
Prostate Cancer Nuclear Medicine Diagnostics Market Report 2026
Global Outlook – By Type (Single Photon Emission Computed Tomography (SPECT), Photon Emission Computed Tomography (PET)), By Test Type (Preliminary Tests, Confirmatory Tests), By End User (Hospitals, Clinics, Other End Users) - Market Size, Trends, And Global Forecast 2026-2035
Prostate Cancer Nuclear Medicine Diagnostics Market Overview
• Prostate Cancer Nuclear Medicine Diagnostics market size has reached to $0.99 billion in 2025 • Expected to grow to $1.84 billion in 2030 at a compound annual growth rate (CAGR) of 13% • Growth Driver: Fueling Growth In Prostate Cancer Nuclear Medicine Diagnostics Market • Market Trend: Targeted Alpha-Particle Radiotherapy Advances Precision Treatment for Prostate Cancer • North America was the largest region in 2025.What Is Covered Under Prostate Cancer Nuclear Medicine Diagnostics Market?
Prostate cancer nuclear medicine diagnostics refers to the process of using radioactive substances to detect and evaluate prostate cancer. These diagnostics help in the detection, staging, and management of prostate cancer by providing detailed functional information about the disease's extent and characteristics. Prostate cancer nuclear medicine diagnostics involve the use of radioactive substances (radiopharmaceuticals) that are introduced into the body, typically through injection, and emit gamma rays or positrons. The main types of prostate cancer nuclear medicine diagnostics are single photon emission computed tomography (SPECT), and photon emission computed tomography (pet). Single photon emission computed tomography (SPECT) is a nuclear imaging technique that generates detailed, three-dimensional images of the body's functional processes. The various test types include preliminary tests and confirmatory tests. These are used by various end-users such as hospitals, clinics, and others.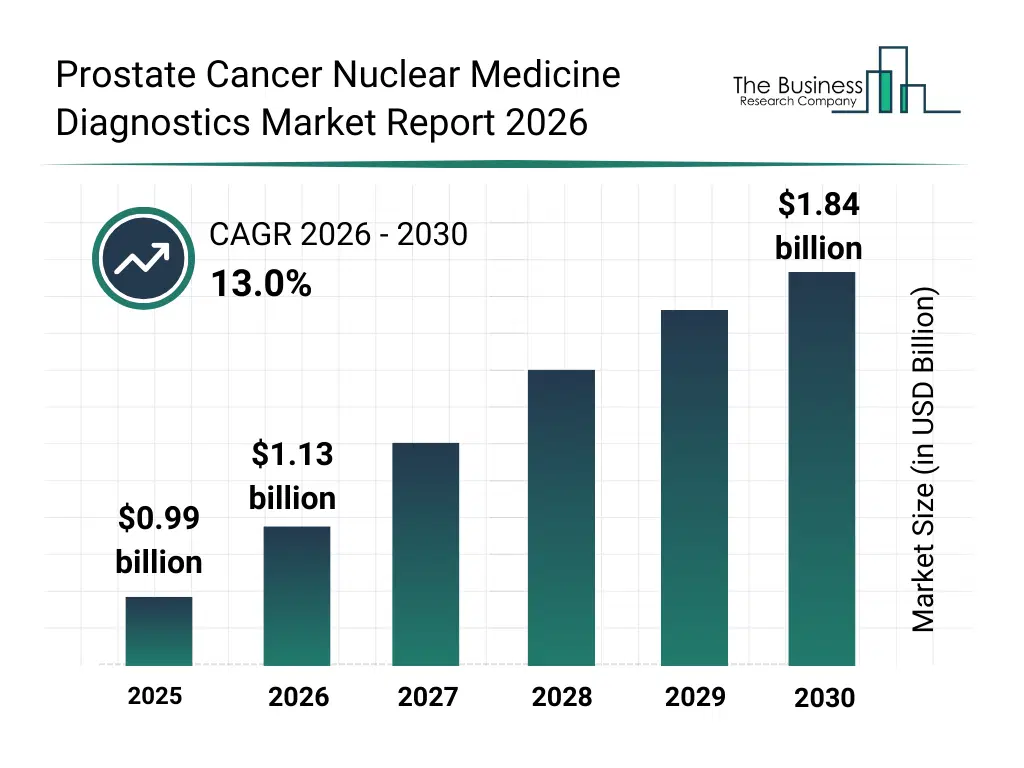
What Is The Prostate Cancer Nuclear Medicine Diagnostics Market Size and Share 2026?
The prostate cancer nuclear medicine diagnostics market size has grown rapidly in recent years. It will grow from $0.99 billion in 2025 to $1.13 billion in 2026 at a compound annual growth rate (CAGR) of 14.6%. The growth in the historic period can be attributed to expansion of nuclear medicine infrastructure, increased use of radiotracers in oncology, availability of gamma camera and pet imaging systems, rising prostate cancer diagnosis rates, growth in hospital-based imaging services.What Is The Prostate Cancer Nuclear Medicine Diagnostics Market Growth Forecast?
The prostate cancer nuclear medicine diagnostics market size is expected to see rapid growth in the next few years. It will grow to $1.84 billion in 2030 at a compound annual growth rate (CAGR) of 13.0%. The growth in the forecast period can be attributed to advancements in radiopharmaceutical development, rising demand for precise cancer staging tools, expansion of personalized oncology diagnostics, increasing investment in hybrid imaging platforms, growing adoption of molecular imaging techniques. Major trends in the forecast period include increasing adoption of pet and spect imaging modalities, rising use of radiopharmaceutical-based diagnostics, growing demand for functional cancer imaging, expansion of hybrid imaging technologies, enhanced focus on accurate disease staging.Global Prostate Cancer Nuclear Medicine Diagnostics Market Segmentation
1) By Type: Single Photon Emission Computed Tomography (SPECT), Photon Emission Computed Tomography (PET) 2) By Test Type: Preliminary Tests, Confirmatory Tests 3) By End User: Hospitals, Clinics, Other End Users Subsegments: 1) By Single Photon Emission Computed Tomography (SPECT): Gamma Camera SPECT, Hybrid SPECT Or CT Imaging 2) By Positron Emission Tomography (PET): PET Or CT Imaging, PET Or MRI ImagingWhat Is The Driver Of The Prostate Cancer Nuclear Medicine Diagnostics Market?
The rise in the prevalence of prostate cancer is expected to propel the growth of the prostate cancer nuclear medicine diagnostics market going forward. Prostate cancer is a type of cancer that occurs in the prostate, a small walnut-shaped gland in males that produces seminal fluid, which nourishes and transports sperm. The rising prevalence of prostate cancer is due to the aging population, lifestyle changes, and genetic factors. Prostate cancer nuclear medicine diagnostics provide valuable insights that enable clinicians to deliver more precise, effective, and personalized care to patients with prostate cancer. For instance, in February 2024, according to the World Health Organization, a Switzerland-based specialized agency responsible for international public health, by 2050, over 35 million new cancer cases are projected, representing a 77% increase from the estimated 20 million cases in 2022. Therefore, the rise in the prevalence of prostate cancer drives the prostate cancer nuclear medicine diagnostics industry.Key Players In The Global Prostate Cancer Nuclear Medicine Diagnostics Market
Major companies operating in the prostate cancer nuclear medicine diagnostics market are Cardinal Health, Bayer AG, Novartis AG, Siemens Healthineers AG, GE Healthcare, Lantheus, Curium Pharma, Telix Pharmaceuticals Ltd, Eckert And Ziegler AG, Point Biopharma Global Inc, Alliance Medical Ltd, SHINE Medical Technologies LLC, PETNET Solution, NorthStar Medical Radioisotopes LLC, Isotope Technologies Garching ITG, Jubilant Pharma Limited, Blue Earth Diagnostics Inc, Institute Of Isotopes Co Ltd, Radiopharm Theranostics, Bracco Diagnostic Inc, NCM USA LLC, ABX Advanced Biochemical Compounds GmbH, Theragnostics Ltd, SpectronRx IncGlobal Prostate Cancer Nuclear Medicine Diagnostics Market Trends and Insights
Major companies operating in the prostate cancer nuclear medicine market are focusing on developing advanced solutions such as targeted alpha-particle radiotherapy to enhance treatment precision and minimize off-target effects. Targeted alpha-particle radiotherapy uses radioactive isotopes that emit alpha particles to selectively destroy cancer cells while sparing healthy tissue, providing a highly localized and effective treatment option. For instance, in March 2025, Actinium Pharmaceuticals Inc., a US-based developer of antibody radiation conjugates and other targeted radiotherapies, launched ATNM-400 featuring actinium-225 (Ac-225) radiotherapy. The therapy targets a non-prostate-specific membrane antigen that is overexpressed in prostate cancer patients and provides an alternative for individuals resistant to PSMA-targeted therapies such as Pluvicto. Key features of ATNM-400 include the use of the alpha-particle emitter Ac-225, which can reduce off-target effects compared with beta-particle therapies, and its targeting of a distinct cancer marker to broaden treatment applicability. The launch includes preclinical data demonstrating biodistribution, imaging, and efficacy analyses across various dose levels, with additional results expected from Pluvicto-resistant prostate cancer models. ATNM-400 is positioned to advance precision radiotherapy and improve treatment options for patients with prostate cancer.What Are Latest Mergers And Acquisitions In The Prostate Cancer Nuclear Medicine Diagnostics Market?
In March 2023, Life Healthcare Group, a South Africa-based healthcare company, acquired TheraMed Nuclear for an undisclosed amount. With this acquisition, Life Healthcare enhances its diagnostic imaging services, particularly in detecting and treating diseases like organ dysfunction and cancer. TheraMed Nuclear is a South Africa-based company that offers prostate cancer nuclear medicine diagnostics.Regional Outlook
North America was the largest region in the prostrate cancer nuclear medicine diagnostics market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Prostate Cancer Nuclear Medicine Diagnostics Market?
The prostate cancer nuclear medicine diagnostics market includes revenues earned by entities by bone scans, prostate-specific membrane antigen (PSMA) PET scan, bone scintigraphy, and CT scans. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Prostate Cancer Nuclear Medicine Diagnostics Market Report 2026?
The prostate cancer nuclear medicine diagnostics market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the prostate cancer nuclear medicine diagnostics industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Prostate Cancer Nuclear Medicine Diagnostics Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.13 billion |
| Revenue Forecast In 2035 | $1.84 billion |
| Growth Rate | CAGR of 14.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Test Type, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Cardinal Health, Bayer AG, Novartis AG, Siemens Healthineers AG, GE Healthcare, Lantheus, Curium Pharma, Telix Pharmaceuticals Ltd, Eckert And Ziegler AG, Point Biopharma Global Inc, Alliance Medical Ltd, SHINE Medical Technologies LLC, PETNET Solution, NorthStar Medical Radioisotopes LLC, Isotope Technologies Garching ITG, Jubilant Pharma Limited, Blue Earth Diagnostics Inc, Institute Of Isotopes Co Ltd, Radiopharm Theranostics, Bracco Diagnostic Inc, NCM USA LLC, ABX Advanced Biochemical Compounds GmbH, Theragnostics Ltd, SpectronRx Inc |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
