
Respiratory Syncytial Virus (RSV) Diagnostics Market Report 2026
Global Outlook – By Product Type (Kits And Assays, Instruments, Reagents, Other Product Types), By Technology (Molecular Diagnostics, Immunoassays, Rapid Antigen Detection Tests, Other Technologies), By Sample Type (Nasal Swabs, Throat Swabs, Bronchoalveolar Lavage), By End User (Hospitals, Diagnostic Laboratories, Home Care Settings, Other End Users) - Market Size, Trends, And Global Forecast 2026-2035
Respiratory Syncytial Virus (RSV) Diagnostics Market Overview
• Respiratory Syncytial Virus (RSV) Diagnostics market size has reached to $1.12 billion in 2025 • Expected to grow to $1.84 billion in 2030 at a compound annual growth rate (CAGR) of 10.4% • Growth Driver: Impact Of Rising Respiratory Disease Prevalence On Diagnostic Demand • Market Trend: Advancements In Multi-Virus Diagnostic Testing For Respiratory Infections • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Respiratory Syncytial Virus (RSV) Diagnostics Market?
Respiratory syncytial virus (RSV) diagnostics refers to the tests and methods used to detect the presence of RSV in patients, typically through respiratory samples like nasal swabs or throat swabs. It is used for identifying RSV infections, which can cause respiratory illness in infants, young children, and the elderly, to guide treatment and management. The main types of respiratory syncytial virus (RSV) diagnostic products are kits and assays, instruments, reagents, and others. Kits and assays are pre-packaged combinations of reagents, materials, and instructions used for specific diagnostic tests. It uses various technologies such as molecular diagnostics, immunoassays, rapid antigen detection tests, and others and uses various sample types, including nasal swabs, throat swabs, and bronchoalveolar lavage. It is used by hospitals, diagnostic laboratories, home care settings, and others.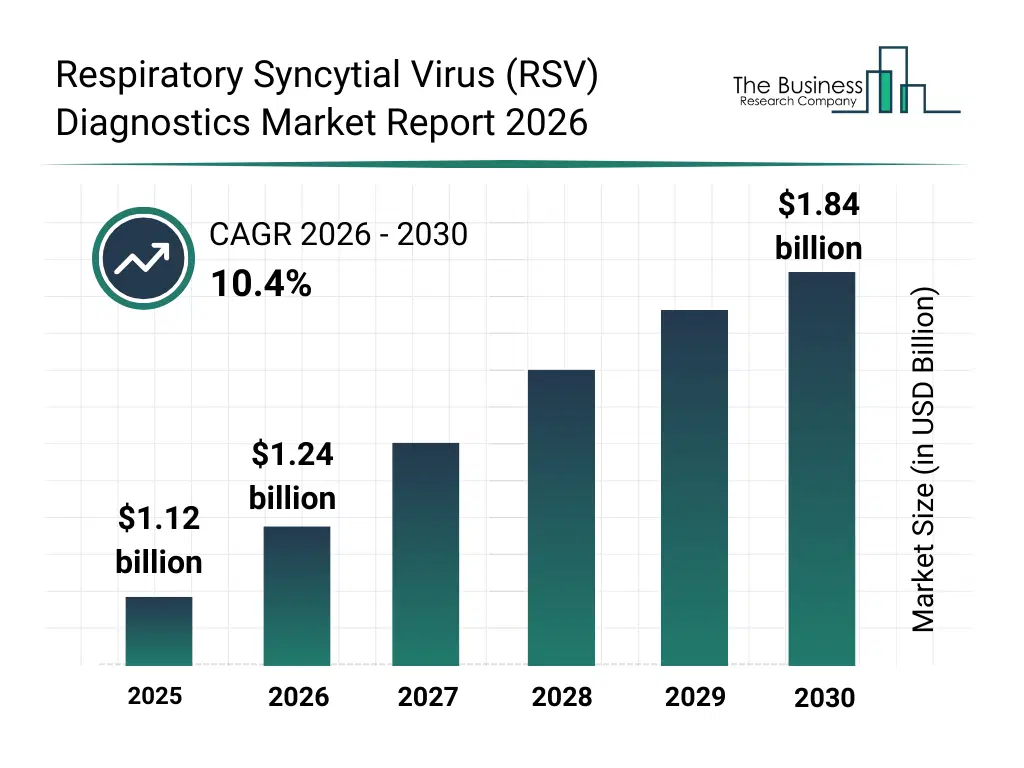
What Is The Respiratory Syncytial Virus (RSV) Diagnostics Market Size and Share 2026?
The respiratory syncytial virus (rsv) diagnostics market size has grown rapidly in recent years. It will grow from $1.12 billion in 2025 to $1.24 billion in 2026 at a compound annual growth rate (CAGR) of 10.7%. The growth in the historic period can be attributed to rising incidence of pediatric respiratory infections, increasing hospitalization rates among elderly populations, wider availability of immunoassay technologies, expansion of diagnostic laboratory networks, increased clinical awareness of rsv.What Is The Respiratory Syncytial Virus (RSV) Diagnostics Market Growth Forecast?
The respiratory syncytial virus (rsv) diagnostics market size is expected to see rapid growth in the next few years. It will grow to $1.84 billion in 2030 at a compound annual growth rate (CAGR) of 10.4%. The growth in the forecast period can be attributed to increasing demand for early and accurate respiratory diagnosis, growing adoption of home testing kits, rising investments in molecular diagnostics, expansion of multiplex testing adoption, increasing focus on pediatric preventive care. Major trends in the forecast period include increasing adoption of rapid point-of-care rsv tests, rising use of molecular diagnostic assays, growing integration of multiplex respiratory panels, expansion of home-based rsv testing solutions, enhanced focus on early infant diagnosis.Global Respiratory Syncytial Virus (RSV) Diagnostics Market Segmentation
1) By Product Type: Kits And Assays, Instruments, Reagents, Other Product Types 2) By Technology: Molecular Diagnostics, Immunoassays, Rapid Antigen Detection Tests, Other Technologies 3) By Sample Type: Nasal Swabs, Throat Swabs, Bronchoalveolar Lavage 4) By End User: Hospitals, Diagnostic Laboratories, Home Care Settings, Other End Users Subsegments: 1) By Kits And Assays: Rapid Antigen Detection Kits, PCR-based Assays, Serological Assays 2) By Instruments: PCR Analyzers, Immunoassay Analyzers 3) By Reagents: PCR Reagents, Antibody Reagents, Enzyme Reagents 4) By Other Product Types: Sample Collection Devices, Transport MediaWhat Is The Driver Of The Respiratory Syncytial Virus (RSV) Diagnostics Market?
The rising prevalence of respiratory diseases is expected to drive the growth of the respiratory syncytial virus (RSV) diagnostics market going forward. Respiratory diseases are a group of disorders affecting the airways and lungs, impairing breathing and oxygen exchange, and include conditions such as asthma, chronic obstructive pulmonary disease (COPD), pneumonia, and respiratory infections. The prevalence of respiratory diseases is rising due to factors such as increasing air pollution, smoking, climate change, urbanization, and the growing prevalence of infections and chronic conditions like asthma and COPD. Respiratory syncytial virus (RSV) diagnostics is essential for respiratory diseases to enable accurate and timely identification of RSV infections, allowing for appropriate treatment, management, and prevention of severe complications, especially in vulnerable populations. For instance, in June 2025, according to the Office for Health Improvement and Disparities (OHID), a UK-based government department, the rate of emergency admissions for respiratory disease in England increased by 7% from the financial year ending 2023 to the financial year ending 2024. Therefore, the rising prevalence of respiratory diseases is fueling the growth of the respiratory syncytial virus (RSV) diagnostics industry.Key Players In The Global Respiratory Syncytial Virus (RSV) Diagnostics Market
Major companies operating in the respiratory syncytial virus (rsv) diagnostics market are F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthineers AG, Merck KGaA, Becton, Dickinson, and Company, Quest Diagnostics Incorporated, Hologic Inc., BIOMÉRIEUX SA, QuidelOrtho Corporation, Bio-Rad Laboratories Inc., DiaSorin S.p.A., Acon Laboratories Inc., Cepheid Inc., Meridian Bioscience Inc., Seegene Inc., LumiraDx Ltd., Sekisui Diagnostics Ltd., Boditech Med Inc., Trinity Biotech plc, R-Biopharm AG, GeneTex Inc., Creative Diagnostics, Certest Biotec S.L., Clarity Diagnostics LLC, Genes 2Me LLP, Coris BioConcept S.A.Global Respiratory Syncytial Virus (RSV) Diagnostics Market Trends and Insights
Major companies operating in the respiratory syncytial virus (RSV) diagnostics market are developing innovative products, such as respiratory virus combination testing, to provide accurate and efficient solutions for customers. Respiratory virus combination testing is a diagnostic method that simultaneously detects multiple respiratory viruses, such as influenza, respiratory syncytial virus (RSV), and others, in a single sample to aid in the identification of viral infections. For instance, in January 2023, Sonora Quest Laboratories, a US-based clinical laboratory testing company, launched the COVID-19, RSC, and influenza diagnostic test. This molecular diagnostic test provides a qualitative, multi-target analysis using a single patient sample to detect COVID-19, respiratory syncytial virus (RSV), and influenza. By combining these three virus tests into one, it streamlines the testing process and reduces the need for multiple samples. The ability to quickly identify all three infections helps healthcare providers make faster treatment decisions, potentially improving patient outcomes. Additionally, this test can contribute to slowing the spread of these viruses, reducing the risk of transmission to others.What Are Latest Mergers And Acquisitions In The Respiratory Syncytial Virus (RSV) Diagnostics Market?
In November 2023, Medix Biochemica, a Finland-based biotechnology company, acquired ViroStat LLC for an undisclosed amount. With this acquisition, Medix aims to expand its infectious disease diagnostic portfolio by incorporating ViroStat's expertise in providing diagnostic solutions, including those for respiratory syncytial virus (RSV). ViroStat LLC is a US-based company that offers a range of diagnostic products, including monoclonal antibodies and antigen detection kits for RSV testing.Regional Outlook
North America was the largest region in the respiratory syncytial virus (RSV) diagnostics market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Respiratory Syncytial Virus (RSV) Diagnostics Market?
The respiratory syncytial virus (RSV) diagnostics market consists of revenues earned by entities by providing services such as diagnostic consultation, research and development support, training and technical support. The market value includes the value of related goods sold by the service provider or included within the service offering. The respiratory syncytial virus (RSV) diagnostics market also includes sales of immunofluorescence test kits, multiplex panels, sample collection devices and automated testing instruments. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Respiratory Syncytial Virus (RSV) Diagnostics Market Report 2026?
The respiratory syncytial virus (rsv) diagnostics market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the respiratory syncytial virus (rsv) diagnostics industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Respiratory Syncytial Virus (RSV) Diagnostics Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.24 billion |
| Revenue Forecast In 2035 | $1.84 billion |
| Growth Rate | CAGR of 10.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Technology, Sample Type, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthineers AG, Merck KGaA, Becton, Dickinson, and Company, Quest Diagnostics Incorporated, Hologic Inc., BIOMÉRIEUX SA, QuidelOrtho Corporation, Bio-Rad Laboratories Inc., DiaSorin S.p.A., Acon Laboratories Inc., Cepheid Inc., Meridian Bioscience Inc., Seegene Inc., LumiraDx Ltd., Sekisui Diagnostics Ltd., Boditech Med Inc., Trinity Biotech plc, R-Biopharm AG, GeneTex Inc., Creative Diagnostics, Certest Biotec S.L., Clarity Diagnostics LLC, Genes 2Me LLP, Coris BioConcept S.A. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
