
Pyrogen Testing Market Report 2026
Global Outlook – By Test Type (LAL Test, In Vitro Test, Rabbit Test), By Product And Service (Assays, Kits, And Reagents, Instruments, Services), By Application (Pharmaceutical And Biologics, Medical Devices, Other Applications) - Market Size, Trends, And Global Forecast 2026-2035
Pyrogen Testing Market Overview
• Pyrogen Testing market size has reached to $1.91 billion in 2025 • Expected to grow to $3.42 billion in 2030 at a compound annual growth rate (CAGR) of 12.2% • Growth Driver: Rising Investments In R&D By Pharmaceutical Companies Boost Pyrogen Testing Market • Market Trend: Rapid Monocyte Activation Test Systems For Pyrogen Testing, Reducing Reliance On Animal Testing • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Pyrogen Testing Market?
The pyrogen testing provide pyrogen testing to check the presence or absence of pyrogens in all aqueous parenteral. Pyrogen testing determines the presence of bacterial toxins in vaccines, biotechnological products, and drugs, which induces fever in humans. It also determines the presence of microbes and their metabolites in drugs during the manufacturing process. The main types of pyrogen tests are LAL test, in vitro test, and rabbit test. The LAL (Limulus Amebocyte Lysate) test is a test for the determination of bacterial endotoxins, which uses the amebocyte lysate of the Limulus crab. The rabbit pyrogen test is a test in which a small amount of batched test material is injected into a rabbit’s bloodstream, and the body temperature of the rabbit is monitored. The products used for pyrogen tests include assays, kits, and reagents, instruments and related services, and are used in pharmaceutical and biologics, medical devices, and other industries.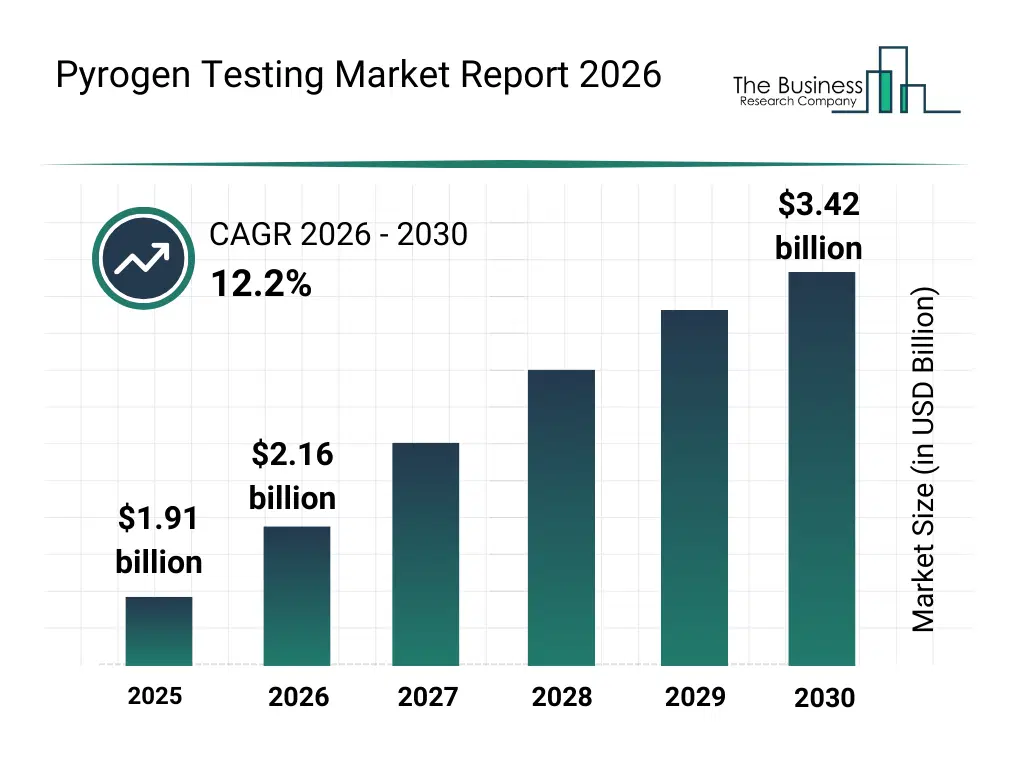
What Is The Pyrogen Testing Market Size and Share 2026?
The pyrogen testing market size has grown rapidly in recent years. It will grow from $1.91 billion in 2025 to $2.16 billion in 2026 at a compound annual growth rate (CAGR) of 13.0%. The growth in the historic period can be attributed to expansion of pharmaceutical manufacturing capacity, increased production of injectable drugs and vaccines, regulatory mandates for pyrogen testing, reliance on LAL-based assays, growth of biologics development.What Is The Pyrogen Testing Market Growth Forecast?
The pyrogen testing market size is expected to see rapid growth in the next few years. It will grow to $3.42 billion in 2030 at a compound annual growth rate (CAGR) of 12.2%. The growth in the forecast period can be attributed to increasing adoption of rFC and MAT assays, rising investments in biopharmaceutical R&D, expansion of cell and gene therapy manufacturing, growing focus on animal-free testing solutions, increasing automation in quality control laboratories. Major trends in the forecast period include increasing adoption of recombinant and in vitro pyrogen tests, rising shift away from animal-based testing methods, growing demand for rapid and high-sensitivity detection assays, expansion of automated endotoxin testing instruments, enhanced focus on regulatory compliance in biopharmaceutical manufacturing.Global Pyrogen Testing Market Segmentation
1) By Test Type: LAL Test, In Vitro Test, Rabbit Test 2) By Product And Service: Assays, Kits, And Reagents, Instruments, Services 3) By Application: Pharmaceutical And Biologics, Medical Devices, Other Applications Subsegments: 1) By Test Limulus Amebocyte Lysate (LAL) Test: Gel Clot LAL Test, Chromogenic LAL Test, Turbidimetric LAL Test 2) By In Vitro Test: Monocyte Activation Test (MAT), Recombinant Factor C (rFC) Assay, Other In Vitro Tests 3) By Rabbit Test: Pyrogen Test in Rabbits, Other Animal-Based TestsWhat Are The Drivers Of The Pyrogen Testing Market?
The rising investments in research and development (R&D) by pharmaceutical and biotechnology companies are expected to propel the growth of the pyrogen testing market going forward. Pyrogen testing is the assessment of drugs and biologics to ensure they are free from fever-inducing contaminants, such as endotoxins, and is critical for safe product manufacturing. The increase in R&D spending is driven by the need to develop advanced therapies and biologics that require stringent quality control and safety testing. Pyrogen testing drives rising investments in R&D by ensuring the safety and regulatory compliance of new drugs and biologics, encouraging pharmaceutical and biotechnology companies to develop innovative therapies with confidence. For instance, in February 2025, according to the National Center for Science and Engineering Statistics (NCSES), a US-based government statistical agency, U.S. research and development expenditures rose from $892 billion in 2022 to $940 billion in 2023, reflecting an increase of $48 billion. Therefore, the rising R&D investments by pharmaceutical companies are driving the growth of the pyrogen testing industry. The rising incidence of tuberculosis (TB) is expected to propel the growth of the pyrogen testing market going forward. Tuberculosis is an infectious disease caused by the bacterium Mycobacterium tuberculosis, which primarily affects the lungs but can spread to other organs, and remains one of the leading causes of preventable death worldwide. The rise in TB cases is driven by factors such as population growth, urbanization, and gaps in healthcare access, leading to higher infection rates in vulnerable populations. Pyrogen testing supports pharmaceutical safety by ensuring that drugs and medical devices are free from fever-inducing contaminants, such as bacterial endotoxins, thereby protecting patients. For instance, in May 2025, according to the World Health Organization (WHO), a Switzerland-based health standards organization, in 2023, approximately 10.8 million people developed TB globally, resulting in 1.25 million deaths. These statistics highlight the growing burden of a highly infectious disease and underscore the critical need for rigorous safety testing in pharmaceutical manufacturing. Therefore, the rising incidence of TB is driving the growth of the pyrogen testing industry.Key Players In The Global Pyrogen Testing Market
Major companies operating in the pyrogen testing market are Thermo Fisher Scientific Inc., Merck KGaA, Charles River Laboratories Inc., Lonza Group Ltd., Ellab A S, Nelson Laboratories LLC, Associates of Cape Cod Inc., GenScript Biotech Corporation, bioMerieux SA, Hyglos GmbH, Wako Chemicals USA Inc., Pyrostar International Inc., Eurofins Scientific SE, SGS SA, WuXi AppTec Group, Sigma Aldrich Co. LLC, Bio Rad Laboratories Inc., Microcoat Biotechnologie GmbH, Sartorius AG, Pacific Biolabs Inc., Toxikon Corporation, STERIS Corporation, Avance Biosciences Inc., BioReliance Corporation, Microbiological Consulting LLC, Microbac Laboratories Inc., North American Science Associates Inc., Pace Analytical Life Sciences LLCGlobal Pyrogen Testing Market Trends and Insights
Major companies operating in the pyrogen testing market are developing innovative products, such as the sustainable monocyte activation test (MAT) systems to advance the business offering and to increase market reach. The monocyte activation test (MAT) System is an in vitro assay used for pyrogen testing in the biopharmaceutical and medical device industry. For instance, in October 2023, Lonza, a Swiss multinational manufacturing company, launched two new rapid monocyte activation test (MAT) systems, the PyroCell MAT Rapid System and the PyroCell MAT Human Serum (HS) Rapid System. The systems which will replace Lonza’s current MAT system kit offerings, contain the new PeliKine Human IL-6 Rapid ELISA Kit that minimises hands-on time and reduces time-to-results from two days to two hours. The new tests give pharma manufacturers easier, faster, and more efficient MAT testing options for product safety testing while helping to reduce the reliance on animals.Regional Outlook
North America was the largest region in the pyrogen testing market in 2025. Asia-Pacific is expected to continue to be the fastest growing region in the pyrogen testing market share during the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Pyrogen Testing Market?
The pyrogen testing market includes revenues earned by entities by endotoxin testing and monocyte activation testing (MAT). The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Pyrogen Testing Market Report 2026?
The pyrogen testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the pyrogen testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Pyrogen Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.16 billion |
| Revenue Forecast In 2035 | $3.42 billion |
| Growth Rate | CAGR of 13.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Test Type, Product And Service, Application |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Merck KGaA, Charles River Laboratories Inc., Lonza Group Ltd., Ellab A S, Nelson Laboratories LLC, Associates of Cape Cod Inc., GenScript Biotech Corporation, bioMerieux SA, Hyglos GmbH, Wako Chemicals USA Inc., Pyrostar International Inc., Eurofins Scientific SE, SGS SA, WuXi AppTec Group, Sigma Aldrich Co. LLC, Bio Rad Laboratories Inc., Microcoat Biotechnologie GmbH, Sartorius AG, Pacific Biolabs Inc., Toxikon Corporation, STERIS Corporation, Avance Biosciences Inc., BioReliance Corporation, Microbiological Consulting LLC, Microbac Laboratories Inc., North American Science Associates Inc., Pace Analytical Life Sciences LLC |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
