
Primary Biliary Cholangitis Therapeutics Market Report 2026
Global Outlook – By Drug (Ursodeoxycholic Acid, Obeticholic Acid, Other Drugs), By Mechanism Of Action (MOA) (Receptor Agonist, Receptor Modulator, Enzyme Activator, Enzyme Inhibitor, Other Mechanisms Of Action), By Distribution Channel (Hospital Pharmacies, Drug Store And Retail Pharmacies, Online Pharmacies), By Use (Human, Veterinary) – Market Size, Trends, Strategies, and Forecast to 2035
Primary Biliary Cholangitis Therapeutics Market Overview
• Primary Biliary Cholangitis Therapeutics market size has reached to $1.01 billion in 2025 • Expected to grow to $1.85 billion in 2030 at a compound annual growth rate (CAGR) of 12.9% • Growth Driver: Increasing Prevalence Of Liver Diseases Fueling The Growth Of The Market Due To Rising Alcohol Consumption And Liver Damage • Market Trend: Breakthrough In PBC Treatment With Selective Pparδ Activation • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Primary Biliary Cholangitis Therapeutics Market?
Primary biliary cholangitis (PBC) therapeutics refer to treatments designed to manage and slow the progression of primary biliary cholangitis, a chronic autoimmune liver disease that damages the bile ducts. These work by enhancing bile flow, minimizing inflammation, and slowing liver fibrosis. The main purpose of these therapeutics is to reduce liver inflammation, improve bile flow, prevent liver fibrosis, and delay the onset of liver failure. The main drug types of primary biliary cholangitis therapeutics are ursodeoxycholic acid, obeticholic acid, and others. Ursodeoxycholic acid is a bile acid used to improve liver function by reducing bile buildup and slowing liver damage, especially in cholestatic liver diseases like primary biliary cholangitis. These work by various mechanisms of action (MOA), such as receptor agonist, receptor modulator, enzyme activator, enzyme inhibitor, and others, and are distributed through several distribution channels, including hospital pharmacies, drug stores and retail pharmacies, and online pharmacies, serving key end users, including human and veterinary purposes.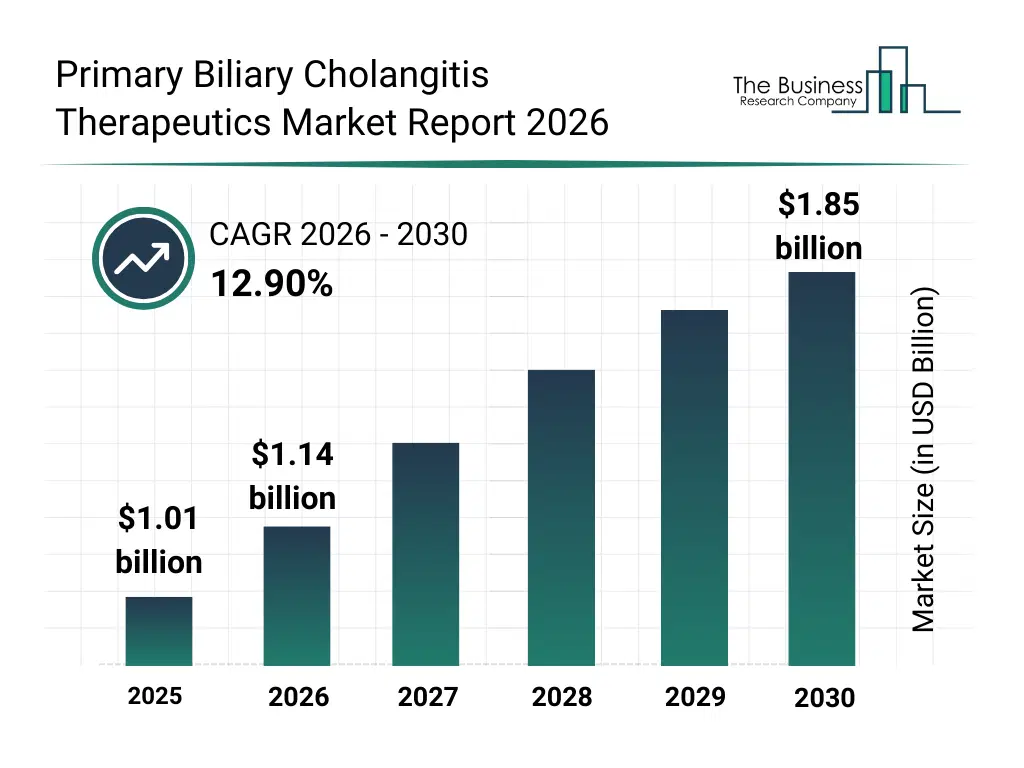
What Is The Primary Biliary Cholangitis Therapeutics Market Size and Share 2026?
The primary biliary cholangitis therapeutics market size has grown rapidly in recent years. It will grow from $1.01 billion in 2025 to $1.14 billion in 2026 at a compound annual growth rate (CAGR) of 12.9%. The growth in the historic period can be attributed to limited treatment options for pbc, growing prevalence of autoimmune liver diseases, increasing liver disease awareness campaigns, reliance on ursodeoxycholic acid monotherapy, expanding hospital infrastructure for liver disease care.What Is The Primary Biliary Cholangitis Therapeutics Market Growth Forecast?
The primary biliary cholangitis therapeutics market size is expected to see rapid growth in the next few years. It will grow to $1.85 billion in 2030 at a compound annual growth rate (CAGR) of 12.9%. The growth in the forecast period can be attributed to development of obeticholic acid and combination therapies, growth in investigational drugs and pipeline candidates, increasing adoption of receptor agonists and modulators, rising patient awareness and early diagnosis initiatives, expansion of online and retail pharmacy distribution networks. Major trends in the forecast period include rising adoption of ursodeoxycholic acid and obeticholic acid therapies, growth in combination therapies and pipeline drugs, increasing focus on early diagnosis and disease monitoring, expansion of hospital and retail pharmacy distribution channels, rising awareness about autoimmune liver diseases and patient education.Global Primary Biliary Cholangitis Therapeutics Market Segmentation
1) By Drug: Ursodeoxycholic Acid, Obeticholic Acid, Other Drugs 2) By Mechanism Of Action (MOA): Receptor Agonist, Receptor Modulator, Enzyme Activator, Enzyme Inhibitor, Other Mechanisms Of Action 3) By Distribution Channel: Hospital Pharmacies, Drug Store And Retail Pharmacies, Online Pharmacies 4) By Use: Human, Veterinary Subsegments: 1) By Ursodeoxycholic Acid: Branded Ursodeoxycholic Acid, Generic Ursodeoxycholic Acid, Combination Therapies With Ursodeoxycholic Acid 2) By Obeticholic Acid: Monotherapy, Combination Therapy, Branded Obeticholic Acid 3) By Other Drugs: Fibrates, Budesonide, Immunosuppressants, Investigational Drugs Or Pipeline CandidatesWhat Is The Driver Of The Primary Biliary Cholangitis Therapeutics Market?
The increasing prevalence of liver diseases is expected to propel the growth of the primary biliary cholangitis therapeutics market going forward. Liver diseases refer to a group of conditions that impair the normal functioning of the liver, often leading to inflammation, scarring, or liver failure. The increasing prevalence of liver diseases is due to rise in alcohol consumption as excessive alcohol intake damages liver cells, leading to inflammation and chronic liver conditions. Primary biliary cholangitis therapeutics help manage liver diseases by targeting bile duct inflammation, slowing disease progression, and preserving liver function. They improve patient outcomes by reducing symptoms, enhancing quality of life, and delaying the need for liver transplantation. For instance, in September 2025, according to the Centers for Disease Control and Prevention (CDC), a US-based federal public health agency, chronic liver disease and cirrhosis caused 52,222 deaths in the United States in 2023, making it the ninth leading cause of death. Therefore, the increasing prevalence of liver diseases is driving the growth of the primary biliary cholangitis therapeutics industry.Key Players In The Global Primary Biliary Cholangitis Therapeutics Market
Major companies operating in the primary biliary cholangitis therapeutics market are Novartis AG, Gilead Sciences Inc., Ipsen Pharma, Intercept Pharmaceuticals, Enanta Pharmaceuticals, Mirum Pharmaceuticals, Calliditas Therapeutics AB, Dr. Falk Pharma GmbH, NGM Biopharmaceuticals Inc., COUR Pharmaceuticals, HighTide Therapeutics Inc., Pliant Therapeutics Inc., Tiziana Life Sciences, Phenex Pharmaceuticals, Synlogic TherapeuticsGlobal Primary Biliary Cholangitis Therapeutics Market Trends and Insights
Major companies operating in the primary biliary cholangitis therapeutics market are focusing on developing advanced products and obtaining approvals, such as for peroxisome proliferator-activated receptor agonists, to improve bile acid metabolism and slow disease progression in patients. Peroxisome proliferator-activated receptor (PPAR) agonists are drugs that activate PPAR proteins, which regulate genes involved in metabolism, inflammation, and energy balance. For instance, in August 2024, Gilead Sciences Inc., a US-based pharmaceutical company, received accelerated approval from the U.S. Food and Drug Administration (FDA) for Livdelzi (seladelpar) for the treatment of primary biliary cholangitis (PBC) in adults who have had an inadequate response to ursodeoxycholic acid (UDCA) or cannot tolerate UDCA. It is an oral, selective PPARδ agonist that works by activating the PPARδ receptor to regulate genes involved in reducing liver inflammation, improving bile acid metabolism, and slowing disease progression in primary biliary cholangitis (PBC).What Are Latest Mergers And Acquisitions In The Primary Biliary Cholangitis Therapeutics Market?
In March 2024, Gilead Sciences Inc., a US-based pharmaceutical company, acquired CymaBay Therapeutics Inc. for $4.3 billion. With this acquisition, Gilead aims to strengthen its liver disease portfolio by adding CymaBay’s investigational therapy, seladelpar, a promising treatment for primary biliary cholangitis (PBC), a rare and chronic liver disease. CymaBay Therapeutics Inc. is a US-based pharmaceutical company that develops and provides innovative therapies for primary biliary cholangitis.Regional Insights
North America was the largest region in the primary biliary cholangitis therapeutics market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Primary Biliary Cholangitis Therapeutics Market?
The primary biliary cholangitis therapeutics market consists of sales of Livdelzi, Iqirvo, Volixibat, fibrates, immunosuppressants, and emerging biologics. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Primary Biliary Cholangitis Therapeutics Market Report 2026?
The primary biliary cholangitis therapeutics market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the primary biliary cholangitis therapeutics industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Primary Biliary Cholangitis Therapeutics Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.14 billion |
| Revenue Forecast In 2035 | $1.85 billion |
| Growth Rate | CAGR of 12.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Drug, Mechanism Of Action (MOA), Distribution Channel, Use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Novartis AG, Gilead Sciences Inc., Ipsen Pharma, Intercept Pharmaceuticals, Enanta Pharmaceuticals, Mirum Pharmaceuticals, Calliditas Therapeutics AB, Dr. Falk Pharma GmbH, NGM Biopharmaceuticals Inc., COUR Pharmaceuticals, HighTide Therapeutics Inc., Pliant Therapeutics Inc., Tiziana Life Sciences, Phenex Pharmaceuticals, Synlogic Therapeutics |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
