
Rare Gastrointestinal Diseases Treatment Market Report 2026
Global Outlook – By Type (Drugs, Device), By Route Of Administration (Oral, Intravenous (IV) Or Injectable, Subcutaneous, Other Routes Of Administration), By Application (Eosinophilic Gastroenteritis, Hercksprung's Disease, Chronic Intestinal Pseudo-Obstruction), By End User (Hospitals, Research Institutes, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Rare Gastrointestinal Diseases Treatment Market Overview
• Rare Gastrointestinal Diseases Treatment market size has reached to $1.45 billion in 2025 • Expected to grow to $1.91 billion in 2030 at a compound annual growth rate (CAGR) of 5.6% • Growth Driver: Rising Diagnostic Testing Fueling The Growth Of The Market Due To Early And Accurate Disease Detection • Market Trend: Innovative Antibody Blocks Disease Pathways To Improve Outcomes • North America was the largest region in 2025.What Is Covered Under Rare Gastrointestinal Diseases Treatment Market?
Rare gastrointestinal (GI) diseases treatment refers to the specialized medical care, therapies, and interventions designed to manage uncommon digestive system disorders that affect a small portion of the population. The main purpose of this treatment is to alleviate symptoms, prevent complications, improve quality of life, and, where possible, achieve long-term disease control or remission. The main types of rare gastrointestinal diseases treatment are drugs and devices. Drugs are substances used to prevent, treat, or manage diseases and medical conditions, including specialized therapies such as biologics and small molecules. They target specific symptoms, reduce inflammation, and improve overall patient health and quality of life. These treatments can be administered via various routes, including oral, intravenous (IV), subcutaneous, and others, and find applications in eosinophilic gastroenteritis, Hirschsprung's disease, and chronic intestinal pseudo-obstruction, serving end users such as hospitals, research institutes, and others.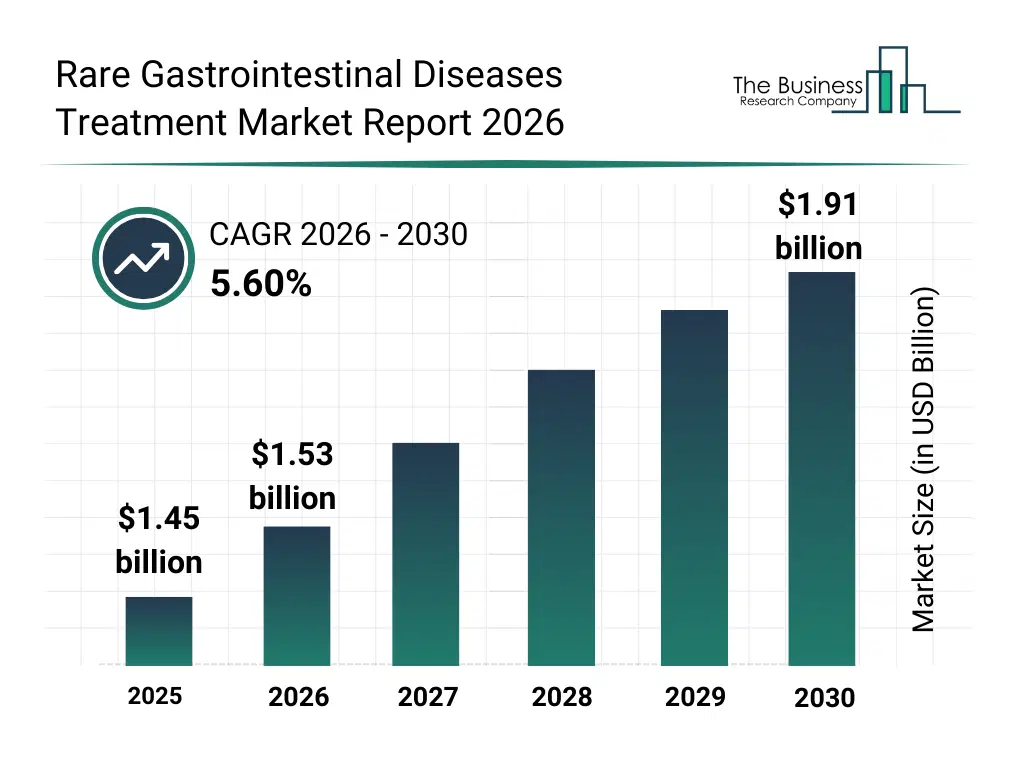
What Is The Rare Gastrointestinal Diseases Treatment Market Size and Share 2026?
The rare gastrointestinal diseases treatment market size has grown strongly in recent years. It will grow from $1.45 billion in 2025 to $1.53 billion in 2026 at a compound annual growth rate (CAGR) of 5.9%. The growth in the historic period can be attributed to increasing recognition of rare GI disorders, expansion of specialty gastroenterology centers, availability of immunosuppressive and anti-inflammatory drugs, rising clinical research activity, improved diagnostic accuracy.What Is The Rare Gastrointestinal Diseases Treatment Market Growth Forecast?
The rare gastrointestinal diseases treatment market size is expected to see strong growth in the next few years. It will grow to $1.91 billion in 2030 at a compound annual growth rate (CAGR) of 5.6%. The growth in the forecast period can be attributed to increasing development of orphan drugs, rising investments in rare disease research, growing adoption of biologic therapies, expansion of precision medicine approaches, increasing patient access to advanced treatments. Major trends in the forecast period include increasing adoption of targeted biologic therapies, rising focus on personalized treatment protocols, growing use of advanced diagnostic devices, expansion of enzyme replacement therapies, enhanced integration of digital patient monitoring.Global Rare Gastrointestinal Diseases Treatment Market Segmentation
1) By Type: Drugs, Device 2) By Route Of Administration: Oral, Intravenous (IV) Or Injectable, Subcutaneous, Other Routes Of Administration 3) By Application: Eosinophilic Gastroenteritis, Hercksprung's Disease, Chronic Intestinal Pseudo-Obstruction 4) By End User: Hospitals, Research Institutes, Other End Users Subsegments: 1) By Drugs: Anti-Inflammatory Drugs, Immunosuppressive Agents, Biologic Therapies, Antibiotics, Proton Pump Inhibitors, Corticosteroids, Enzyme Replacement Therapies 2) By Devices: Endoscopic Devices, Diagnostic Imaging Systems, Surgical Instruments, Feeding Tubes, Stent Devices, Biopsy DevicesWhat Is The Driver Of The Rare Gastrointestinal Diseases Treatment Market?
The rising diagnostic testing is expected to propel the growth of the rare gastrointestinal diseases treatment market going forward. Diagnostic tests refer to medical tests performed to detect, diagnose, or monitor diseases, conditions, or infections in a patient. The increasing demand for diagnostic tests is primarily driven by growing awareness about the importance of early disease detection, which enables timely interventions and better patient outcomes. Diagnostic testing aids in rare gastrointestinal diseases treatment by enabling early and accurate detection of underlying conditions. It helps identify specific disease pathways, supports personalized treatment plans, and improves patient outcomes through timely intervention. For instance, in December 2024, according to the National Health Service, a UK-based government department, the number of diagnostic tests by activity has reached 2,544,809 in 2024, an increase from 2,312,030 in 2023. Therefore, the rising diagnostic testing is driving the growth of the rare gastrointestinal diseases treatment industry.Key Players In The Global Rare Gastrointestinal Diseases Treatment Market
Major companies operating in the rare gastrointestinal diseases treatment market are Johnson & Johnson, Merck & Co. Inc., Pfizer Inc., Bayer AG, Sanofi S.A., Zealand Pharma A/S, Novartis AG, AstraZeneca plc, GlaxoSmithKline plc, Eli Lilly and Company, Takeda Pharmaceutical Company Limited, Teva Pharmaceutical Industries Ltd., Regeneron Pharmaceuticals Inc., Ipsen S.A., PTC Therapeutics Inc., Ironwood Pharmaceuticals Inc., Mirum Pharmaceuticals Inc., Albireo Pharma Inc., Orphalan S.A., Ellodi Pharmaceuticals Inc.Global Rare Gastrointestinal Diseases Treatment Market Trends and Insights
Major companies operating in the rare gastrointestinal diseases treatment market are focusing on developing innovative treatments, such as fully human monoclonal antibodies, to target disease pathways and improve patient outcomes. Fully human monoclonal antibodies are lab-engineered antibodies made entirely from human proteins that specifically target disease-causing molecules, helping to precisely block or modulate them and improve treatment effectiveness while reducing side effects. For instance, in August 2023, Regeneron Pharmaceuticals Inc., a US-based biotechnology company, received U.S. Food and Drug Administration (FDA) approval for Veopoz (pozelimab-bbfg) for CHAPLE disease, a rare and life-threatening hereditary gastrointestinal disorder. Veopoz is a fully human monoclonal antibody designed to target complement factor C5, helping to regulate the overactive complement system that causes protein loss and gastrointestinal damage in patients with CHAPLE. This targeted mechanism of action addresses the underlying cause of the disease, preventing debilitating symptoms and potentially life-threatening complications.What Are Latest Mergers And Acquisitions In The Rare Gastrointestinal Diseases Treatment Market?
In May 2023, Ironwood Pharmaceuticals Inc., a US-based gastrointestinal healthcare company, acquired VectivBio Holding AG for approximately $1 billion. With this acquisition, Ironwood Pharmaceuticals aims to expand its gastrointestinal portfolio by adding VectivBio’s late-stage asset apraglutide for short bowel syndrome with intestinal failure, leveraging its commercial strength and clinical expertise to accelerate growth, deliver innovative therapies, and enhance long-term shareholder value. VectivBio Holding AG is a Switzerland-based clinical-stage biotechnology company that specializes in developing treatments for severe and rare gastrointestinal conditions.Regional Insights
North America was the largest region in the rare gastrointestinal diseases treatment market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Rare Gastrointestinal Diseases Treatment Market?
The rare gastrointestinal diseases treatment market consists of revenues earned by entities by providing services such as diagnosis of rare gastrointestinal disorders, prescription of specialized therapies and medications, management and monitoring of patient treatment plans, and support programs for patients with rare digestive conditions. The market value includes the value of related goods sold by the service provider or included within the service offering. The rare gastrointestinal diseases treatment market also includes sales of specialized drugs, biologics, nutritional therapies, and diagnostic kits. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Rare Gastrointestinal Diseases Treatment Market Report 2026?
The rare gastrointestinal diseases treatment market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the rare gastrointestinal diseases treatment industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Rare Gastrointestinal Diseases Treatment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.53 billion |
| Revenue Forecast In 2035 | $1.91 billion |
| Growth Rate | CAGR of 5.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Route Of Administration, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Johnson & Johnson, Merck & Co. Inc., Pfizer Inc., Bayer AG, Sanofi S.A., Zealand Pharma A/S, Novartis AG, AstraZeneca plc, GlaxoSmithKline plc, Eli Lilly and Company, Takeda Pharmaceutical Company Limited, Teva Pharmaceutical Industries Ltd., Regeneron Pharmaceuticals Inc., Ipsen S.A., PTC Therapeutics Inc., Ironwood Pharmaceuticals Inc., Mirum Pharmaceuticals Inc., Albireo Pharma Inc., Orphalan S.A., Ellodi Pharmaceuticals Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
