
Real World Evidence Solutions Market Report 2026
Global Outlook – By Component (Services, Data Sets, Clinical Setting Data, Claims Data, Pharmacy Data, Patient Powered Data), By Therapeutic Area (Oncology, Cardiovascular, Neurology, Immunology, Other Therapeutic Areas), By Application (Drug Development And Approvals, Medical Device Development And Approvals, Reimbursement/Coverage And Regulatory Decision Making, Post Market Safety And Adverse Events Monitoring), By End-Users (Pharmaceutical And Medical Devices Companies, Healthcare Providers, Healthcare Payers, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Real World Evidence Solutions Market Overview
• Real World Evidence Solutions market size has reached to $2.33 billion in 2025 • Expected to grow to $4.81 billion in 2030 at a compound annual growth rate (CAGR) of 15.6% • Growth Driver: Geriatric Population Spurs Demand For Real-World Evidence Solutions • Market Trend: Real-World Evidence Solutions Market Advances With Maxis Clinical Sciences Rwe Solutions Service • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Real World Evidence Solutions Market?
Real-world evidence (RWE) consists of clinical insights derived from data gathered outside of controlled clinical trials, using sources such as electronic health records, insurance claims, and patient registries. It helps assess the safety, effectiveness, and usage of medical treatments in real-life healthcare environments. The main components of real-world evidence solutions are services, data sets, clinical setting data, claims data, pharmacy data, and patient-powered data. Services refer to an activity or the performance of a task with a commercial purpose. They are used in oncology, cardiovascular disease, neurology, immunology, and other therapeutic areas. They are applied in drug development and approval, medical device development and approval, reimbursement/coverage and regulatory decision making, and post-market safety and adverse event monitoring. The various end-users are pharmaceutical and medical device companies, healthcare providers, healthcare payers, and other end-users.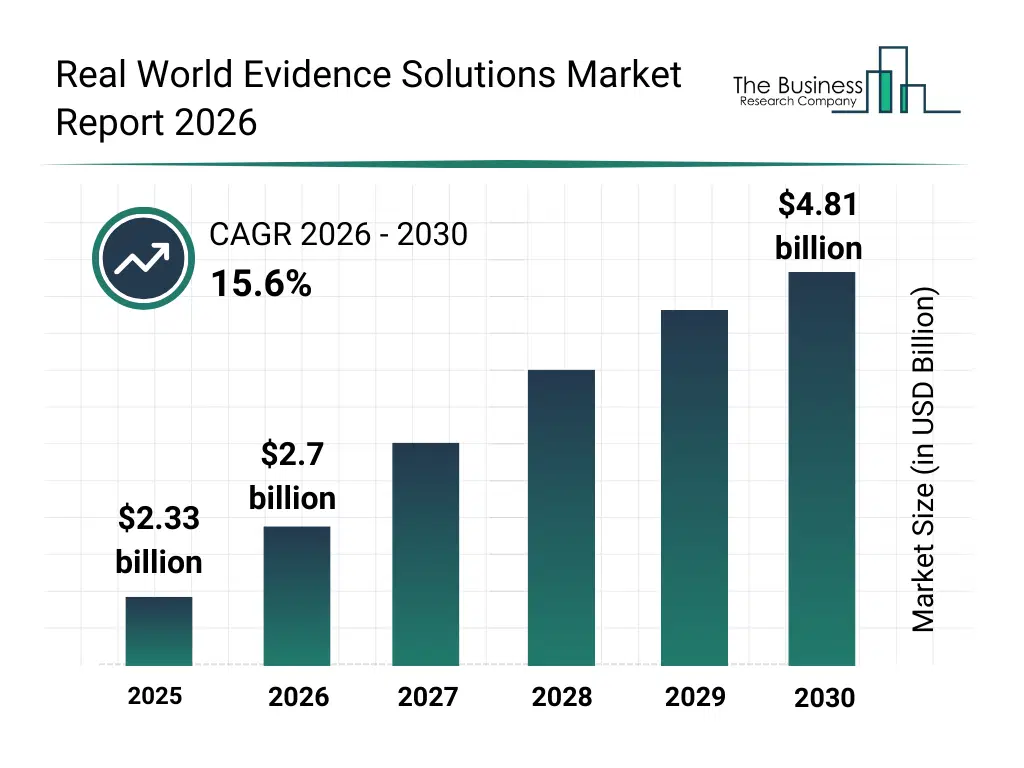
What Is The Real World Evidence Solutions Market Size and Share 2026?
The real world evidence solutions market size has grown rapidly in recent years. It will grow from $2.33 billion in 2025 to $2.7 billion in 2026 at a compound annual growth rate (CAGR) of 15.9%. The growth in the historic period can be attributed to increasing availability of real-world healthcare data, rising demand for post-approval evidence, growing focus on drug safety monitoring, expansion of electronic health record adoption, early regulatory acceptance of rwe.What Is The Real World Evidence Solutions Market Growth Forecast?
The real world evidence solutions market size is expected to see rapid growth in the next few years. It will grow to $4.81 billion in 2030 at a compound annual growth rate (CAGR) of 15.6%. The growth in the forecast period can be attributed to data-driven regulatory decision making, growing use of rwe in reimbursement decisions, expansion of precision medicine initiatives, increasing integration of real-time healthcare data, rising adoption of ai-enabled evidence platforms. Major trends in the forecast period include increasing use of advanced analytics in rwe studies, growing integration of ehr and claims data, rising adoption of AI-driven evidence generation, expansion of rwe in regulatory submissions, enhanced focus on post-market safety monitoring.Global Real World Evidence Solutions Market Segmentation
1) By Component: Services, Data Sets, Clinical Setting Data, Claims Data, Pharmacy Data, Patient Powered Data 2) By Therapeutic Area: Oncology, Cardiovascular, Neurology, Immunology, Other Therapeutic Areas 3) By Application: Drug Development And Approvals, Medical Device Development And Approvals, Reimbursement/Coverage And Regulatory Decision Making, Post Market Safety And Adverse Events Monitoring 4) By End-Users: Pharmaceutical And Medical Devices Companies, Healthcare Providers, Healthcare Payers, Other End-Users Subsegments: 1) By Services: Consulting Services, Data Management Services, Regulatory Services, Analytics Services 2) By Data Sets: Electronic Health Records (EHR), Patient Registries, Clinical Trials Data, Observational Study Data 3) By Clinical Setting Data: Hospital Data, Outpatient Data, Specialty Care Data, Claims Data 4) By Insurance Claims Data: Medicare/Medicaid Data, Commercial Claims Data, Pharmacy Data 5) By Prescription Data: Pharmacy Claims Data, Drug Utilization Data 6) By Patient Powered Data: Patient Surveys, Patient-Reported Outcomes (PRO), Wearable Device DataWhat Is The Driver Of The Real World Evidence Solutions Market?
The growing geriatric population is expected to propel the growth of the real-world evidence solutions market. The geriatric population refers to people aged 65 and over, who are at higher risk of falling for several reasons, including postural hypotension, and balance or gait impairment. Aging increases the threat of chronic diseases such as strokes, cardiovascular diseases, type 2 diabetes, arthritis, and cancer. For instance, in January 2024, according to the data published by the Population Reference Bureau, a US-based nonprofit organization, the population of Americans aged 65 and older is expected to grow from 58 million in 2022 to 82 million by 2050, a 47% increase. Additionally, their share of the total population is projected to rise from 17% to 23%. Therefore, the growing geriatric population is driving the growth of the real-world evidence solutions market.Key Players In The Global Real World Evidence Solutions Market
Major companies operating in the real world evidence solutions market are Anthem Inc., Cegedim Health Data, Clarivate PLC, Cognizant Technology Solutions Corporation, Flatiron Health Inc., International Business Machines Corporation, ICON plc, IQVIA Holdings Inc., Medpace Holdings Inc., Optum Inc., Oracle Corporation, PAREXEL International Corporation, PPD Inc., SAS Institute Inc., Syneos Health, Symphony Innovation LLC, UnitedHealth Group Incorporated, Deloitte Touche Tohmatsu Limited, Informa PLC, Palantir Technologies, UDG Healthcare PLC, Elevance Health Inc., Covance Consulting Ltd., Clinipace Worldwide, Evidera Inc., Pharmerit International, Analysis Group Inc., HealthCore Inc., Inovalon Holdings Inc., KMK Consulting Inc.Global Real World Evidence Solutions Market Trends and Insights
Major companies operating in the real-world evidence solutions market are developing innovative products, such as the new real-world evidence (RWE) solutions service, which is set to optimize evidence-based medicine. Real-world evidence (RWE) solutions services are designed to generate and analyze clinical evidence on the usage, potential benefits, and risks of medical products using real-world data (RWD) resulting from routine healthcare delivery. For instance, in October 2023, Maxis Clinical Sciences, a US-based clinical research consultancy for small to medium-sized pharmaceutical and life sciences companies worldwide, launched new Real-World Evidence (RWE) Solutions service. Maxis Clinical Sciences' RWE Solutions has been established to combine several RWD sources. These include patient-reported outcomes as well as data from wearable devices, which were designed to improve patient participation and data quality.What Are Latest Mergers And Acquisitions In The Real World Evidence Solutions Market?
In March 2024, eMed LLC, a US-based telehealth and diagnostics company, acquired Science 37 Holdings Inc. through a tender offer valued at $5.75 per share. With this acquisition, eMed seeks to enhance its real-world evidence (RWE) solutions by integrating Science 37’s decentralized Metasite™ platform, which enables patients to participate in clinical studies remotely and supports the capture of real-world data outside traditional research sites. Science 37 Holdings Inc. is a US-based decentralized clinical trials company focused on expanding access to studies by connecting participants virtually and generating robust real-world patient data.Regional Outlook
North America was the largest region in the real world evidence solutions market in 2025. Asia-Pacific is expected to be the fastest-growing region in the real world evidence solutions market share during the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Real World Evidence Solutions Market?
The real-world evidence solutions market consists of sales of solutions that are used in randomized trials, large simple trials, pragmatic trials, and observational studies. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Real World Evidence Solutions Market Report 2026?
The real world evidence solutions market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the real world evidence solutions industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Real World Evidence Solutions Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.7 billion |
| Revenue Forecast In 2035 | $4.81 billion |
| Growth Rate | CAGR of 15.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Component, Therapeutic Area, Application, End-Users |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Anthem Inc., Cegedim Health Data, Clarivate PLC, Cognizant Technology Solutions Corporation, Flatiron Health Inc., International Business Machines Corporation, ICON plc, IQVIA Holdings Inc., Medpace Holdings Inc., Optum Inc., Oracle Corporation, PAREXEL International Corporation, PPD Inc., SAS Institute Inc., Syneos Health, Symphony Innovation LLC, UnitedHealth Group Incorporated, Deloitte Touche Tohmatsu Limited, Informa PLC, Palantir Technologies, UDG Healthcare PLC, Elevance Health Inc., Covance Consulting Ltd., Clinipace Worldwide, Evidera Inc., Pharmerit International, Analysis Group Inc., HealthCore Inc., Inovalon Holdings Inc., KMK Consulting Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
