmentation
1) By Product Type: Devices, Consumables, Software 2) By Technology: Electrochemical, Optical, Others 3) By Distribution Channel: Direct Sales, Retail Pharmacies, Online Or E-Commerce, Others 4) By Application: Hospitals, Clinics, Home Care, Diagnostic Centers, Others 5) By End-User: Healthcare Professionals, Patients Subsegments: 1) By Devices: Portable Testing Devices, Handheld Testing Devices, Desktop Testing Devices 2) By Consumables: Test Strips, Cartridges, Control Solutions 3) By Software: Data Management Software, Remote Monitoring Software, Analytical SoftwareWhat Is The Driver Of The Point-Of-Care International Normalized Ratio (INR) Testing Market?
The rising prevalence of cardiovascular diseases is expected to propel the growth of the point-of-care International Normalized Ratio (INR) testing market going forward. Cardiovascular diseases are conditions that affect the heart and blood vessels, including heart attacks, strokes, and hypertension. Cardiovascular diseases are increasing due to unhealthy lifestyles such as poor diet, lack of exercise, and smoking. Point-of-care International Normalized Ratio (INR) testing aids in cardiovascular disease diagnosis by enabling rapid assessment of blood coagulation status at the bedside or clinic. It improves clinical decision-making by supporting timely detection of clotting abnormalities and guiding anticoagulation therapy. For instance, in October 2024, according to the Centers for Disease Control, a US-based government agency, in 2023, approximately 919,032 people died from cardiovascular disease, accounting for 1 in every 3 deaths. Therefore, the rising prevalence of cardiovascular diseases is driving the growth of the point-of-care International Normalized Ratio (INR) testing industry.Key Players In The Global Point-Of-Care International Normalized Ratio (INR) Testing Market
Major companies operating in the point-of-care international normalized ratio (inr) testing market are Abbott Laboratories, Danaher Corporation, Siemens Healthineers AG, Becton, Dickinson and Company, Hoffmann-La Roche Ltd., QuidelOrtho Corporation, Sysmex Corporation, Bio-Rad Laboratories Inc., Werfen S.A., HORIBA Ltd., Microvisk Ltd., Randox Laboratories Ltd., Nova Biomedical Corporation, Agappe Diagnostics Ltd., Visby Medical Inc., EKF Diagnostics Holdings plc, Trinity Biotech plc, HemoSonics LLC, Human Diagnostics Worldwide GmbH, CoaguSense Inc.Global Point-Of-Care International Normalized Ratio (INR) Testing Market Trends and Insights
Major companies operating in the point-of-care International Normalized Ratio (INR) testing market are focusing on developing advanced products, such as patient self-monitoring coagulation analyzers, to expand decentralized testing and shift recurring demand toward lower-cost consumables. Patient self-monitoring coagulation analyzers are portable devices that enable patients to perform PT/INR testing at home using a small capillary blood sample, supporting frequent monitoring while reducing dependence on clinical visits. For instance, in March 2024, Universal Biosensors, an Australia-based medical device company, launched the Xprecia Prime 4U Coagulation Analyzer for patient self-monitoring. This self-monitoring coagulation analyzer fits in the palm of the hand, delivers rapid PT/INR results from a single drop of capillary blood, and uses an electrochemical strip format that supports regulatory approvals, including 510(k) clearance and CLIA waiver for the U.S. market. This launch represents an important advancement in INR testing by enabling broader access to home monitoring, increasing test frequency, and improving cost efficiency through higher consumable utilization while maintaining clinical accuracy.What Are Latest Mergers And Acquisitions In The Point-Of-Care International Normalized Ratio (INR) Testing Market?
In September 2025, Una Health Ltd., a UK‑based medical technology company, partnered with iLine Microsystems Pvt. Ltd. to expand the distribution of International Normalised Ratio (INR) monitoring solutions that simplify anticoagulation monitoring. Through this distribution agreement, Una Health Ltd. and iLine Microsystems Pvt. Ltd. aim to increase accessibility of easy‑to‑use portable INR testing devices for patients on blood‑thinning therapies, facilitating more frequent self‑monitoring and better clinical outcomes. iLine Microsystems Pvt. Ltd. is an India‑based diagnostics and point‑of‑care device manufacturer.Regional Insights
North America was the largest region in the Point-of-Care INR Testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Point-Of-Care International Normalized Ratio (INR) Testing Market?
The point-of-care International Normalized Ratio (INR) testing market consists of revenues earned by entities by providing services such as calibration services, maintenance services, device repair services, data management services, remote monitoring services, technical support services, and training services. The market value includes the value of related goods sold by the service provider or included within the service offering. The point-of-care inr testing market also includes sales of point-of-care INR testing devices, test strips, lancets, control solutions, replacement batteries, carrying cases, and charging adapters. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Point-Of-Care International Normalized Ratio (INR) Testing Market Report 2026?
The point-of-care international normalized ratio (inr) testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the point-of-care international normalized ratio (inr) testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Point-Of-Care International Normalized Ratio (INR) Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.75 billion |
| Revenue Forecast In 2035 | $2.56 billion |
| Growth Rate | CAGR of 10.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Technology, Distribution Channel, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Abbott Laboratories, Danaher Corporation, Siemens Healthineers AG, Becton, Dickinson and Company, Hoffmann-La Roche Ltd., QuidelOrtho Corporation, Sysmex Corporation, Bio-Rad Laboratories Inc., Werfen S.A., HORIBA Ltd., Microvisk Ltd., Randox Laboratories Ltd., Nova Biomedical Corporation, Agappe Diagnostics Ltd., Visby Medical Inc., EKF Diagnostics Holdings plc, Trinity Biotech plc, HemoSonics LLC, Human Diagnostics Worldwide GmbH, CoaguSense Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Point-Of-Care International Normalized Ratio (INR) Testing Market Report 2026 market was valued at $1.58 billion in 2025, increased to $1.75 billion in 2026, and is projected to reach $2.56 billion by 2030.
request a sample hereThe expected CAGR for the Point-Of-Care International Normalized Ratio (INR) Testing market during the forecast period 2025–2030 is 10.1%.
request a sample hereMajor growth driver of the market includes: Rising Prevalence Of Cardiovascular Diseases Fueling The Growth Of The Market Due To The Need For Rapid Coagulation Monitoring And Timely Anticoagulation Management in the Point-Of-Care International Normalized Ratio (INR) Testing market. For further insights on this market,
request a sample hereThe point-of-care international normalized ratio (inr) testing market covered in this report is segmented –
1) By Product Type: Devices, Consumables, Software
2) By Technology: Electrochemical, Optical, Others
3) By Distribution Channel: Direct Sales, Retail Pharmacies, Online Or E-Commerce, Others
4) By Application: Hospitals, Clinics, Home Care, Diagnostic Centers, Others
5) By End-User: Healthcare Professionals, Patients Subsegments:
1) By Devices: Portable Testing Devices, Handheld Testing Devices, Desktop Testing Devices
2) By Consumables: Test Strips, Cartridges, Control Solutions
3) By Software: Data Management Software, Remote Monitoring Software, Analytical Software
request a sample here1) By Product Type: Devices, Consumables, Software
2) By Technology: Electrochemical, Optical, Others
3) By Distribution Channel: Direct Sales, Retail Pharmacies, Online Or E-Commerce, Others
4) By Application: Hospitals, Clinics, Home Care, Diagnostic Centers, Others
5) By End-User: Healthcare Professionals, Patients Subsegments:
1) By Devices: Portable Testing Devices, Handheld Testing Devices, Desktop Testing Devices
2) By Consumables: Test Strips, Cartridges, Control Solutions
3) By Software: Data Management Software, Remote Monitoring Software, Analytical Software
Major trend in this market includes: Developing Compact Patient Self-Monitoring INR Test Meters To Expand Decentralized Testing And Improve Cost-Efficient Anticoagulation Management For further insights on this market,
request a sample hereMajor companies operating in the Point-Of-Care International Normalized Ratio (INR) Testing market are Major companies operating in the point-of-care international normalized ratio (inr) testing market are Abbott Laboratories, Danaher Corporation, Siemens Healthineers AG, Becton, Dickinson and Company, Hoffmann-La Roche Ltd., QuidelOrtho Corporation, Sysmex Corporation, Bio-Rad Laboratories Inc., Werfen S.A., HORIBA Ltd., Microvisk Ltd., Randox Laboratories Ltd., Nova Biomedical Corporation, Agappe Diagnostics Ltd., Visby Medical Inc., EKF Diagnostics Holdings plc, Trinity Biotech plc, HemoSonics LLC, Human Diagnostics Worldwide GmbH, CoaguSense Inc.
request a sample hereNorth America was the largest region in the Point-of-Care INR Testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the point-of-care international normalized ratio (inr) testing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
request a sample here