
Primary Ciliary Dyskinesia (PCD) Market Report 2026
Global Outlook – By Treatment (Antibiotics, Airway Clearance Therapies, Surgical Interventions, Other Treatments), By Method (Genetic Testing, Imaging Techniques, Clinical Evaluation, Lung Function Tests), By Distribution Channel (Direct Sales, Online Pharmacies, Third-party Distributors, Retail Pharmacies), By End-User (Hospitals, Specialty Clinics, Diagnostic Laboratories, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Primary Ciliary Dyskinesia (PCD) Market Overview
• Primary Ciliary Dyskinesia (PCD) market size has reached to $0.49 billion in 2025 • Expected to grow to $0.68 billion in 2030 at a compound annual growth rate (CAGR) of 6.6% • Growth Driver: The Rising Demand For Gene Therapy Is Fueling The Growth Of The Market Due To Advances In Genetic Research And Treatment Development • Market Trend: Advancements In mRNA-Based Therapies For Primary Ciliary Dyskinesia Treatment • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Primary Ciliary Dyskinesia (PCD) Market?
Primary ciliary dyskinesia (PCD) is a rare inherited disorder that impairs cilia's normal function and structure, a microscopic, hair-like structure found in the airways, ears, and reproductive tract. The primary purpose of understanding and diagnosing PCD is to manage symptoms early and improve quality of life through targeted therapies. The main types of primary ciliary dyskinesia (PCD) treatment are antibiotics, airway clearance therapies, surgical interventions, and others. Antibiotics support primary ciliary dyskinesia by targeting respiratory bacterial infections, helping prevent lung damage, and maintaining airway health. It is diagnosed through various methods such as genetic testing, imaging techniques, clinical evaluation, and lung function tests, and is distributed through several distribution channels, including direct sales, online pharmacies, third-party distributors, and retail pharmacies. It is used by various end-users, including hospitals, specialty clinics, diagnostic laboratories, and others.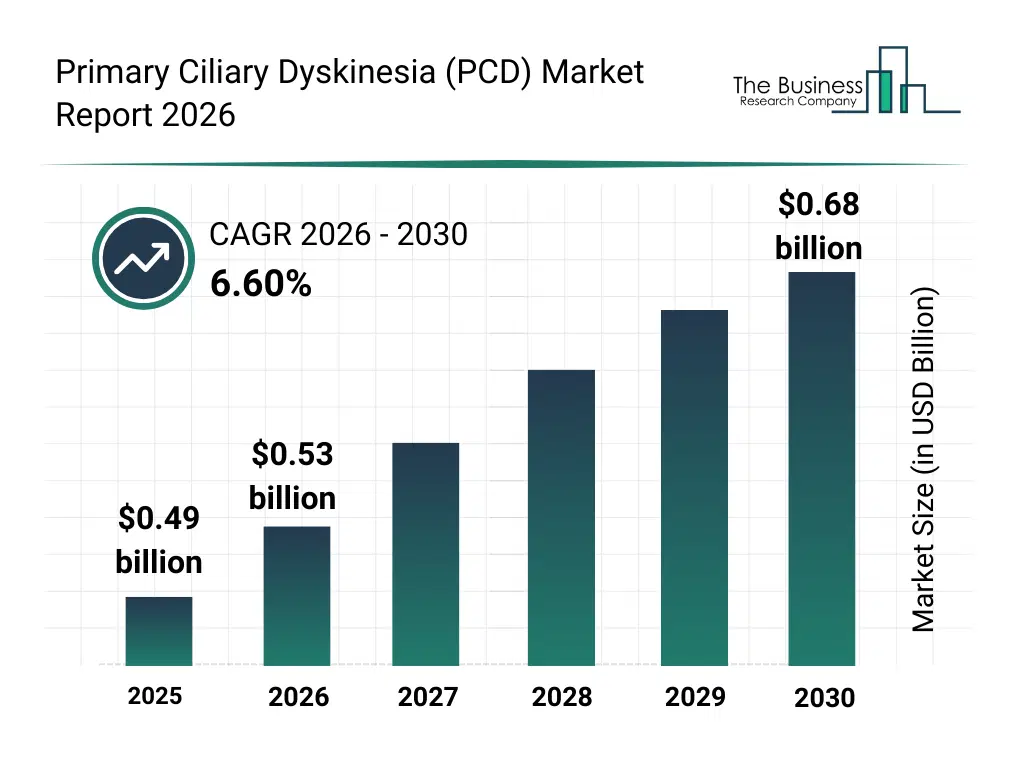
What Is The Primary Ciliary Dyskinesia (PCD) Market Size and Share 2026?
The primary ciliary dyskinesia (pcd) market size has grown strongly in recent years. It will grow from $0.49 billion in 2025 to $0.53 billion in 2026 at a compound annual growth rate (CAGR) of 6.7%. The growth in the historic period can be attributed to limited awareness about pcd, lack of standardized diagnostic protocols, reliance on conventional antibiotic therapies, minimal availability of specialized clinics, low adoption of airway clearance devices.What Is The Primary Ciliary Dyskinesia (PCD) Market Growth Forecast?
The primary ciliary dyskinesia (pcd) market size is expected to see strong growth in the next few years. It will grow to $0.68 billion in 2030 at a compound annual growth rate (CAGR) of 6.6%. The growth in the forecast period can be attributed to development of gene therapy and stem cell interventions, increasing investments in diagnostic laboratories, rising prevalence of rare respiratory disorders, adoption of personalized medicine approaches, integration of digital health and remote monitoring solutions. Major trends in the forecast period include increasing adoption of genetic testing for early diagnosis, rising use of advanced imaging techniques for monitoring, expansion of airway clearance therapy devices, growing demand for personalized antibiotic treatments, focus on innovative therapies such as gene and stem cell therapy.Global Primary Ciliary Dyskinesia (PCD) Market Segmentation
1) By Treatment: Antibiotics, Airway Clearance Therapies, Surgical Interventions, Other Treatments 2) By Method: Genetic Testing, Imaging Techniques, Clinical Evaluation, Lung Function Tests 3) By Distribution Channel: Direct Sales, Online Pharmacies, Third-party Distributors, Retail Pharmacies 4) By End-User: Hospitals, Specialty Clinics, Diagnostic Laboratories, Other End-Users Subsegments: 1) By Antibiotics: Macrolides, Penicillins, Cephalosporins, Fluoroquinolones, Aminoglycosides 2) By Airway Clearance Therapies: Chest Physiotherapy, High-Frequency Chest Wall Oscillation (HFCWO), Positive Expiratory Pressure (PEP) Therapy, Intrapulmonary Percussive Ventilation (IPV), Oscillatory Positive Expiratory Pressure (OPEP) 3) By Surgical Interventions: Sinus Surgery, Tympanostomy Tube Placement, Lung Transplantation 4) By Other Treatments: Mucolytics, Immunomodulatory Therapies, Gene Therapy, Stem Cell TherapyWhat Is The Driver Of The Primary Ciliary Dyskinesia (PCD) Market?
The rising demand for gene therapy is expected to propel the growth of the primary ciliary dyskinesia market going forward. Gene therapy is a treatment that modifies or replaces faulty genes in a patient's cells to cure or prevent diseases. The demand for gene therapy is growing due to progress in genetic research, enabling more accurate treatments that target and correct genetic disorders at the molecular level, enhancing patient outcomes. Researchers are developing gene therapy for Primary Ciliary Dyskinesia (PCD) to correct the genetic mutations that cause faulty cilia function in the respiratory system. This therapy introduces healthy genes to restore normal ciliary movement, improve mucus clearance, and reduce respiratory infections. For instance, in July 2023, according to the American Society of Gene & Cell Therapy (ASGCT), a US-based professional membership organization, in the first quarter of 2023, there were 247 gene therapy programs in Phase II clinical trials, and this number increased by 5%, reaching a total of 260 ongoing Phase II gene therapy trials by the end of the second quarter. Therefore, the rising demand for gene therapy is driving the growth of the primary ciliary dyskinesia market.Key Players In The Global Primary Ciliary Dyskinesia (PCD) Market
Major companies operating in the primary ciliary dyskinesia (pcd) market are Centogene NV, Revvity Inc, Quest Diagnostics Incorporated, Nationwide Children’s Hospital, Children’s Healthcare of Atlanta, National Jewish Health, Guy's and St Thomas' NHS Foundation Trust, University Hospital Southampton, Vanderbilt University Medical Center, Stanford Medicine, Mayo Foundation for Medical Education and Research, UNC School of Medicine, UAB Health System, University Hospital Münster, DNA Labs India Pvt Ltd, Parion Sciences Inc, ReCode Therapeutics Inc, PCD Foundation, PCD Research Consortium, European PCD NetworkGlobal Primary Ciliary Dyskinesia (PCD) Market Trends and Insights
Major companies operating in the primary ciliary dyskinesia market are focusing on developing innovative solutions, such as investigational mRNA-based therapeutics, to improve symptom management and enhance patients’ quality of life. An investigational mRNA-based therapeutic is a developing treatment that utilizes messenger RNA (mRNA) to direct cells to produce proteins that may treat or prevent diseases. This cutting-edge method targets conditions at the molecular level, paving the way for personalized medicine. For instance, in June 2024, ReCode Therapeutics, Inc., a US-based biotechnology company, launched RCT1100, an investigational inhaled mRNA therapy. It received approval from the Food and Drug Administration (FDA) for orphan drug designation to treat primary ciliary dyskinesia. RCT1100 aims to restore ciliary function by delivering DNAI1 mRNA directly to airway cells, addressing a critical unmet need for the estimated 45,000 affected individuals in the U.S., as there are currently no disease-modifying treatments or cures for PCD.What Are Latest Mergers And Acquisitions In The Primary Ciliary Dyskinesia (PCD) Market?
In October 2024, PCD Research, a UK-based charitable organization specializing in primary ciliary dyskinesia (PCD) research, partnered with Nucleic Acid Therapy Accelerator (NATA) to advance therapy for primary ciliary dyskinesia. The partnership aims to co-fund a grant focused on advancing therapies for primary ciliary dyskinesia (PCD), a rare genetic disorder impacting respiratory function. This collaboration aims to support innovative research that enhances treatment options and outcomes for individuals affected by this progressive condition. Nucleic Acid Therapy Accelerator (NATA) is a UK-based research initiative specializing in nucleic acid therapeutics.Regional Outlook
North America was the largest region in the primary ciliary dyskinesia market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Primary Ciliary Dyskinesia (PCD) Market?
The primary ciliary dyskinesia market consists of revenues earned by entities by providing services such as pulmonary rehabilitation, home healthcare services, and genetic counselling. The market value includes the value of related goods sold by the service provider or included within the service offering. The primary ciliary dyskinesia market includes sales of immunofluorescence microscopy, transmission electron tomography, and whole-exome sequencing kits. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Primary Ciliary Dyskinesia (PCD) Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $0.53 billion |
| Revenue Forecast In 2035 | $0.68 billion |
| Growth Rate | CAGR of 6.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Treatment, Method, Distribution Channel, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Centogene NV, Revvity Inc, Quest Diagnostics Incorporated, Nationwide Children’s Hospital, Children’s Healthcare of Atlanta, National Jewish Health, Guy's and St Thomas' NHS Foundation Trust, University Hospital Southampton, Vanderbilt University Medical Center, Stanford Medicine, Mayo Foundation for Medical Education and Research, UNC School of Medicine, UAB Health System, University Hospital Münster, DNA Labs India Pvt Ltd, Parion Sciences Inc, ReCode Therapeutics Inc, PCD Foundation, PCD Research Consortium, European PCD Network |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
