Post-Marketing Pharmacovigilance And Medical Information Market Report 2026

Post-Marketing Pharmacovigilance And Medical Information Market Report 2026
Global Outlook – By Type (Spontaneous Reporting, Intensified Adverse Drug Reaction (ADR) Reporting, Targeted Spontaneous Reporting, Cohort Event Monitoring, Electronic Health Record (EHR) Mining), By Publication Type (Books, Online Media, Journals), By End User (Hospitals, Research Organizations, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Post-Marketing Pharmacovigilance And Medical Information Market Overview
• Post-Marketing Pharmacovigilance And Medical Information market size has reached to $6.9 billion in 2025 • Expected to grow to $11.89 billion in 2030 at a compound annual growth rate (CAGR) of 11.4% • Growth Driver: Rising Incidence Of Adverse Drug Reactions Fueling The Growth Of The Market Due To Increasing Use Of Complex Biologics And Specialty Medications • Market Trend: AI-Powered Pharmacovigilance Solutions Driving Efficiency And Compliance In Drug Safety Monitoring • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Post-Marketing Pharmacovigilance And Medical Information Market?
Post-marketing pharmacovigilance is the continuous monitoring of a drug’s safety once it is available on the market, focusing on identifying, evaluating, and preventing adverse effects or other drug-related issues. Medical information services deliver precise, scientific, and current responses to healthcare providers and patients about a drug’s usage, safety, and effectiveness. Together, these processes safeguard patient health, ensure regulatory compliance, and enhance treatment outcomes. The main types of post-marketing pharmacovigilance and medical information are spontaneous reporting, intensified adverse drug reaction (ADR) reporting, targeted spontaneous reporting, cohort event monitoring, and electronic health record (EHR) mining. Spontaneous reporting involves healthcare professionals or patients voluntarily noticing and communicating unexpected reactions associated with medication usage to authorized bodies. It includes product types, such as books, online media, and journals, and caters to diverse end users, including hospitals, research organizations, and others.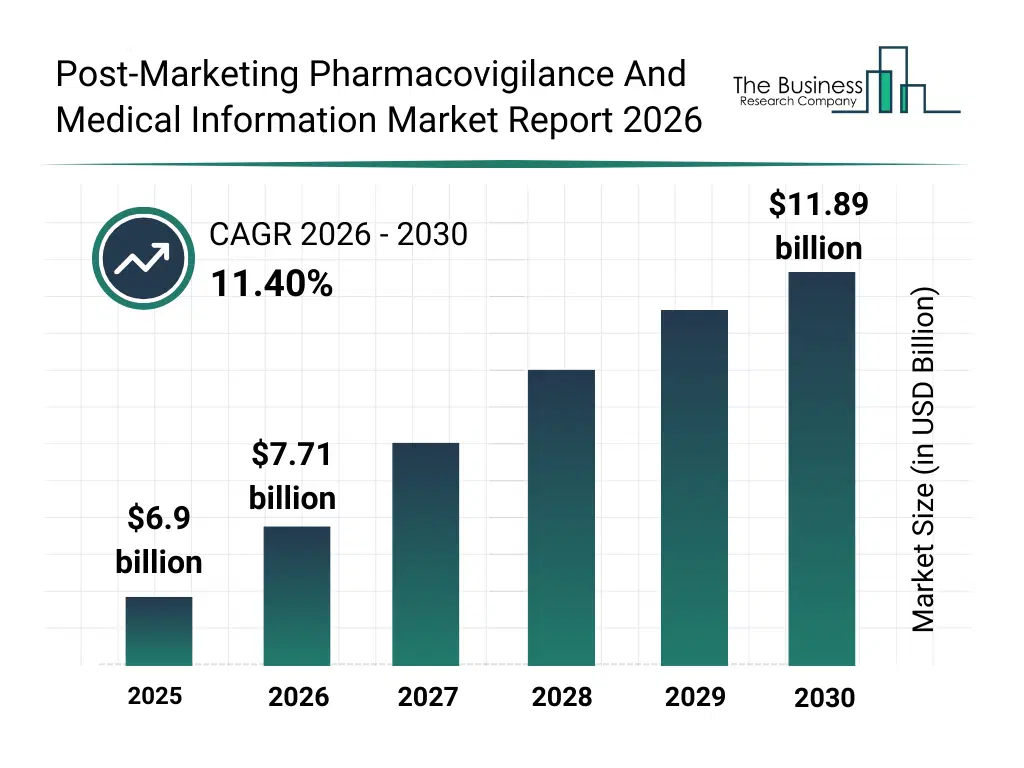
What Is The Post-Marketing Pharmacovigilance And Medical Information Market Size and Share 2026?
The post-marketing pharmacovigilance and medical information market size has grown rapidly in recent years. It will grow from $6.9 billion in 2025 to $7.71 billion in 2026 at a compound annual growth rate (CAGR) of 11.7%. The growth in the historic period can be attributed to limited post-marketing surveillance infrastructure, reliance on manual ADR reporting, growing regulatory requirements for drug safety, increased number of new drug approvals, lack of integrated hospital data systems.What Is The Post-Marketing Pharmacovigilance And Medical Information Market Growth Forecast?
The post-marketing pharmacovigilance and medical information market size is expected to see rapid growth in the next few years. It will grow to $11.89 billion in 2030 at a compound annual growth rate (CAGR) of 11.4%. The growth in the forecast period can be attributed to integration of ai and ml algorithms in signal detection, adoption of longitudinal patient data analysis, expansion of disease registry-linked monitoring, increasing digital app-based reporting, rising investments in real-time pharmacovigilance technologies. Major trends in the forecast period include increasing adoption of electronic health record (ehr) mining, rising use of real-time adverse drug reaction (adr) monitoring systems, growth in risk population-based and therapeutic class-based reporting, expansion of cohort event monitoring for new drug users, integration of digital and mobile platforms for pharmacovigilance reporting.Global Post-Marketing Pharmacovigilance And Medical Information Market Segmentation
1) By Type: Spontaneous Reporting, Intensified Adverse Drug Reaction (ADR) Reporting, Targeted Spontaneous Reporting, Cohort Event Monitoring, Electronic Health Record (EHR) Mining 2) By Publication Type: Books, Online Media, Journals 3) By End User: Hospitals, Research Organizations, Other End-Users Subsegments: 1) By Spontaneous Reporting: Consumer Reporting, Healthcare Professional Reporting, Regulatory Authority Reporting, Pharmaceutical Company Reporting, Digital App-Based Reporting 2) By Intensified Adverse Drug Reaction (ADR) Reporting: Hospital-Based Surveillance, Program-Specific Monitoring, Disease-Specific Monitoring, Product-Specific Monitoring, Real-Time Monitoring Systems 3) By Targeted Spontaneous Reporting: Risk Population-Based Reporting, Therapeutic Class-Based Reporting, Adverse Drug Reaction Focused Reporting, Region-Specific Reporting, Healthcare Setting-Specific Reporting 4) By Cohort Event Monitoring: Prospective Cohort Monitoring, Retrospective Cohort Monitoring, Active Follow-Up Monitoring, New Drug User Monitoring, Disease Registry-Linked Monitoring 5) By Electronic Health Record (EHR) Mining: Natural Language Processing-Based Mining, Artificial Intelligence (AI) And Machine Learning Algorithms, Rule-Based Signal Detection, Longitudinal Patient Data Analysis, Integrated Hospital Data SystemsWhat Is The Driver Of The Post-Marketing Pharmacovigilance And Medical Information Market?
The rising incidence of adverse drug reactions is expected to propel the growth of the post-marketing pharmacovigilance and medical information market going forward. Adverse drug reactions refer to harmful or unintended effects that occur when a medication is taken at its recommended dosage for prevention, diagnosis, or treatment purposes. The incidence of adverse drug reactions is rising as the aging population increasingly requires treatment for multiple chronic conditions, leading to the simultaneous use of several medications that heighten the likelihood of harmful drug interactions and unexpected side effects. Post-marketing pharmacovigilance and medical information support the management of adverse drug reactions by detecting, tracking, and sharing details on potential side effects after a medicine is launched, helping maintain its safety and effectiveness. For instance, in January 2023, according to Navikenz, a US-based artificial intelligence-focused IT services company, in the US, adverse drug reactions are estimated to cause 200,000 to 400,000 deaths annually, exceeding the toll from stroke and diabetes combined, with around 2.7 million cases each year resulting in over 100,000 hospitalizations and more than 15,000 deaths. Therefore, the rising incidence of adverse drug reactions is driving the growth of the post-marketing pharmacovigilance and medical information industry.Key Players In The Global Post-Marketing Pharmacovigilance And Medical Information Market
Major companies operating in the post-marketing pharmacovigilance and medical information market are Accenture plc, Sanofi S.A., Capgemini SE, Merck & Co. Inc., Cognizant Technology Solutions Corporation, IQVIA Holdings Inc., HCL Technologies Limited, ICON plc, Syneos Health Inc., Genpact Limited, Parexel International Corporation, Avalere Health LLC, Quanticate Limited, Inizio Consulting LLCGlobal Post-Marketing Pharmacovigilance And Medical Information Market Trends and Insights
Major companies operating in the post-marketing pharmacovigilance and medical information market are focusing on developing advanced solutions, such as AI-powered pharmacovigilance workflows, to enhance adverse event detection accuracy and enable real-time safety monitoring for better patient outcomes. AI-powered pharmacovigilance workflows use artificial intelligence to automate drug safety monitoring, enabling faster adverse event detection and improved regulatory compliance. For instance, in January 2024, PubHive Ltd., a UK-based cloud-based software-as-a-service (SaaS) company, introduced a centralized Summary of Product Characteristics (SmPC) management for life science companies and research organizations. The platform consolidates critical drug safety information into a single, easily accessible repository, automates literature monitoring and regulatory reporting, enhances team collaboration, and streamlines compliance processes through advanced AI capabilities. This enables pharmacovigilance teams to operate more efficiently and proactively, ensuring both patient safety and adherence to regulatory standards.What Are Latest Mergers And Acquisitions In The Post-Marketing Pharmacovigilance And Medical Information Market?
In October 2023, Ergomed plc, a UK-based pharmaceutical services company, acquired Panacea Pharma Projects Limited for an undisclosed amount. With this acquisition, Ergomed aims to expand its pharmacovigilance business, PrimeVigilance, strengthen its capabilities, and broaden its geographic reach, supporting its strategy to become a leading provider of pharmacovigilance services. Panacea Pharma Projects Limited is a UK-based pharmaceutical consultancy firm that specializes in pharmacovigilance (PV) and medical information services, including post-marketing support.Regional Insights
North America was the largest region in the post-marketing pharmacovigilance and medical information market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Post-Marketing Pharmacovigilance And Medical Information Market?
The post-marketing pharmacovigilance and medical information market includes revenues earned by entities by providing services such as adverse event reporting and management, signal detection and risk assessment, drug safety database, medical call center services, and regulatory compliance support. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Post-Marketing Pharmacovigilance And Medical Information Market Report 2026?
The post-marketing pharmacovigilance and medical information market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the post-marketing pharmacovigilance and medical information industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Post-Marketing Pharmacovigilance And Medical Information Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $7.71 billion |
| Revenue Forecast In 2035 | $11.89 billion |
| Growth Rate | CAGR of 11.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Publication Type, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Accenture plc, Sanofi S.A., Capgemini SE, Merck & Co. Inc., Cognizant Technology Solutions Corporation, IQVIA Holdings Inc., HCL Technologies Limited, ICON plc, Syneos Health Inc., Genpact Limited, Parexel International Corporation, Avalere Health LLC, Quanticate Limited, Inizio Consulting LLC |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Post-Marketing Pharmacovigilance And Medical Information market was valued at $6.9 billion in 2025, increased to $7.71 billion in 2026, and is projected to reach $11.89 billion by 2030.
request a sample hereThe global Post-Marketing Pharmacovigilance And Medical Information market is expected to grow at a CAGR of 11.4% from 2026 to 2035 to reach $11.89 billion by 2035.
request a sample hereSome Key Players in the Post-Marketing Pharmacovigilance And Medical Information market Include, Accenture plc, Sanofi S.A., Capgemini SE, Merck & Co. Inc., Cognizant Technology Solutions Corporation, IQVIA Holdings Inc., HCL Technologies Limited, ICON plc, Syneos Health Inc., Genpact Limited, Parexel International Corporation, Avalere Health LLC, Quanticate Limited, Inizio Consulting LLC .
request a sample hereMajor trend in this market includes: AI-Powered Pharmacovigilance Solutions Driving Efficiency And Compliance In Drug Safety Monitoring. For further insights on this market.
request a sample hereNorth America was the largest region in the post-marketing pharmacovigilance and medical information market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the post-marketing pharmacovigilance and medical information market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
request a sample here