
Pressure Ulcer Detection Device Market Report 2026
Global Outlook – By Product (In-Shoe Sensor, Surface Electrode, SEM (Skin And Tissue Assessment Monitoring Scanner) Scanner, Pressure Measuring Mattress), By Application (Stage 1, Stage 2, Stage 3, Stage 4), By End User (Hospitals, Nursing Homes, Homecare Settings, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Pressure Ulcer Detection Device Market Overview
• Pressure Ulcer Detection Device market size has reached to $1.51 billion in 2025 • Expected to grow to $1.87 billion in 2030 at a compound annual growth rate (CAGR) of 4.4% • Growth Driver: Increasing Prevalence of Diabetes Propels Growth Of Pressure Ulcer Detection Device Market • Market Trend: IR-MED Clinically Validates Handheld Device For Early Detection Of Pressure Injuries • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Pressure Ulcer Detection Device Market?
A pressure ulcer detection device is a medical device designed to monitor and detect the presence of pressure ulcers, also known as bedsores or pressure sores. Pressure ulcers are skin injuries that develop when prolonged pressure is applied to a specific body area. Pressure ulcer detection devices are valuable tools in healthcare settings for the prevention, early detection, and management of pressure ulcers and contribute to better patient care, reduced healthcare costs, and improved overall outcomes for individuals at risk of developing these painful and potentially life-threatening skin injuries. The main product types of pressure ulcer detection devices are in-shoe sensors, surface electrodes, sem (skin and tissue assessment monitoring scanner) scanners, and pressure measuring mattresses. In-shoe sensor is a specialized device or sensor system designed to be placed inside a shoe to collect data related to various aspects of foot movement, pressure distribution, and gait dynamics. The various applications include Stage 1, stage 2, stage 3, and stage 4. These are used by various end-users such as hospitals, nursing homes, home care settings, and others.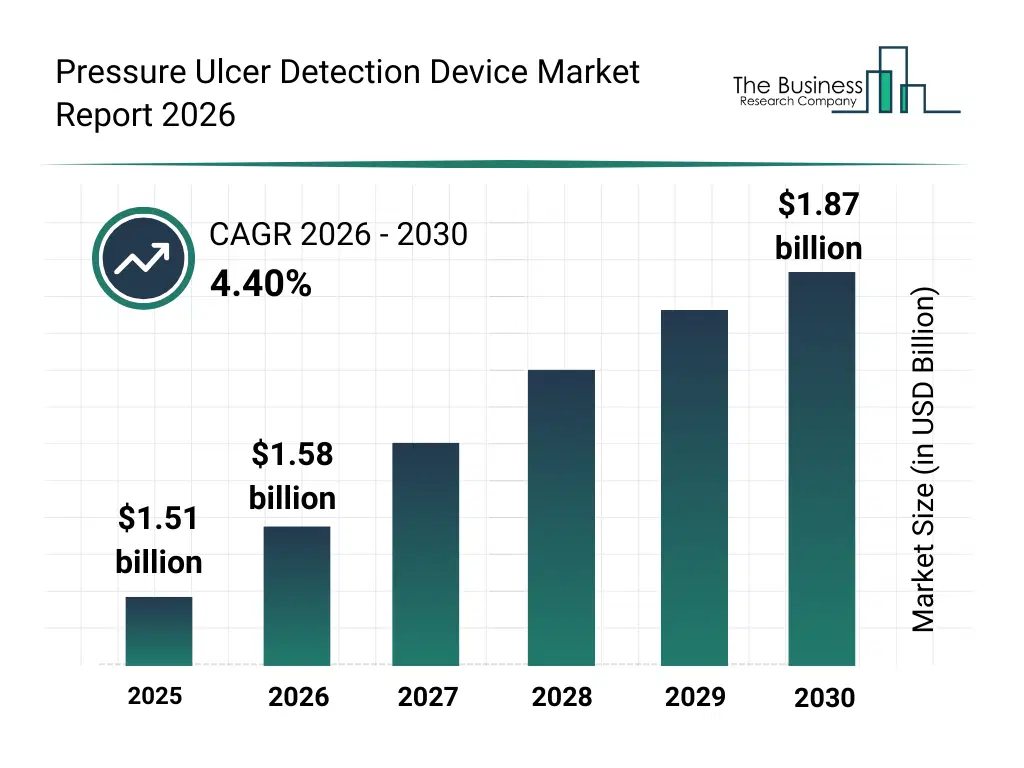
What Is The Pressure Ulcer Detection Device Market Size and Share 2026?
The pressure ulcer detection device market size has grown steadily in recent years. It will grow from $1.51 billion in 2025 to $1.58 billion in 2026 at a compound annual growth rate (CAGR) of 4.4%. The growth in the historic period can be attributed to rising incidence of pressure ulcers among immobile and geriatric patients, increasing awareness of early detection to reduce treatment costs, early adoption of surface electrodes and mattress-based pressure monitoring systems, expansion of hospital and nursing home prevention programs, growing clinical emphasis on pressure injury risk assessment protocols.What Is The Pressure Ulcer Detection Device Market Growth Forecast?
The pressure ulcer detection device market size is expected to see steady growth in the next few years. It will grow to $1.87 billion in 2030 at a compound annual growth rate (CAGR) of 4.4%. The growth in the forecast period can be attributed to rising demand for continuous real-time pressure monitoring technologies, increasing uptake of wearable and in-shoe pressure sensor devices, growing utilization of sem scanners for early-stage ulcer identification, expanding use of detection devices in homecare and remote care environments, rising focus on outcome-based care and complication reduction in high-risk patient populations. Major trends in the forecast period include rising adoption of sem scanners and tissue monitoring devices for early pressure ulcer detection, increasing utilization of pressure measuring mattresses in hospital and long-term care settings, growing use of in-shoe and wearable pressure sensors for foot ulcer prevention, expansion of continuous pressure monitoring solutions across homecare and nursing facilities, increasing clinical focus on stage-based assessment and prevention of pressure ulcers.Global Pressure Ulcer Detection Device Market Segmentation
1) By Product: In-Shoe Sensor, Surface Electrode, SEM (Skin And Tissue Assessment Monitoring Scanner) Scanner, Pressure Measuring Mattress 2) By Application: Stage 1, Stage 2, Stage 3, Stage 4 3) By End User: Hospitals, Nursing Homes, Homecare Settings, Other End Users Subsegments: 1) By In-Shoe Sensor: Smart Insoles, Pressure-Sensitive Insoles, Wearable Pressure Sensors For Foot Ulcers 2) By Surface Electrode: Electrodes For Continuous Pressure Monitoring, Electrode Pads For Early Detection, Disposable Surface Electrodes 3) By SEM (Skin And Tissue Assessment Monitoring) Scanner: Handheld SEM Scanners, Wireless SEM Devices, SEM Scanners For Real-Time Monitoring 4) By Pressure Measuring Mattress: Pressure Mapping Mattress Systems, Air-Flow Pressure Measuring Mattresses, Foam Pressure Measuring MattressesWhat Is The Driver Of The Pressure Ulcer Detection Device Market?
The increasing prevalence of diabetes is expected to propel the growth of the pressure ulcer detection device market going forward. Diabetes is a long-term metabolic condition marked by high blood glucose, which can seriously harm the heart, blood vessels, eyes, kidneys, and nerves over time. Diabetic individuals are at an increased risk of developing pressure ulcers due to neuropathy, poor circulation, and others. Pressure ulcer detection devices play a significant role in identifying early signs of pressure-related skin damage and assisting healthcare professionals in preventing the development of pressure ulcers in individuals with diabetes. For instance, in January 2023, according to a report by the International Diabetes Federation (IDF), a Belgium-based diabetes association, the prevalence of diabetes would rise by 25% in 2030 and by 51% by 2045. Therefore, the increasing prevalence of diabetes drives the pressure ulcer detection device industry.Key Players In The Global Pressure Ulcer Detection Device Market
Major companies operating in the pressure ulcer detection device market are 3M Company, Hill Rom Holdings Inc., Stryker Corp., Smith and Nephew plc, Coloplast Group, Pall Hartmann AG, Integra LifeSciences Corporation, Acelity Holdings Inc., Invacare Corporation, Tactile Systems Technology Inc., Veristride Inc., Julius Zorn GmbH, Tekscan Inc., Orpyx Medical Technologies, Medi GmbH & Co., Xsensor Technology Corporation, Sensoria Inc., Moticon GmbH, ArjoHunleigh Inc., Bruin Biometrics LLCGlobal Pressure Ulcer Detection Device Market Trends and Insights
Major companies operating in the pressure ulcer detection device market are focusing on technological innovation, such as AI-powered, infrared spectrographic decision support systems to improve patient outcomes and prevent complications. AI-powered infrared spectrographic systems integrate handheld optical monitoring, real-time tissue analysis, and predictive algorithms to identify subdermal pressure injuries with high sensitivity and specificity, reducing reliance on visual inspection and supporting timely clinical intervention. For instance, in July 2023, IR-MED Inc., a US-based medical technology company, launched PressureSafe, a groundbreaking handheld medical device that leverages infrared optical spectroscopy combined with artificial intelligence to support the early detection of pressure injuries. Designed for point-of-care use, PressureSafe offers a non-invasive, skin tone-agnostic assessment tool that accurately identifies pressure ulcers in their early stages, including suspected deep tissue injuries. The device has demonstrated impressive clinical efficacy, with sensitivity rates around 92% to 94% and specificity close to 88%, which enables healthcare providers to promptly detect and intervene in pressure injury cases, significantly reducing incidence rates by up to 50%.What Are Latest Mergers And Acquisitions In The Pressure Ulcer Detection Device Market?
In March 2023, Syst'Am, a France-based company that specializes in medical equipment, acquired Jarven Health Care Oy for an undisclosed amount. Syst'am strengthens its position in anti-decubitus prevention and care devices with this acquisition. Further, this acquisition would expedite Järven's commercial expansion while maintaining its identity, making it the best option. Jarven Health Care Oy is a Sweden-based medical equipment manufacturer specializing in preventing and treating pressure ulcers.Regional Outlook
North America was the largest region in the pressure ulcer detection device market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Pressure Ulcer Detection Device Market?
The pressure ulcer detection device market consists of sales of thermographic imaging system, wearable pressure sensors, and surface pressure mapping systems. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Pressure Ulcer Detection Device Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.58 billion |
| Revenue Forecast In 2035 | $1.87 billion |
| Growth Rate | CAGR of 4.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | 3M Company, Hill Rom Holdings Inc., Stryker Corp., Smith and Nephew plc, Coloplast Group, Pall Hartmann AG, Integra LifeSciences Corporation, Acelity Holdings Inc., Invacare Corporation, Tactile Systems Technology Inc., Veristride Inc., Julius Zorn GmbH, Tekscan Inc., Orpyx Medical Technologies, Medi GmbH & Co., Xsensor Technology Corporation, Sensoria Inc., Moticon GmbH, ArjoHunleigh Inc., Bruin Biometrics LLC |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
