
Rapid Diagnostic Test Kits Market Report 2026
Global Outlook – By Product Type (Antigen Test Kits, Antibody Test Kits, Nucleic Acid Test Kits, Multi-Analyte Test Kits), By Technology (Fluorescent Immunoassay, Enzyme-Linked Immunosorbent Assay (ELISA), Colloidal Gold-Based Test, Lateral Flow Assays), By Application (Infectious Disease Testing, Cardiology Testing, Oncology Testing, Pregnancy And Fertility Testing, Toxicology Testing, Glucose Monitoring, Other Applications), By End-User (Hospitals, Diagnostic Laboratories, Home Care Settings, Pharmacies) - Market Size, Trends, And Global Forecast 2026-2035
Rapid Diagnostic Test Kits Market Overview
• Rapid Diagnostic Test Kits market size has reached to $24.08 billion in 2025 • Expected to grow to $32.22 billion in 2030 at a compound annual growth rate (CAGR) of 5.9% • Growth Driver: Increasing Prevalence Of Infectious Diseases Driving The Market Growth Due To Antimicrobial Resistance • Market Trend: Technological Innovations Drive Non-Invasive And Convenient Early Detection Alternatives • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Rapid Diagnostic Test Kits Market?
Rapid diagnostic test kits are portable, easy-to-use medical tools designed to quickly detect the presence or absence of specific diseases, infections, or health conditions, often delivering results within minutes, making them valuable for point-of-care testing, early diagnosis, and immediate treatment decisions in both clinical and remote settings. The main product type of rapid diagnostic test kits includes antigen test kits, antibody test kits, nucleic acid test kits, and multi-analyte test kits. Antigen test kits are diagnostic tools that detect specific proteins from a pathogen, such as a virus, to quickly identify an active infection. They are categorized by technology into fluorescent immunoassay, enzyme-linked immunosorbent assay, colloidal gold-based test, and lateral flow assays. They are divided by application into infectious disease testing, cardiology testing, oncology testing, pregnancy and fertility testing, toxicology testing, glucose monitoring, and others, and cater to end-users such as hospitals, diagnostic laboratories, home care settings, and pharmacies.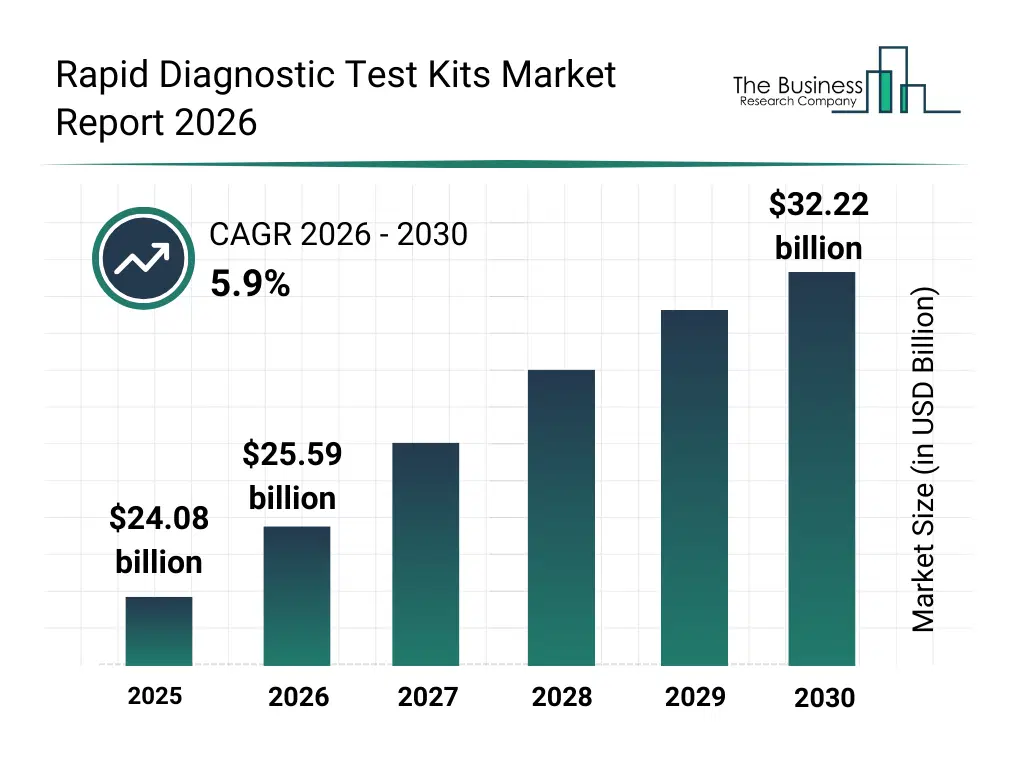
What Is The Rapid Diagnostic Test Kits Market Size and Share 2026?
The rapid diagnostic test kits market size has grown strongly in recent years. It will grow from $24.08 billion in 2025 to $25.59 billion in 2026 at a compound annual growth rate (CAGR) of 6.3%. The growth in the historic period can be attributed to increasing burden of infectious diseases, expansion of decentralized healthcare delivery, rising demand for quick diagnostic results, growth in outpatient and homecare testing, availability of lateral flow and immunoassay technologies.What Is The Rapid Diagnostic Test Kits Market Growth Forecast?
The rapid diagnostic test kits market size is expected to see strong growth in the next few years. It will grow to $32.22 billion in 2030 at a compound annual growth rate (CAGR) of 5.9%. The growth in the forecast period can be attributed to increasing investments in rapid diagnostic innovation, rising adoption of self-testing kits, expansion of infectious disease surveillance programs, growing demand for affordable diagnostics in emerging markets, increasing integration of digital readout technologies. Major trends in the forecast period include increasing adoption of point-of-care testing solutions, rising demand for multiplex rapid test kits, growing use of home-based diagnostic testing, expansion of portable and disposable test formats, enhanced focus on early disease detection.Global Rapid Diagnostic Test Kits Market Segmentation
1) By Product Type: Antigen Test Kits, Antibody Test Kits, Nucleic Acid Test Kits, Multi-Analyte Test Kits 2) By Technology: Fluorescent Immunoassay, Enzyme-Linked Immunosorbent Assay (ELISA), Colloidal Gold-Based Test, Lateral Flow Assays 3) By Application: Infectious Disease Testing, Cardiology Testing, Oncology Testing, Pregnancy And Fertility Testing, Toxicology Testing, Glucose Monitoring, Other Applications 4) By End-User: Hospitals, Diagnostic Laboratories, Home Care Settings, Pharmacies Subsegments: 1) By Antigen Test Kits: COVID-19 Antigen Test Kits, Influenza Antigen Test Kits, Malaria Antigen Test Kits, Respiratory Syncytial Virus Antigen Test Kits, Streptococcus Antigen Test Kits 2) By Antibody Test Kits: HIV Antibody Test Kits, Hepatitis Antibody Test Kits, COVID-19 Antibody Test Kits, Dengue Antibody Test Kits, Syphilis Antibody Test Kits 3) By Nucleic Acid Test Kits: HIV Nucleic Acid Test Kits, Hepatitis B And C Nucleic Acid Test Kits, Human Papillomavirus Nucleic Acid Test Kits, Tuberculosis Nucleic Acid Test Kits, COVID-19 Nucleic Acid Test Kits 4) By Multi-Analyte Test Kits: Respiratory Panel Test Kits, Sexually Transmitted Infections Panel Test Kits, Gastrointestinal Panel Test Kits, Febrile Illness Panel Test Kits, Sepsis Marker Panel Test KitsWhat Is The Driver Of The Rapid Diagnostic Test Kits Market?
The increasing prevalence of infectious diseases is expected to propel the growth of the rapid diagnostic test kits market going forward. Infectious diseases refer to illnesses caused by pathogenic microorganisms such as bacteria, viruses, fungi, or parasites that can spread directly or indirectly from one person to another. The rising prevalence of infectious diseases is driven by antimicrobial resistance and evolving pathogen strains, as widespread antibiotic use has led to drug-resistant bacterial infections while viruses continue to mutate, creating new variants that evade existing immunity and require updated detection methods. Growing infectious disease prevalence supports increased utilization of point-of-care testing devices that enable rapid identification of pathogens, allowing healthcare providers to quickly diagnose infections, initiate appropriate treatment, and implement timely infection control measures. For instance, in March 2024, according to the Centers for Disease Control and Prevention, a US-based government agency, the number of tuberculosis cases increased from 8,320 in 2022 to 9,615 in 2023, representing a rise of 1,295 cases. Therefore, the increasing prevalence of infectious diseases is driving the growth of the rapid diagnostic test kits industry.Key Players In The Global Rapid Diagnostic Test Kits Market
Major companies operating in the rapid diagnostic test kits market are F Hoffmann La Roche Ltd, Thermo Fisher Scientific Inc, Abbott Laboratories, Danaher Corporation, Siemens Healthineers AG, Becton Dickinson and Company, Hologic Inc, bioMerieux SA, QuidelOrtho Corporation, Bio Rad Laboratories Inc, QIAGEN NV, Cepheid Inc, OraSure Technologies Inc, Nova Biomedical Corporation, Sekisui Diagnostics LLC, EKF Diagnostics Holdings plc, Trinity Biotech plc, ACON Laboratories Inc, Fluxergy LLC, Lumos Diagnostics Holdings Limited, Abingdon Health plc, Abionic SAGlobal Rapid Diagnostic Test Kits Market Trends and Insights
Major companies operating in the rapid diagnostic test kits market are focusing on developing novel solutions, such as saliva-based pregnancy test kits, to gain a competitive edge in the market. Saliva-based pregnancy test kits are diagnostic tools designed to detect pregnancy by analyzing a woman's saliva for the presence of hormonal changes, particularly human chorionic gonadotropin (hCG), which is produced during pregnancy. For instance, in February 2023, Abingdon Health plc, a UK-based, near-patient diagnostics company, launched Salistick, the first rapid saliva-based pregnancy test in the UK and Ireland, marking a significant innovation in home pregnancy testing by offering a cleaner, more convenient alternative to traditional urine-based tests. This highly accurate test detects the pregnancy hormone β-hCG in saliva, allowing women to perform early pregnancy detection anywhere and anytime, while also enabling a more personal and shareable testing experience.What Are Latest Mergers And Acquisitions In The Rapid Diagnostic Test Kits Market?
In June 2023, Avacta Group, a UK-based biotechnology company, acquired Coris Bioconcept SRL for $8.42 million. Through this acquisition, Avacta aims to expand its diagnostics portfolio by integrating Coris’s rapid test products and expertise, enhancing its capabilities in both centralized laboratory and decentralized point-of-care testing, while strengthening its position in the growing markets of infectious disease and antimicrobial resistance diagnostics. Coris BioConcept SRL is a Belgium-based company that offers rapid diagnostic kits for the detection of human respiratory, gastroenteric, and blood-borne pathogens.Regional Outlook
North America was the largest region in the rapid diagnostic test kits market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Rapid Diagnostic Test Kits Market?
The rapid diagnostic test kits market consists of sales of influenza test kits, malaria test kits, pregnancy test kits, glucose monitoring kits, hepatitis test kits, and dengue test kits. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Rapid Diagnostic Test Kits Market Report 2026?
The rapid diagnostic test kits market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the rapid diagnostic test kits industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Rapid Diagnostic Test Kits Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $25.59 billion |
| Revenue Forecast In 2035 | $32.22 billion |
| Growth Rate | CAGR of 6.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Technology, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F Hoffmann La Roche Ltd, Thermo Fisher Scientific Inc, Abbott Laboratories, Danaher Corporation, Siemens Healthineers AG, Becton Dickinson and Company, Hologic Inc, bioMerieux SA, QuidelOrtho Corporation, Bio Rad Laboratories Inc, QIAGEN NV, Cepheid Inc, OraSure Technologies Inc, Nova Biomedical Corporation, Sekisui Diagnostics LLC, EKF Diagnostics Holdings plc, Trinity Biotech plc, ACON Laboratories Inc, Fluxergy LLC, Lumos Diagnostics Holdings Limited, Abingdon Health plc, Abionic SA |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
