
Pulmonology Devices Market Report 2026
Global Outlook – By Product Type (Endobronchial Ultrasound (EBUS) Needles, Pulmonary Biopsy Devices, Airway Stents, Single-Use Bronchoscopes, Other Product Types), By Indication (Chronic Obstructive Pulmonary Disease (COPD), Lung Cancer, Tracheal And Bronchial Stenosis, Foreign Body Extraction, Other Indications), By End-User (Pulmonology Clinics, Hospitals, Ambulatory Surgical Centers) – Market Size, Trends, Strategies, and Forecast to 2035
Pulmonology Devices Market Overview
• Pulmonology Devices market size has reached to $1.5 billion in 2025 • Expected to grow to $2.36 billion in 2030 at a compound annual growth rate (CAGR) of 9.3% • Growth Driver: Respiratory Disease Prevalence Drives Pulmonology Devices Market Growth • Market Trend: Enhanced Bi-level Respiratory Support For Sleep Apnea In Pulmonology Devices Market • North America was the largest region in 2025.What Is Covered Under Pulmonology Devices Market?
Pulmonology devices are medical instruments and equipment used in the diagnosis, treatment, and management of respiratory system conditions and diseases. These devices play a crucial role in assessing lung function, monitoring respiratory health, providing therapeutic interventions, and supporting patients with respiratory disorders. The main types of products in the pulmonology devices market are endobronchial ultrasound (EBUS) needles, pulmonary biopsy devices, airway stents, single-use bronchoscopes, and others. Endobronchial ultrasound (EBUS) needles are specialized instruments used during bronchoscopy to collect tissue samples from the lungs and adjacent lymph nodes. The various indications include chronic obstructive pulmonary disease (COPD), lung cancer, tracheal and bronchial stenosis, foreign body extraction, and others. The devices are utilized in pulmonology clinics, hospitals, and ambulatory surgical centers.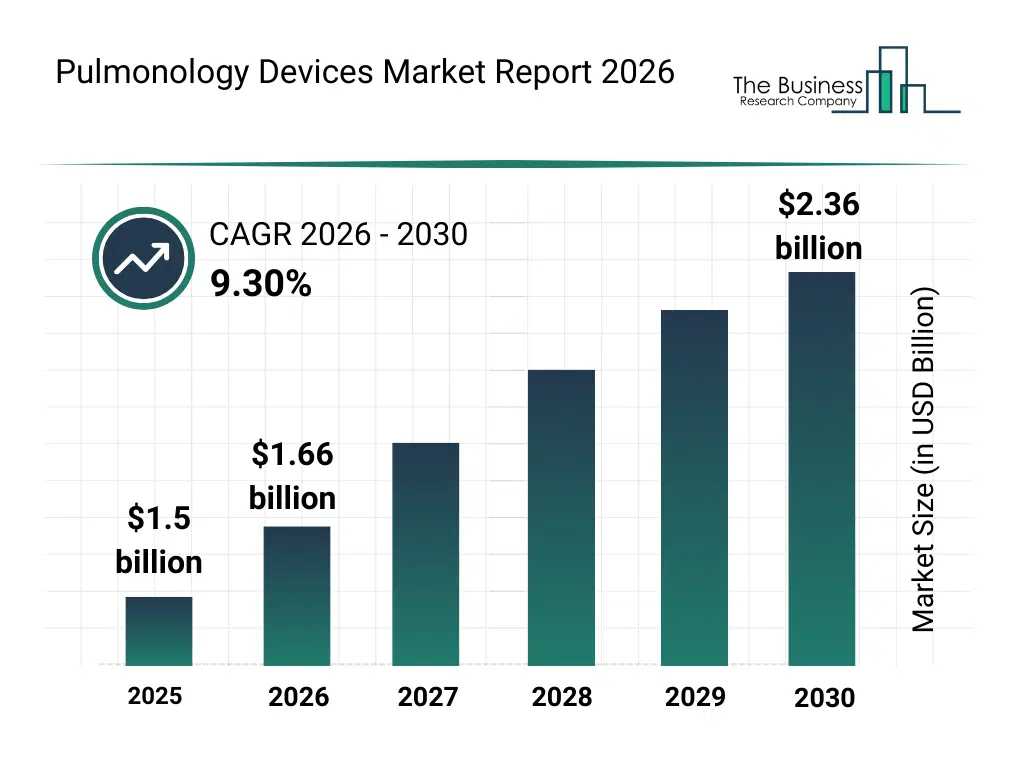
What Is The Pulmonology Devices Market Size and Share 2026?
The pulmonology devices market size has grown rapidly in recent years. It will grow from $1.5 billion in 2025 to $1.66 billion in 2026 at a compound annual growth rate (CAGR) of 10.4%. The growth in the historic period can be attributed to rising prevalence of respiratory diseases, increasing hospital-based pulmonary procedures, growth in diagnostic bronchoscopy adoption, expansion of pulmonology specialty clinics, availability of advanced airway management devices.What Is The Pulmonology Devices Market Growth Forecast?
The pulmonology devices market size is expected to see strong growth in the next few years. It will grow to $2.36 billion in 2030 at a compound annual growth rate (CAGR) of 9.3%. The growth in the forecast period can be attributed to growing geriatric population with respiratory disorders, increasing demand for early lung cancer detection, rising adoption of disposable medical devices, expansion of outpatient pulmonary care, technological advancements in pulmonary diagnostics. Major trends in the forecast period include increasing adoption of single-use bronchoscopes, rising demand for minimally invasive pulmonary diagnostics, growing integration of advanced imaging-guided biopsy devices, expansion of portable pulmonary function testing equipment, enhanced focus on infection control and patient safety.Global Pulmonology Devices Market Segmentation
1) By Product Type: Endobronchial Ultrasound (EBUS) Needles, Pulmonary Biopsy Devices, Airway Stents, Single-Use Bronchoscopes, Other Product Types 2) By Indication: Chronic Obstructive Pulmonary Disease (COPD), Lung Cancer, Tracheal And Bronchial Stenosis, Foreign Body Extraction, Other Indications 3) By End-User: Pulmonology Clinics, Hospitals, Ambulatory Surgical Centers Subsegments: 1) By Endobronchial Ultrasound (EBUS) Needles: Radial EBUS Needles, Linear EBUS Needles, Biopsy EBUS Needles, EBUS Needles With Safety Features, EBUS Needles For Peripheral Lung Lesions 2) By Pulmonary Biopsy Devices: Core Biopsy Needles, Fine Needle Aspiration (FNA) Biopsy Devices, Vacuum-Assisted Biopsy Devices, Transthoracic Biopsy Devices, Endoscopic Pulmonary Biopsy Devices 3) By Airway Stents: Self-Expanding Airway Stents, Balloon-Expandable Airway Stents, Silicone Airway Stents, Metal Airway Stents, Hybrid Airway Stents (Silicone + Metal) 4) By Single-Use Bronchoscopes: Single-Use Flexible Bronchoscopes, Single-Use Rigid Bronchoscopes, Single-Use Pediatric Bronchoscopes, Single-Use Disposable Video Bronchoscopes, Single-Use Bronchoscopes With Integrated Suction 5) By Other Product Types: Pulmonary Function Testing Devices, Nebulizers For Pulmonary Disease Treatment, Spirometers, Pulmonary Inhalation Devices, Non-Invasive Ventilation (NIV) DevicesWhat Is The Driver Of The Pulmonology Devices Market?
The rise in the prevalence of respiratory diseases is expected to propel the growth of the pulmonology devices market going forward. Respiratory diseases are a group of medical conditions that affect the organs and tissues involved in breathing, including the airways, lungs, and respiratory muscles. The cases of respiratory diseases are occurring due to rising air pollution, smoking, and occupational hazards. Pulmonology devices aid in respiratory disease management by enabling accurate diagnosis, continuous monitoring, and effective treatment through tools like spirometers, inhalers, pulse oximeters, and ventilators, ultimately improving patient outcomes and quality of life. For instance, in December 2023, according to a research study published by JAMA Network Open, a medical journal by the American Medical Association, the global burden of chronic obstructive pulmonary disease (COPD) projected that the cases worldwide could reach nearly 600 million by 2050, marking a 23% increase in prevalence. Therefore, the rise in the prevalence of respiratory diseases is driving the growth of the pulmonology devices industry.Key Players In The Global Pulmonology Devices Market
Major companies operating in the pulmonology devices market are Medtronic, Koninklijke Philips N.V., GE Healthcare, Boston Scientific Corporation, Medline Industries LP, Olympus Corporation, Omron Healthcare, ResMed, Getinge, Fisher And Paykel Healthcare Limited, Ambu A/S, Vyaire Medical Inc., Drive DeVilbiss Healthcare, PENTAX Medical, Hamilton Medical, Nonin Medical, CAIRE Inc., Pulmonx Corporation, Verathon Inc., Draegerwerk AG And Co. KGaA, NIOXGlobal Pulmonology Devices Market Trends and Insights
Major companies operating in the pulmonology devices market are developing innovative products with advanced technologies, such as bilevel positive airway pressure (BiPAP) technology, to enhance patient comfort, improve treatment efficacy, and address various respiratory conditions. The BiPAP technology provides two pressure settings, one for inhalation (IPAP) and one for exhalation (EPAP), which can be more comfortable for users with continuous pressure. For instance, in February 2024, ResMed, a US-based company providing cloud-connected devices, launched AirCurve 11 bilevel sleep apnea devices, a line of advanced bilevel positive airway pressure (BiPAP) machines aimed to improve comfort and compliance for sleep apnea and other respiratory disorders. The AirCurve 11 VAuto model provides higher and lower pressure during exhalation, designed to better align with the patient's natural breathing pattern. The AirCurve 11 ASV is an adaptive servo-ventilation (ASV) device that tracks 13 points on each breath cycle 50 times per second to stabilize the patient's ventilation.What Are Latest Mergers And Acquisitions In The Pulmonology Devices Market?
In January 2025, Molex LLC, a US-based electronics and manufacturing company, acquired Vectura Group Ltd. for an undisclosed amount. With this acquisition, Phillips Medisize, Molex’s CDMO arm, significantly bolsters its inhalation drug‑delivery capabilities, enabling a broader portfolio of combination products and device development. Vectura Group Limited is a UK-based pharmaceutical technology company that specializes in providing provides pulmonology devices, specifically inhalation drug delivery solutions for respiratory conditions like asthma and COPD.Regional Outlook
North America was the largest region in the pulmonology devices market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Pulmonology Devices Market?
The pulmonary devices market consists of sales of spirometers, peak flow meters, nebulizers, and pulse oximeters. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Pulmonology Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.66 billion |
| Revenue Forecast In 2035 | $2.36 billion |
| Growth Rate | CAGR of 10.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Indication, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Medtronic, Koninklijke Philips N.V., GE Healthcare, Boston Scientific Corporation, Medline Industries LP, Olympus Corporation, Omron Healthcare, ResMed, Getinge, Fisher And Paykel Healthcare Limited, Ambu A/S, Vyaire Medical Inc., Drive DeVilbiss Healthcare, PENTAX Medical, Hamilton Medical, Nonin Medical, CAIRE Inc., Pulmonx Corporation, Verathon Inc., Draegerwerk AG And Co. KGaA, NIOX |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
