
Point-of-Care (POC) Coagulation Testing Devices Market Report 2026
Global Outlook – By Product Type (Coagulation Monitors, Consumables, Coagulation Analyzers), By Technology (Optical Technology, Mechanical Technology, Electrochemical Technology), By Method (Prothrombin Time And International Normalized Ratio, Viscoelastic Coagulation Monitoring, Other Methods), By Application (Coagulation Testing, Other Applications), By End-User (Hospitals, Diagnostic Laboratories, Clinics, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Point-of-Care (POC) Coagulation Testing Devices Market Overview
• Point-of-Care (POC) Coagulation Testing Devices market size has reached to $2.32 billion in 2025 • Expected to grow to $3.49 billion in 2030 at a compound annual growth rate (CAGR) of 8.2% • Growth Driver: Rising Prevalence of Blood Clotting Disorders Drives Growth of Point-of-Care (POC) Coagulation Testing Devices Market • Market Trend: Revolutionizing Coagulation Testing with Immunofluorescence-Based Quantitative Analysis • North America was the largest region in 2025.What Is Covered Under Point-of-Care (POC) Coagulation Testing Devices Market?
Point-of-care (POC) coagulation testing devices are portable, easy-to-use medical devices used to assess the clotting ability of blood outside of a traditional laboratory setting. They are designed to provide rapid and accurate results, allowing healthcare providers to make immediate treatment decisions for patients with coagulation disorders or those at risk of abnormal bleeding or clotting. The main products of point-of-care (POC) coagulation testing devices are coagulation monitors, consumables, and coagulation analyzers. Coagulation monitors are medical devices used to measure the clotting ability of blood, typically in a point-of-care (POC) setting. They work by using various technologies include optical technology, mechanical technology, and electrochemical technology and follow prothrombin time and international normalized ratio, viscoelastic coagulation monitoring, and other methods. The different applications include blood glucose testing, infectious diseases testing, cardiac markers testing, coagulation testing, blood gas and electrolyte testing, and others and ar used by hospitals, diagnostic laboratories, clinics, and others.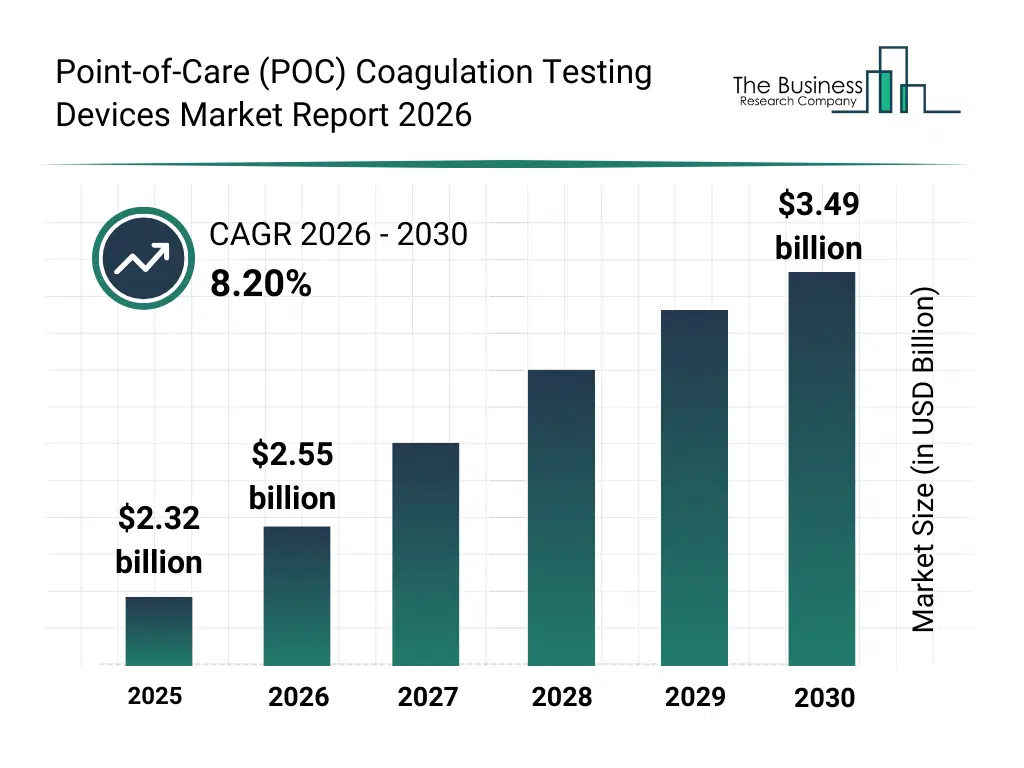
What Is The Point-of-Care (POC) Coagulation Testing Devices Market Size and Share 2026?
The point-of-care (poc) coagulation testing devices market size has grown strongly in recent years. It will grow from $2.32 billion in 2025 to $2.55 billion in 2026 at a compound annual growth rate (CAGR) of 9.7%. The growth in the historic period can be attributed to rising prevalence of coagulation disorders, increased use of anticoagulant therapies, expansion of decentralized healthcare settings, growing adoption of point-of-care diagnostics, improvements in coagulation testing technologies.What Is The Point-of-Care (POC) Coagulation Testing Devices Market Growth Forecast?
The point-of-care (poc) coagulation testing devices market size is expected to see strong growth in the next few years. It will grow to $3.49 billion in 2030 at a compound annual growth rate (CAGR) of 8.2%. The growth in the forecast period can be attributed to increasing demand for home healthcare diagnostics, rising focus on personalized medicine, expansion of telehealth-enabled diagnostics, growing geriatric population, continuous innovation in microfluidic testing platforms. Major trends in the forecast period include increasing adoption of portable coagulation monitors, rising demand for rapid diagnostic testing at point of care, growing use of home-based coagulation testing kits, expansion of wireless data connectivity in poc devices, enhanced focus on test accuracy and reliability.Global Point-of-Care (POC) Coagulation Testing Devices Market Segmentation
1) By Product Type: Coagulation Monitors, Consumables, Coagulation Analyzers 2) By Technology: Optical Technology, Mechanical Technology, Electrochemical Technology 3) By Method: Prothrombin Time And International Normalized Ratio, Viscoelastic Coagulation Monitoring, Other Methods 4) By Application: Coagulation Testing, Other Applications 5) By End-User: Hospitals, Diagnostic Laboratories, Clinics, Other End-Users Subsegments: 1) By Coagulation Monitors: Prothrombin Time (Pt) Monitors, International Normalized Ratio (Inr) Monitors, Activated Partial Thromboplastin Time (Aptt) Monitors, Whole Blood Coagulation Monitors, Clotting Time Monitors 2) By Consumables: Test Strips, Reagents, Control Solutions, Calibration Kits, Lancets And Needles 3) By Coagulation Analyzers: Portable Coagulation Analyzers, Benchtop Coagulation Analyzers, Fully Automated Coagulation Analyzers, Semi-Automated Coagulation Analyzers, Coagulation Testing Kits (For Home Use)What Is The Driver Of The Point-of-Care (POC) Coagulation Testing Devices Market?
The rise in the prevalence of blood clotting disorders is expected to propel the growth of the point-of-care (POC) coagulation testing devices market going forward. Blood clotting disorders impair the blood's ability to clot properly. Lifestyle factors can contribute to blood clotting disorders, including smoking, excessive alcohol consumption, and a sedentary lifestyle. These behaviors can affect blood vessel health, clotting factor levels, and blood flow, increasing the risk of abnormal clot formation. POC coagulation testing devices enable healthcare providers to quickly assess a patient's coagulation status, allowing for timely adjustments to medication dosages and treatment plans. For instance, in July 2025, according to the National Blood Authority, an Australia-based blood management agency, there were 2,788 patients living with hereditary haemophilia A (HMA) in 2023–2024, up from 2,681 patients in 2022–2023. Therefore, the rise in the prevalence of blood clotting disorders drives the growth of the point-of-care (POC) coagulation testing devices industry.Key Players In The Global Point-of-Care (POC) Coagulation Testing Devices Market
Major companies operating in the point-of-care (poc) coagulation testing devices market are Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Bio-Rad Laboratories Inc., Werfen, Haemonetics Corporation, HemoSonics LLC, Sysmex Corporation, Nihon Kohden Corporation, HORIBA Medical, Helena Laboratories Corporation, Diagnostica Stago, Sekisui Diagnostics, Instrumentation Laboratory, Trinity Biotech plc, CoaguSense Inc., iLine Microsystems, Medirox AB, Hemochron Systems, Axis-Shield DiagnosticsGlobal Point-of-Care (POC) Coagulation Testing Devices Market Trends and Insights
Major companies operating in the point-of-care (POC) coagulation testing devices market are developing immunofluorescence-based quantitative analyzers to improve the diagnosis and management of coagulation disorders. Immunofluorescence-based quantitative analyzers are diagnostic instruments used in laboratory settings to measure and quantify specific proteins or molecules involved in the coagulation process, aiding in diagnosing and managing blood clotting disorders. For instance, in January 2023, Cipla Limited, an India-based pharmaceutical company, launched Cippoint. This immunofluorescence-based quantitative analyzer utilizes immunofluorescence technology to provide rapid and accurate test results in 3 to 15 minutes, facilitating faster clinical decision-making processes. With its user-friendly interface, quick turnaround time, and comprehensive testing capabilities, Cippoint is crucial in enhancing diagnostic efficiency, enabling timely clinical decisions, and improving patient care outcomes.What Are Latest Mergers And Acquisitions In The Point-of-Care (POC) Coagulation Testing Devices Market?
In August 2024, Perosphere Technologies Inc, a US-based medical devices manufacturing company partnered with M3 Medical for an undisclosed amount. With this partnership, they aim to improve patient outcomes, reduce healthcare system costs, and establish a new standard of care for patients using anticoagulant therapies or at risk for bleeding in Ireland. M3 Medical is an Ireland-based medical device company.Regional Outlook
North America was the largest region in the point-of-care (POC) coagulation testing devices market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Point-of-Care (POC) Coagulation Testing Devices Market?
The point-of-care (POC) coagulation testing devices market consists of revenues earned by entities by providing services such as increased patient comfort, improved workflow efficiency, better patient management, remote monitoring capabilities, emergency use for immediate testing, and assessment of anticoagulant reversal effectiveness. The market value includes the value of related goods sold by the service provider or included within the service offering. The point-of-care (POC) coagulation testing devices market also includes sales of viscoelastic testing devices, platelet function testing devices, and D-Dimer testing devices. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Point-of-Care (POC) Coagulation Testing Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.55 billion |
| Revenue Forecast In 2035 | $3.49 billion |
| Growth Rate | CAGR of 9.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Technology, Method, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Bio-Rad Laboratories Inc., Werfen, Haemonetics Corporation, HemoSonics LLC, Sysmex Corporation, Nihon Kohden Corporation, HORIBA Medical, Helena Laboratories Corporation, Diagnostica Stago, Sekisui Diagnostics, Instrumentation Laboratory, Trinity Biotech plc, CoaguSense Inc., iLine Microsystems, Medirox AB, Hemochron Systems, Axis-Shield Diagnostics |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
