
Scleroderma Diagnostics And Therapeutics Market Report 2026
Global Outlook – By Drug Type (Corticosteroids, Immunosuppressive Agents, Endothelin Receptor Antagonists, Calcium Channel Blockers, Phosphodiesterase Type 5 (PDE-5) Inhibitors, Chelating Agents, Prostacyclin Analogues, Other Drug Types), By Diagnostic Test Type (Blood Tests, Imaging Techniques, Skin Biopsy, Pulmonary Function Tests, Electrocardiogram And Echocardiogram), By Indication (Systemic Scleroderma, Localized Scleroderma), By Route Of Administration (Oral, Injectable, Topical), By End-User (Hospitals, Diagnostic Centers, Private Laboratories, Government Laboratories, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Scleroderma Diagnostics And Therapeutics Market Overview
• Scleroderma Diagnostics And Therapeutics market size has reached to $2.94 billion in 2025 • Expected to grow to $4.39 billion in 2030 at a compound annual growth rate (CAGR) of 8.3% • Growth Driver: Rising Prevalence Of Autoimmune Diseases Fueling The Growth Of The Market Due To Genetic Predisposition And Increasing Disease Burden • Market Trend: Advancements In CD19 CAR T-Cell Therapy Drive Innovation In Scleroderma Treatment • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Scleroderma Diagnostics And Therapeutics Market?
Scleroderma diagnostics and therapeutics involve advanced testing methods and targeted treatments designed to detect and manage scleroderma, a chronic autoimmune disorder characterized by the hardening and tightening of the skin and connective tissues. These methods help detect symptoms such as Raynaud’s phenomenon, skin thickening, and internal organ involvement. The main drug types of scleroderma diagnostics and therapeutics are corticosteroids, immunosuppressive agents, endothelin receptor antagonists, calcium channel blockers, phosphodiesterase type 5 (PDE-5) inhibitors, chelating agents, prostacyclin analogues, and others. Corticosteroids are a class of steroid hormones that help reduce inflammation and suppress the immune system. The various types of diagnostic tests include blood tests, imaging techniques, skin biopsy, pulmonary function tests, electrocardiograms, and echocardiograms, and they are classified into systemic scleroderma and localized scleroderma based on their indications. These can be administered through different routes, including oral, injectable, and topical, serving a range of end-users such as hospitals, diagnostic centers, private laboratories, government laboratories, and others.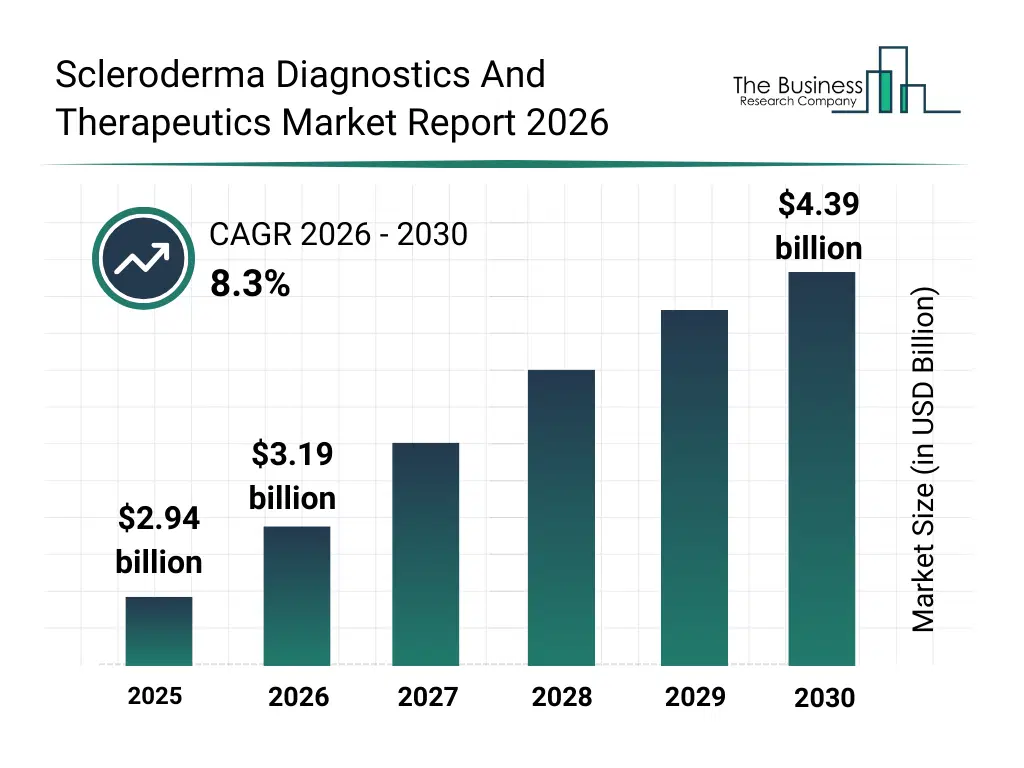
What Is The Scleroderma Diagnostics And Therapeutics Market Size and Share 2026?
The scleroderma diagnostics and therapeutics market size has grown strongly in recent years. It will grow from $2.94 billion in 2025 to $3.19 billion in 2026 at a compound annual growth rate (CAGR) of 8.6%. The growth in the historic period can be attributed to limited awareness of scleroderma, lack of targeted therapies, conventional diagnostic methods, low early diagnosis rates, regional healthcare disparities.What Is The Scleroderma Diagnostics And Therapeutics Market Growth Forecast?
The scleroderma diagnostics and therapeutics market size is expected to see strong growth in the next few years. It will grow to $4.39 billion in 2030 at a compound annual growth rate (CAGR) of 8.3%. The growth in the forecast period can be attributed to development of targeted biologics, ai-powered diagnostics, expansion of rare disease centers, telemedicine adoption, growing patient advocacy and awareness. Major trends in the forecast period include early diagnosis adoption, novel biologic therapies, minimally invasive treatment preference, integration of ai in diagnostics, telehealth for rare diseases.Global Scleroderma Diagnostics And Therapeutics Market Segmentation
1) By Drug Type: Corticosteroids, Immunosuppressive Agents, Endothelin Receptor Antagonists, Calcium Channel Blockers, Phosphodiesterase Type 5 (PDE-5) Inhibitors, Chelating Agents, Prostacyclin Analogues, Other Drug Types 2) By Diagnostic Test Type: Blood Tests, Imaging Techniques, Skin Biopsy, Pulmonary Function Tests, Electrocardiogram And Echocardiogram 3) By Indication: Systemic Scleroderma, Localized Scleroderma 4) By Route Of Administration: Oral, Injectable, Topical 5) By End-User: Hospitals, Diagnostic Centers, Private Laboratories, Government Laboratories, Other End-Users Subsegments: 1) By Corticosteroids: Prednisone, Methylprednisolone, Dexamethasone 2) By Immunosuppressive Agents: Methotrexate, Mycophenolate Mofetil, Azathioprine, Cyclophosphamide 3) By Endothelin Receptor Antagonists: Bosentan, Ambrisentan, Macitentan 4) By Calcium Channel Blockers: Nifedipine, Amlodipine, Diltiazem 5) By Phosphodiesterase Type 5 Inhibitors (Pde-5) Inhibitors: Sildenafil, Tadalafil, Vardenafil 6) By Chelating Agents: D-Penicillamine, Deferoxamine 7) By Prostacyclin Analogues: Epoprostenol, Iloprost, Treprostinil 8) By Other Drug Types: Rituximab (Monoclonal Antibody), Abatacept (T-Cell Modulator), Tocilizumab (Il-6 Inhibitor)What Is The Driver Of The Scleroderma Diagnostics And Therapeutics Market?
The rising prevalence of autoimmune diseases is expected to propel the growth of the scleroderma diagnostics and therapeutics market going forward. Autoimmune diseases are conditions where the immune system mistakenly attacks the body's own healthy cells. The prevalence of autoimmune diseases is increasing due to genetic predisposition, as specific gene variations influence immune system activity and connective tissue abnormalities. Scleroderma diagnostics and therapeutics improve autoimmune disease management by enabling early detection and personalized treatment strategies that streamline clinical decision-making, ultimately improving patient outcomes and overall quality of life. For instance, in June 2024, according to the Australian Institute of Health and Welfare, an Australia-based government agency, 514,000 (2.0%) people were living with rheumatoid arthritis in 2022, contributing to 2.0% of the total disease burden and 16% of the burden of the musculoskeletal condition in 2023, with 1,322 deaths (5.1 deaths per 100,000 population) recorded in 2022, accounting for 0.7% of all deaths. Therefore, the rising prevalence of autoimmune diseases is driving the growth of the scleroderma diagnostics and therapeutics industry.Key Players In The Global Scleroderma Diagnostics And Therapeutics Market
Major companies operating in the scleroderma diagnostics and therapeutics market are Johnson and Johnson, F. Hoffmann-La Roche Ltd., Merck And Co. Inc., Bayer AG, Sanofi S.A., GSK plc, Boehringer Ingelheim International GmbH, Galapagos NV, Inventiva S.A., Redx Pharma plc, Kadmon Holdings Inc., Certa Therapeutics Pty Ltd, Corbus Pharmaceuticals Holdings Inc., aTyr Pharma Inc., MediciNova Inc., Pfizer Inc., Novartis AG, Abbvie Inc., Bristol Myers Squibb Company, AstraZeneca plc, Takeda Pharmaceutical Company Limited, Eli Lilly And Company, Amgen Inc., UCB Pharma, CSL Behring, Mitsubishi Tanabe Pharma, Chugai Pharmaceutical Co. Ltd., Kyowa KirinGlobal Scleroderma Diagnostics And Therapeutics Market Trends and Insights
Major companies operating in the scleroderma diagnostics and therapeutics market are focusing on developing advancements in diagnostic techniques, such as fully human CD19 CAR T-cell therapy, to improve disease management and patient outcomes. Human CD19 CAR T-cell therapy refers to a type of immunotherapy in which a patient's own T cells (a type of immune cell) are genetically modified to express a chimeric antigen receptor (CAR) that specifically targets CD19, a protein found on the surface of B cells. For instance, in October 2023, Kyverna Therapeutics Inc., a US-based biopharmaceutical company, received FDA clearance for its Investigational New Drug (IND) application for KYV-101, a fully human CD19 CAR T-cell therapy, to initiate a Phase 1/2 clinical trial for the treatment of diffuse cutaneous systemic sclerosis (scleroderma). The upcoming KYSA-5 trial will assess the safety and efficacy of KYV-101, which specifically targets B cells involved in the progression of autoimmune diseases. This therapy is designed to reset the immune system and enhance outcomes for patients with scleroderma.What Are Latest Mergers And Acquisitions In The Scleroderma Diagnostics And Therapeutics Market?
In October 2023, Amgen Inc., a US-based biotechnology company, acquired Horizon Therapeutics plc for $27.8 billion. Through this acquisition, Amgen aims to strengthen its portfolio in rare autoimmune and inflammatory diseases by leveraging Horizon Therapeutics' expertise and innovative drug pipeline. Horizon Therapeutics plc is an Ireland-based biotechnology company focusing on developing treatments for scleroderma, particularly through innovative therapies targeting fibrosis and immune system regulation.Regional Outlook
North America was the largest region in the scleroderma diagnostics and therapeutics market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Scleroderma Diagnostics And Therapeutics Market?
The scleroderma diagnostics and therapeutics market includes revenues earned by entities by providing services such as advanced diagnostic imaging and laboratory testing, biomarker and genetic testing, targeted therapeutic development, personalized treatment planning, and remote patient monitoring. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Scleroderma Diagnostics And Therapeutics Market Report 2026?
The scleroderma diagnostics and therapeutics market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the scleroderma diagnostics and therapeutics industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Scleroderma Diagnostics And Therapeutics Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.19 billion |
| Revenue Forecast In 2035 | $4.39 billion |
| Growth Rate | CAGR of 8.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Drug Type, Diagnostic Test Type, Indication, Route Of Administration, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Johnson and Johnson, F. Hoffmann-La Roche Ltd., Merck And Co. Inc., Bayer AG, Sanofi S.A., GSK plc, Boehringer Ingelheim International GmbH, Galapagos NV, Inventiva S.A., Redx Pharma plc, Kadmon Holdings Inc., Certa Therapeutics Pty Ltd, Corbus Pharmaceuticals Holdings Inc., aTyr Pharma Inc., MediciNova Inc., Pfizer Inc., Novartis AG, Abbvie Inc., Bristol Myers Squibb Company, AstraZeneca plc, Takeda Pharmaceutical Company Limited, Eli Lilly And Company, Amgen Inc., UCB Pharma, CSL Behring, Mitsubishi Tanabe Pharma, Chugai Pharmaceutical Co. Ltd., Kyowa Kirin |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
