
Respiratory Syncytial Virus (RSV) Therapeutics Market Report 2026
Global Outlook – By Drug (Palivizumab, Ribavirin, Other Drugs), By Treatment (Immune Prophylaxis, Supportive Care, Antiviral Medications), By Dosage Form (Oral, Injectable, Other Dosage Forms), By Patient Type (Pediatrics, Adults), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies) - Market Size, Trends, And Global Forecast 2026-2035
Respiratory Syncytial Virus (RSV) Therapeutics Market Overview
• Respiratory Syncytial Virus (RSV) Therapeutics market size has reached to $2 billion in 2025 • Expected to grow to $5.63 billion in 2030 at a compound annual growth rate (CAGR) of 23% • Growth Driver: The Rise In The Prevalence Of RSV Infection • Market Trend: Pfizer's Abrysvo Approval Marks A Significant Advancement In Respiratory Syncytial Virus (RSV) Therapeutics • North America was the largest region in 2025.What Is Covered Under Respiratory Syncytial Virus (RSV) Therapeutics Market?
Respiratory syncytial virus (RSV) therapeutics refers to the treatments and medications employed to manage and treat infections caused by the respiratory syncytial virus. Respiratory syncytial virus is a common respiratory virus that can cause mild to severe respiratory illness, particularly in infants, young children, older adults, and individuals with weakened immune systems. The main types of drugs in respiratory syncytial virus (RSV) therapeutics are palivizumab, ribavirin, and others. Palivizumab is a monoclonal antibody developed through the application of DNA recombination for respiratory infections and used in children with infections to prevent major complications resulting from respiratory syncytial virus infection. Treatments are categorized into immune prophylaxis, supportive care, and antiviral medications, which are administered in dosage forms such as oral, injectable, and others for patients such as pediatrics and adults. These therapeutics are distributed through hospital pharmacies, retail pharmacies, and online pharmacies.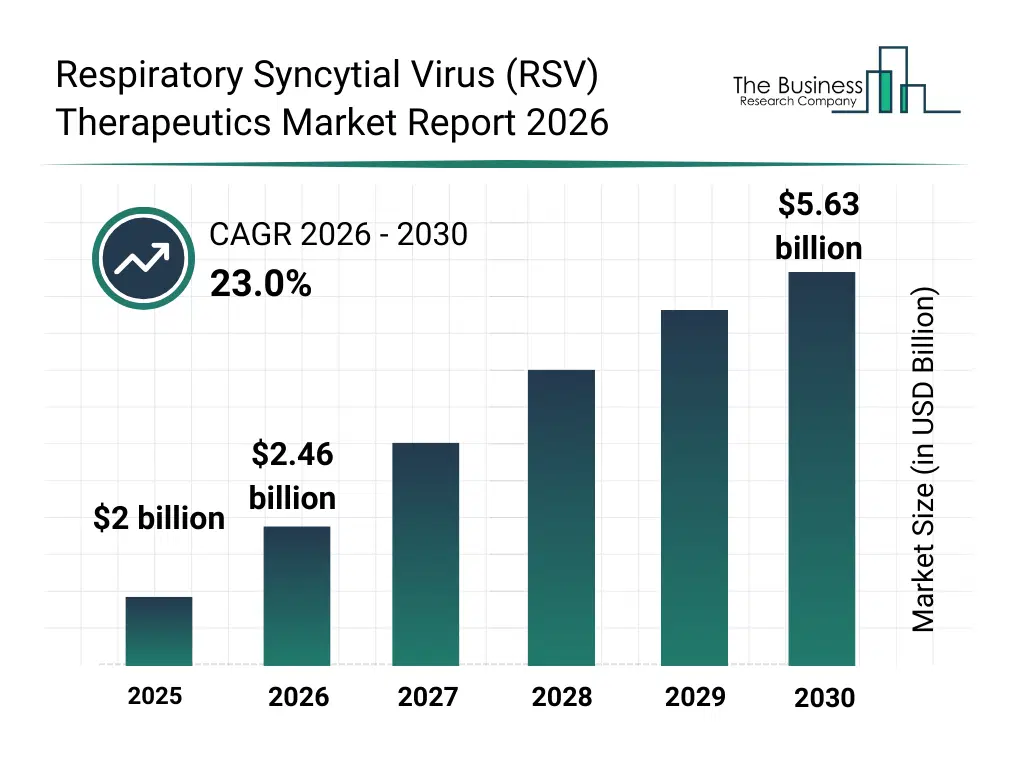
What Is The Respiratory Syncytial Virus (RSV) Therapeutics Market Size and Share 2026?
The respiratory syncytial virus (rsv) therapeutics market size has grown exponentially in recent years. It will grow from $2 billion in 2025 to $2.46 billion in 2026 at a compound annual growth rate (CAGR) of 23.2%. The growth in the historic period can be attributed to increasing rsv-related hospitalization rates, reliance on supportive care treatments, early adoption of palivizumab therapy, growing pediatric healthcare expenditure, expanding hospital-based treatment access.What Is The Respiratory Syncytial Virus (RSV) Therapeutics Market Growth Forecast?
The respiratory syncytial virus (rsv) therapeutics market size is expected to see exponential growth in the next few years. It will grow to $5.63 billion in 2030 at a compound annual growth rate (CAGR) of 23.0%. The growth in the forecast period can be attributed to increasing investments in rsv vaccine and drug development, rising focus on long-acting antibodies, growing demand for pediatric antiviral therapies, expansion of outpatient treatment options, increasing regulatory approvals for novel rsv drugs. Major trends in the forecast period include increasing development of monoclonal antibody therapies, rising focus on antiviral drug innovation, growing adoption of immune prophylaxis treatments, expansion of pediatric-specific rsv therapies, enhanced focus on long-acting preventive drugs.Global Respiratory Syncytial Virus (RSV) Therapeutics Market Segmentation
1) By Drug: Palivizumab, Ribavirin, Other Drugs 2) By Treatment: Immune Prophylaxis, Supportive Care, Antiviral Medications 3) By Dosage Form: Oral, Injectable, Other Dosage Forms 4) By Patient Type: Pediatrics, Adults 5) By Distribution Channel: Hospital Pharmacies, Retail Pharmacies, Online Pharmacies Subsegments: 1) By Palivizumab: Monoclonal Antibody, Long-Acting Monoclonal Antibody 2) By Ribavirin: Antiviral Medication, Inhalation Antiviral Formulation 3) By Other Drugs: Cidofovir, Nitazoxanide, Other Experimental Or Off-Label TreatmentsWhat Is The Driver Of The Respiratory Syncytial Virus (RSV) Therapeutics Market?
The rise in the prevalence of RSV infection is driving the growth of the respiratory syncytial virus (RSV) therapeutics market going forward. RSV is a prevalent respiratory illness that can result in mild to severe respiratory infections, especially in newborns, young children, elderly adults, and people with compromised immune systems. Increasing RSV infections prompt the development of innovative treatments to address the rising need and mitigate the severity of RSV-related complications. For instance, in September 2023, according to Centres for Disease Control and Prevention, a US-based national public health agency, significant increase in RSV-associated hospitalization rates among children under 4 years, escalating from 2.0 hospitalizations per 100,000 population for the week ending August 5, 2023, to 7.0 hospitalizations per 100,000 population by August 19, 2023. Therefore, the rise in the prevalence of RSV infection propels the growth of the respiratory syncytial virus (RSV) therapeutics industry.Key Players In The Global Respiratory Syncytial Virus (RSV) Therapeutics Market
Major companies operating in the respiratory syncytial virus (rsv) therapeutics market are F Hoffmann La Roche Ltd, Merck And Co Inc, Novartis AG, Sanofi S.A., AstraZeneca PLC, GlaxoSmithKline plc, Gilead Sciences Inc, Moderna Inc, Regeneron Pharmaceuticals Inc, Novavax AB, Vir Biotechnology Inc, Enanta Pharmaceuticals Inc, Kyorin Pharmaceutical Co Ltd, Bavarian Nordic A S, Lupin Limited, Hetero Healthcare Ltd, Inovio Pharmaceuticals Inc, Ascletis Pharma Inc, NanoBio Corporation, Vaxart Inc, Ark Biosciences Private Limited, Pulmocide Ltd, Pfizer Inc, Takeda Pharmaceutical Company LimitedGlobal Respiratory Syncytial Virus (RSV) Therapeutics Market Trends and Insights
Major companies operating in the respiratory syncytial virus (RSV) therapeutics market are focused on developing innovative products such as next-generation prefusion F-protein vaccines to strengthen immune protection and reduce severe RSV-related illness. The innovative name refers to advanced RSV vaccines engineered using stabilized prefusion F-protein antigens, which are designed to trigger a more potent and targeted immune response than traditional formulations. For instance, in May 2023, Pfizer Inc., a US-based pharmaceutical manufacturing company, received approval from the Food and Drug Administration, a US-based federal agency responsible for protecting and promoting public health, for ABRYSVO. The bivalent RSV prefusion F (RSVpreF) vaccine ABRYSVO (Respiratory Syncytial Virus Vaccine) is administered to prevent lower respiratory tract sickness caused by RSV in people 60 and older. ABRYSVO is an unadjuvanted vaccine made up of a pair of preF proteins tailored to maximize immunity to RSV A and B varieties, and it has been clinically proven to be safe and productive.What Are Latest Mergers And Acquisitions In The Respiratory Syncytial Virus (RSV) Therapeutics Market?
In June 2023, Meissa Vaccines, a US-based clinical-stage biotechnology company, entered into a CGMP manufacturing partnership with Exothera S.A. to support the production of its pediatric RSV vaccine candidate for Phase 2 clinical trials. Through this partnership, Meissa aims to secure reliable, scalable, and regulatory-compliant manufacturing capacity to advance its MV-012-968 RSV program toward later-stage development. Exothera S.A. is a Belgium-based provider of process development, viral vector manufacturing, and large-scale CGMP production services for vaccine and cell/gene therapy developers.Regional Outlook
North America was the largest region in the respiratory syncytial virus (RSV) therapeutics market in 2025. Asia Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Respiratory Syncytial Virus (RSV) Therapeutics Market?
The respiratory syncytial virus (RSV) therapeutics market includes revenues earned by entities by providing services such as home care and respiratory support services. The market value includes the value of related goods sold by the service provider or included within the service offering. The respiratory syncytial virus (RSV) therapeutics market consists of sales of antiviral drugs, monoclonal antibodies, and vaccines. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Respiratory Syncytial Virus (RSV) Therapeutics Market Report 2026?
The respiratory syncytial virus (rsv) therapeutics market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the respiratory syncytial virus (rsv) therapeutics industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Respiratory Syncytial Virus (RSV) Therapeutics Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.46 billion |
| Revenue Forecast In 2035 | $5.63 billion |
| Growth Rate | CAGR of 23.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Drug, Treatment, Dosage Form, Patient Type, Distribution Channel |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F Hoffmann La Roche Ltd, Merck And Co Inc, Novartis AG, Sanofi S.A., AstraZeneca PLC, GlaxoSmithKline plc, Gilead Sciences Inc, Moderna Inc, Regeneron Pharmaceuticals Inc, Novavax AB, Vir Biotechnology Inc, Enanta Pharmaceuticals Inc, Kyorin Pharmaceutical Co Ltd, Bavarian Nordic A S, Lupin Limited, Hetero Healthcare Ltd, Inovio Pharmaceuticals Inc, Ascletis Pharma Inc, NanoBio Corporation, Vaxart Inc, Ark Biosciences Private Limited, Pulmocide Ltd, Pfizer Inc, Takeda Pharmaceutical Company Limited |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
