
Remicade Biosimilar Market Report 2026
Global Outlook – By Type (100mg/10ml, 500mg/50ml), By Disease Indication (Ulcerative Colitis, Rheumatoid Arthritis, Ankylosing Spondylitis, Crohn’s Disease, Psoriatic Arthritis, Plaque Psoriasis), By Application (Blood Disorders, Oncology Diseases) – Market Size, Trends, Strategies, and Forecast to 2035
Remicade Biosimilar Market Overview
• Remicade Biosimilar market size has reached to $4.89 billion in 2025 • Expected to grow to $13.31 billion in 2030 at a compound annual growth rate (CAGR) of 22.2% • Growth Driver: Surge In Autoimmune Diseases Fuels Remicade Biosimilar Market Growth • Market Trend: Innovations in the Remicade Biosimilar Market With Subcutaneous Formulations • Europe was the largest region in 2025.What Is Covered Under Remicade Biosimilar Market?
Remicade biosimilar refers to a biological drug like Remicade used to treat certain autoimmune diseases in adults and children. It treats autoimmune inflammatory conditions such as psoriasis, rheumatoid arthritis, Crohn's disease, and ulcerative colitis. The main types of biosimilars in remicade biosimilars are 100 mg/10 ml and 500 mg/50 ml. 100 mg/10 ml refers to the amount of drug in the dosage form or a unit of the dosage form that is equal to 100 mg/10 ml. It includes ulcerative colitis, rheumatoid arthritis, ankylosing spondylitis, Crohn’s disease, psoriatic arthritis, and plaque psoriasis. These are used for several applications, including blood disorders and oncology diseases.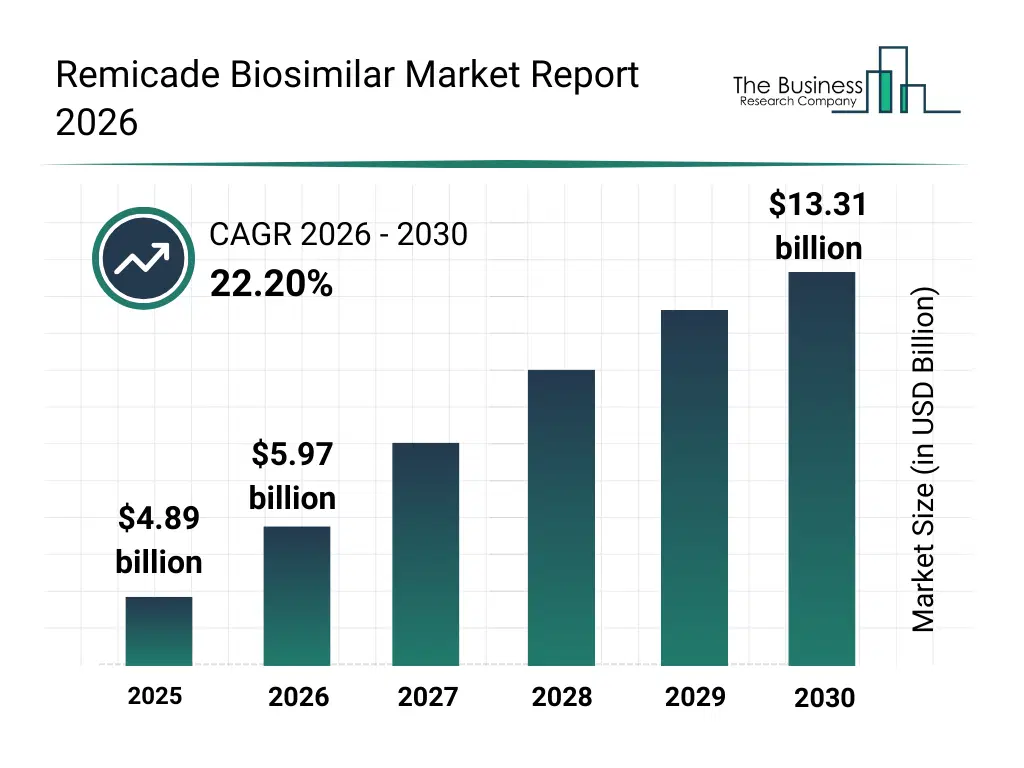
What Is The Remicade Biosimilar Market Size and Share 2026?
The remicade biosimilar market size has grown exponentially in recent years. It will grow from $4.89 billion in 2025 to $5.97 billion in 2026 at a compound annual growth rate (CAGR) of 22.2%. The growth in the historic period can be attributed to high cost burden of branded biologics, patent expiry of remicade, increasing prevalence of autoimmune diseases, government support for biosimilar approvals, hospital-driven bulk procurement models.What Is The Remicade Biosimilar Market Growth Forecast?
The remicade biosimilar market size is expected to see exponential growth in the next few years. It will grow to $13.31 billion in 2030 at a compound annual growth rate (CAGR) of 22.2%. The growth in the forecast period can be attributed to growing demand for affordable biologic therapies, expansion of biosimilar pipelines, rising healthcare expenditure in emerging economies, increasing acceptance among clinicians and patients, favorable reimbursement frameworks. Major trends in the forecast period include increasing adoption of cost-effective biosimilars, rising prescriber confidence in infliximab biosimilars, expansion of indications across autoimmune disorders, competitive pricing and tender-based procurement, growing penetration in emerging healthcare markets.Global Remicade Biosimilar Market Segmentation
1) By Type: 100mg/10ml, 500mg/50ml 2) By Disease Indication: Ulcerative Colitis, Rheumatoid Arthritis, Ankylosing Spondylitis, Crohn’s Disease, Psoriatic Arthritis, Plaque Psoriasis 3) By Application: Blood Disorders, Oncology DiseasesWhat Are The Drivers Of The Remicade Biosimilar Market?
The rise in the incidence of autoimmune diseases is expected to propel the growth of the remicade biosimilar market going forward. Autoimmune diseases are disorders in which the immune system mistakenly attacks and damages the body's tissues and organs. Remicade biosimilars are used in treating autoimmune diseases by providing an effective therapeutic option to manage the symptoms and reduce inflammation associated with these. For instance, in February 2024, according to Arthritis Australia, an Australia-based charitable not-for-profit organisation advocating for people with arthritis and musculoskeletal conditions, by 2025, approximately 562,378 Australians are expected to be living with rheumatoid arthritis (RA), accounting for 14% of the total arthritis population. The number of people with RA is projected to rise by 33%, reaching around 748,721 by 2040. Therefore, the rise in the incidence of autoimmune diseases is driving the growth of the remicade biosimilar industry. The growing healthcare expenditure is significantly contributing to the growth of the remicade biosimilar market going forward. Healthcare expenditure refers to the total amount of money spent on healthcare goods and services within a specific period, typically on an annual basis. Biologic drugs, including Remicade, tend to be expensive. As healthcare expenditure increases, there is a greater focus on cost containment and more efficient allocation of resources. Biosimilars offer a more cost-effective alternative to their originator biologics, including Remicade. For instance, in November 2023, according to the Canadian Institute for Health Information, a Canada-based Institute for Health Information, in 2023, total health spending in Canada is projected to reach $344 billion, equating to $8,740 per person. This marks a 2.8% increase compared to the previous year, which saw only a 1.5% growth in health spending in 2022, indicating a notable rebound in healthcare investment. Therefore, the growing healthcare expenditure will drive the remicade biosimilar industry.Key Players In The Global Remicade Biosimilar Market
Major companies operating in the remicade biosimilar market are Novartis AG, Amgen Inc, Viatris Inc., Teva Pharmaceutical Industries Ltd., Biogen Inc., LG Chem Ltd., Nippon Kayaku Co. Ltd., Celltrion Inc., Nichi-iko Pharmaceutical Co. Ltd., Biocon Limited, Alvotech S.A., MabPharm Ltd., Genor Biopharma Co. Ltd, Shanghai Biomabs Pharmaceuticals Co. Ltd., Samsung Bioepis Co. Ltd., Biocad Biopharmaceutical Co, EPIRUS Biopharmaceuticals Inc., BioXpress Therapeutics SA, Mabion S.AGlobal Remicade Biosimilar Market Trends and Insights
Major companies operating in the remicade biosimilar market are focusing on developing innovative solutions such as subcutaneous formulations to gain a competitive advantage. Subcutaneous formulations of Remicade provide an alternative administration route for the treatment of autoimmune diseases, allowing for self-administration and potentially improving patient adherence to therapy. For instance, in March 2024, Celltrion, a South Korea-based biopharmaceutical company, launched Zymfentra, a biosimilar to Remicade (infliximab) for $6,181.08 for two doses over four weeks, approved by the FDA as the first subcutaneous infliximab formulation for maintaining therapy in adults with moderate-to-severe ulcerative colitis and Crohn's disease. Eligible commercially insured patients can receive copay assistance of $5 per month, while uninsured or underinsured patients may get Zymfentra at no cost.Regional Insights
Europe was the largest region in the remicade biosimilar market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Remicade Biosimilar Market?
The remicade biosimilar market consists of sales of inflectra, renflexis, avolsa, remsima, infimab, and avolsa. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Remicade Biosimilar Market Report 2026?
The remicade biosimilar market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the remicade biosimilar industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Remicade Biosimilar Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $5.97 billion |
| Revenue Forecast In 2035 | $13.31 billion |
| Growth Rate | CAGR of 22.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Disease Indication, Application |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Novartis AG, Amgen Inc, Viatris Inc., Teva Pharmaceutical Industries Ltd., Biogen Inc., LG Chem Ltd., Nippon Kayaku Co. Ltd., Celltrion Inc., Nichi-iko Pharmaceutical Co. Ltd., Biocon Limited, Alvotech S.A., MabPharm Ltd., Genor Biopharma Co. Ltd, Shanghai Biomabs Pharmaceuticals Co. Ltd., Samsung Bioepis Co. Ltd., Biocad Biopharmaceutical Co, EPIRUS Biopharmaceuticals Inc., BioXpress Therapeutics SA, Mabion S.A |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
