
Safety Syringe Market Report 2026
Global Outlook – By Product (Retractable Safety Syringes, Non Retractable Safety Syringes), By Therapy (Insulin, Glucagon-like peptide-I (CLP-I), Tuberculosis, Growth Hormones), By Application (Subcutaneous, Intramuscular), By End-user (Hospitals, Ambulatory Surgical Centers, Long-term Care Facilities, Specialty Clinics, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Safety Syringe Market Overview
• Safety Syringe market size has reached to $9.68 billion in 2025 • Expected to grow to $14.77 billion in 2030 at a compound annual growth rate (CAGR) of 8.8% • Growth Driver: Surge In Blood-Borne Diseases Accelerates Demand For Safety Syringes In Healthcare • Market Trend: Advancing Needle-Stick Injury Prevention Through Affordable Single-Use Safety Syringe Innovations • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Safety Syringe Market?
A safety syringe is a medical injection device designed with built-in safety features to prevent accidental needle stick injuries or reuse. These syringes are specifically engineered to prevent needle stick injuries, which can occur when healthcare workers or individuals handling syringes accidentally puncture their skin with a used needle. The main products of safety syringes are retractable safety syringes and nonretractable safety syringes. Retractable safety syringes are a type of medical syringe designed with a built-in mechanism that allows the needle to retract or withdraw into the syringe barrel after the injection is administered. It is used for therapies such as insulin, glucagon-like peptide-i (clp-i), tuberculosis, and growth hormones, and are applied for subcutaneous, and intramuscular. It is utilized in hospitals, ambulatory surgical centers, long-term care facilities, specialty clinics, and others.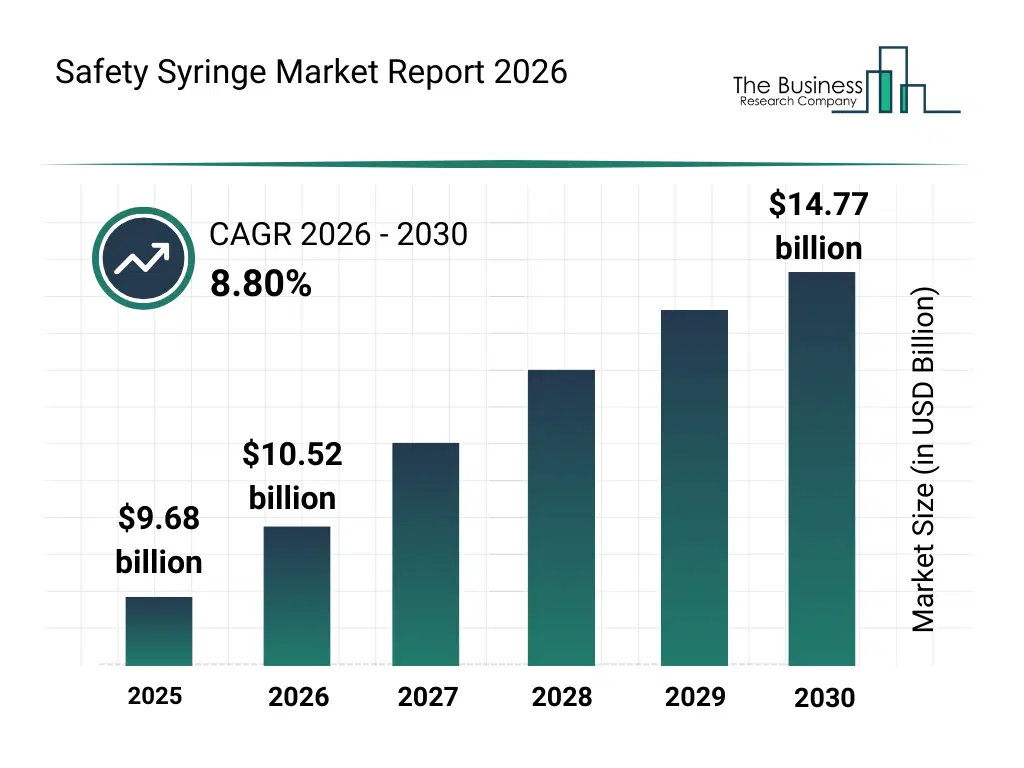
What Is The Safety Syringe Market Size and Share 2026?
The safety syringe market size has grown strongly in recent years. It will grow from $9.68 billion in 2025 to $10.52 billion in 2026 at a compound annual growth rate (CAGR) of 8.7%. The growth in the historic period can be attributed to rising incidence of needle stick injuries among healthcare workers, increasing regulatory emphasis on safe injection and single-use devices, early availability of fixed needle and shielded needle safety syringes, expansion of hospital immunization and therapeutic injection programs, growing awareness of infection prevention and occupational safety.What Is The Safety Syringe Market Growth Forecast?
The safety syringe market size is expected to see strong growth in the next few years. It will grow to $14.77 billion in 2030 at a compound annual growth rate (CAGR) of 8.8%. The growth in the forecast period can be attributed to rising demand for automatic and manual retractable safety syringes, increasing utilization in insulin clp-i tuberculosis and growth hormone therapies, growing adoption across ambulatory surgical centers and specialty clinics, expanding procurement in long-term care and chronic disease management settings, rising focus on preventing device reuse and cross-contamination. Major trends in the forecast period include rising adoption of safety syringes to minimize needle stick injury risks in healthcare settings, increasing utilization of retractable and non-retractable safety syringes across therapeutic injections, growing preference for automatic and manual retractable safety syringe designs, expansion of safety syringe use in subcutaneous and intramuscular drug administration, increasing deployment of safety syringes across hospitals ascs and long-term care facilities.Global Safety Syringe Market Segmentation
1) By Product: Retractable Safety Syringes, Non Retractable Safety Syringes 2) By Therapy: Insulin, Glucagon-like peptide-I (CLP-I), Tuberculosis, Growth Hormones 3) By Application: Subcutaneous, Intramuscular 4) By End-user: Hospitals, Ambulatory Surgical Centers, Long-term Care Facilities, Specialty Clinics, Other End Users Subsegments: 1) By Retractable Safety Syringes: Automatic Retractable Safety Syringes, Manual Retractable Safety Syringes 2) By Non-Retractable Safety Syringes: Fixed Needle Safety Syringes, Shielded Needle Safety SyringesWhat Is The Driver Of The Safety Syringe Market?
The rising number of blood-borne diseases is expected to propel the growth of the safety syringe market going forward. A blood-borne disease is an infectious disease caused by microorganisms, such as bacteria, viruses, or parasites, that are transmitted through the blood. The use of safety syringes is a proactive measure to protect healthcare workers and patients from the risks associated with blood-borne diseases by reducing the likelihood of accidental exposures and promoting best practices in medical procedures. For instance, in November 2024, according to the UK Health Security Agency, a UK-based government agency, over the first 24 months, 34 emergency departments carried out 1,981,590 HIV tests, 1,502,799 hepatitis C tests, and 1,185,678 hepatitis B tests, leading to a substantial rise in the yearly number of bloodborne virus tests in England. Therefore, the rising number of blood-borne diseases is driving the growth of the safety syringe industry.Key Players In The Global Safety Syringe Market
Major companies operating in the safety syringe market are Cardinal Health Inc., Retractable Technologies Inc., Novo Nordisk A/s, Becton Dickinson and Company, Baxter International Inc., Medline Industries LP, Terumo Corporation, B. Braun Melsungen AG, West Pharmaceutical Services Inc., Fresenius Kabi AG, Nemera, Smiths Medical International Ltd., Nipro Medical Corporation, Henke-Sass Wolf GmbH, Revolutions Medical Inc., Sol-Millennium Inc., UltiMed Inc., Unilife Corporation, AdvaCare Pharma, Axol Bio Corporation, Vita Needle Company, Numedico Technologies Pty Ltd., Duopross Meditech Corporation, DMC Medical Ltd., Guangdong Haiou Medical Apparatus Co. Ltd., Kendall Healthcare Group Ltd.Global Safety Syringe Market Trends and Insights
Major companies operating in the safety syringe market are developing new products, such as glass vaccine syringe, to gain a competitive edge in the market. A glass vaccine syringe is a medical device made of glass used for injecting vaccines into patients, offering a sterile and reusable option for administering medications. For instance, on March 2024, Hindustan Syringes and Medical Devices, an India-based medical device manufacturer, launched the Dispojekt single-use safety syringe with an integrated safety shield called SIP Shield. The Dispojekt syringe is designed to reduce needle-stick injuries, cut infection-control and disposal costs, and improve workplace safety for healthcare workers; HMD invested about Rs 70 crore in the first production phase and plans to scale to 300 million units per year. Dispojekt is pitched as an affordable, domestically manufactured safety syringe aimed at the developing world and is claimed to be roughly half the cost of comparable safety syringes used in the USA and Europe, supporting wider adoption and stronger infection-prevention outcomes.What Are Latest Mergers And Acquisitions In The Safety Syringe Market?
In December 2024, Novo Holdings A/S, a Denmark-based life sciences investment company, acquired Catalent, Inc. for approximately US$16.5 billion. The acquisition aims to bolster Novo Holdings’ scale in sterile fill-and-finish, prefilled syringe production and injectable supply-chain capabilities, thereby enabling its portfolio companies to meet rising global demand for injectable biologics and drug-device combinations. Catalent, Inc. is a US-based CDMO specializing in drug-delivery technologies, sterile manufacturing, prefilled syringes and biologics supply for leading pharmaceutical and biotech firms.Regional Outlook
North America was the largest region in the safety syringes market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Safety Syringe Market?
The safety syringe market consists of sales of retractable syringes, needleless syringes, safety syringe accessories, needle removers and blood collection devices. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Safety Syringe Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $10.52 billion |
| Revenue Forecast In 2035 | $14.77 billion |
| Growth Rate | CAGR of 8.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Therapy, Application, End-user |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Cardinal Health Inc., Retractable Technologies Inc., Novo Nordisk A/s, Becton Dickinson and Company, Baxter International Inc., Medline Industries LP, Terumo Corporation, B. Braun Melsungen AG, West Pharmaceutical Services Inc., Fresenius Kabi AG, Nemera, Smiths Medical International Ltd., Nipro Medical Corporation, Henke-Sass Wolf GmbH, Revolutions Medical Inc., Sol-Millennium Inc., UltiMed Inc., Unilife Corporation, AdvaCare Pharma, Axol Bio Corporation, Vita Needle Company, Numedico Technologies Pty Ltd., Duopross Meditech Corporation, DMC Medical Ltd., Guangdong Haiou Medical Apparatus Co. Ltd., Kendall Healthcare Group Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
