
Sexually Transmitted Infection (STI) And Vaginitis Polymerase Chain Reaction (PCR) Testing Market Report 2026
Global Outlook – By Test Type (Sexually Transmitted Infection (STR) Polymerase Chain Reaction (PCR) Panels, Vaginitis Polymerase Chain Reaction (PCR) Panels), By Condition (Sexually Transmitted Infections, Vaginal Infections), By End User (Hospitals And Clinics, Diagnostic Laboratories, Homecare Or At-Home Testing, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Sexually Transmitted Infection (STI) And Vaginitis Polymerase Chain Reaction (PCR) Testing Market Overview
• Sexually Transmitted Infection (STI) And Vaginitis Polymerase Chain Reaction (PCR) Testing market size has reached to $0.68 billion in 2025 • Expected to grow to $1.08 billion in 2030 at a compound annual growth rate (CAGR) of 9.5% • Growth Driver: Rise In Sexual Health Screenings Driving Growth Of The Market Due To Increasing Demand For Early Detection And Treatment • Market Trend: Innovations Driving Growth In The STI And Vaginitis PCR Testing Market To Enhance Diagnostic Accuracy And Speed • North America was the largest region in 2025.What Is Covered Under Sexually Transmitted Infection (STI) And Vaginitis Polymerase Chain Reaction (PCR) Testing Market?
Sexually transmitted infection (STI) and vaginitis polymerase chain reaction (PCR) testing is a precise molecular diagnostic method that amplifies and detects the genetic material of various pathogens responsible for sexually transmitted infections and vaginal infections. This technique enables rapid and highly accurate identification of bacteria, viruses, and other microorganisms, allowing healthcare providers to diagnose infections more effectively and tailor treatments accordingly. The main test types of sexually transmitted infection (STI) and vaginitis polymerase chain reaction (PCR) testing are sexually transmitted infection (STI) polymerase chain reaction (PCR) panels and vaginitis polymerase chain reaction (PCR) panels. Sexually transmitted infections (STIs) are illnesses caused by bacteria, viruses, fungi, or parasites that spread largely via sexual contact, such as vaginal, anal, and oral sex. These tests cover a range of conditions, including sexually transmitted infections and vaginal infections, and serve various end users such as hospitals and clinics, diagnostic laboratories, homecare or at-home testing, and others.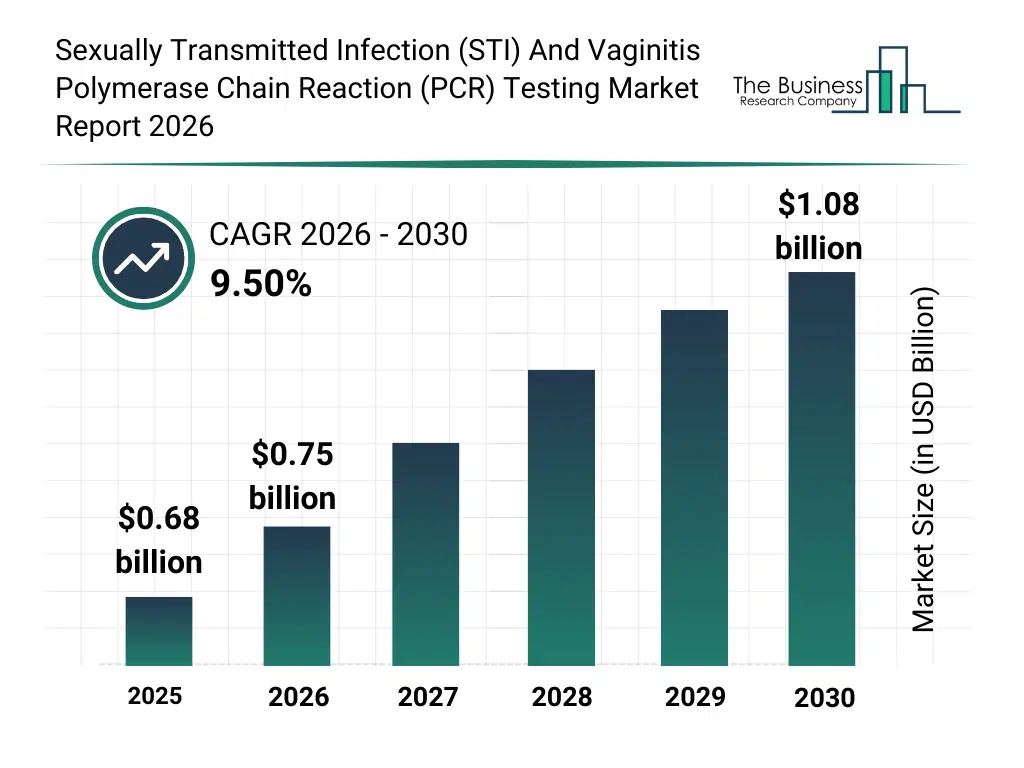
What Is The Sexually Transmitted Infection (STI) And Vaginitis Polymerase Chain Reaction (PCR) Testing Market Size and Share 2026?
The sexually transmitted infection (sti) and vaginitis polymerase chain reaction (pcr) testing market size has grown rapidly in recent years. It will grow from $0.68 billion in 2025 to $0.75 billion in 2026 at a compound annual growth rate (CAGR) of 10.3%. The growth in the historic period can be attributed to increasing prevalence of sexually transmitted infections, limitations of traditional diagnostic methods, growing awareness of women’s reproductive health, expansion of clinical diagnostic laboratories, adoption of standard pcr testing protocols.What Is The Sexually Transmitted Infection (STI) And Vaginitis Polymerase Chain Reaction (PCR) Testing Market Growth Forecast?
The sexually transmitted infection (sti) and vaginitis polymerase chain reaction (pcr) testing market size is expected to see strong growth in the next few years. It will grow to $1.08 billion in 2030 at a compound annual growth rate (CAGR) of 9.5%. The growth in the forecast period can be attributed to rising demand for point-of-care molecular diagnostics, technological advancements in multiplex and real-time pcr, increasing adoption of home-based testing solutions, growing investments in molecular diagnostic infrastructure, expanding screening programs for sti and vaginitis. Major trends in the forecast period include rising adoption of multiplex pcr panels for sti and vaginitis detection, increasing preference for rapid and high-sensitivity molecular diagnostics, growing demand for at-home and self-collected pcr testing kits, expansion of pcr testing in routine women’s health screening, enhanced focus on early and accurate pathogen identification.Global Sexually Transmitted Infection (STI) And Vaginitis Polymerase Chain Reaction (PCR) Testing Market Segmentation
1) By Test Type: Sexually Transmitted Infection (STR) Polymerase Chain Reaction (PCR) Panels, Vaginitis Polymerase Chain Reaction (PCR) Panels 2) By Condition: Sexually Transmitted Infections, Vaginal Infections 3) By End User: Hospitals And Clinics, Diagnostic Laboratories, Homecare Or At-Home Testing, Other End Users Subsegments: 1) By Sexually Transmitted Infection (STR) Polymerase Chain Reaction (PCR) Panels: Chlamydia Trachomatis Polymerase Chain Reaction, Neisseria Gonorrhoeae Polymerase Chain Reaction, Trichomonas Vaginalis Polymerase Chain Reaction, Herpes Simplex Virus Polymerase Chain Reaction, Human Papillomavirus Polymerase Chain Reaction, Mycoplasma Genitalium Polymerase Chain Reaction, Syphilis Polymerase Chain Reaction, Multiplex Sexually Transmitted Infection Polymerase Chain Reaction Panels 2) By Vaginitis Polymerase Chain Reaction (PCR) Panels: Bacterial Vaginosis Polymerase Chain Reaction, Candida Species Polymerase Chain Reaction, Trichomoniasis Polymerase Chain Reaction, Mixed Vaginitis Polymerase Chain Reaction Panels, Aerobic Vaginitis Polymerase Chain Reaction, Atrophic Vaginitis Polymerase Chain ReactionWhat Is The Driver Of The Sexually Transmitted Infection (STI) And Vaginitis Polymerase Chain Reaction (PCR) Testing Market?
The increasing sexual health screenings are expected to propel the growth of the sexually transmitted infection (STI) and vaginitis polymerase chain reaction (PCR) testing market going forward. Sexual health screenings refer to medical tests conducted to detect sexually transmitted infections (STIs) and other related conditions to ensure early diagnosis, treatment, and prevention of disease spread. Sexual health screenings are increasing due to the rising prevalence of sexually transmitted infections worldwide, which has heightened the need for early detection and timely treatment to prevent complications and reduce the spread of infections. Sexual health screenings play a crucial role in STI and vaginitis PCR testing by facilitating early and precise identification of various infections, often even in the absence of symptoms, which supports prompt treatment and helps prevent complications and the spread of infections. For instance, in July 2025, according to the Bristol City Council, a UK-based government agency, in 2024, sexual health services conducted 25,410 STI tests (excluding chlamydia screening for people under 25), equating to 5,260.9 tests per 100,000 population. This rate exceeds the England average of 4,088.8 per 100,000. Therefore, the increasing sexual health screenings are driving the growth of the sexually transmitted infection (STI) and vaginitis polymerase chain reaction (PCR) testing industry.Key Players In The Global Sexually Transmitted Infection (STI) And Vaginitis Polymerase Chain Reaction (PCR) Testing Market
Major companies operating in the sexually transmitted infection (sti) and vaginitis polymerase chain reaction (pcr) testing market are F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Abbott Laboratories, Danaher Corporation, Becton Dickinson and Company, Hologic Inc., QIAGEN N.V., ARUP Laboratories Inc., Seegene Inc., LetsGetChecked Ltd., Sansure Biotech Inc., Visby Medical Inc., R-Biopharm AG, Savyon Diagnostics Ltd., altona Diagnostics GmbH, SpeeDx Pty Ltd., CERTEST BIOTEC S.L., ALPCO Diagnostics, Atila Biosystems Inc., Ridacom Diagnostics Ltd., Sinochips Diagnostics Co Ltd., Vitassay S.L., Bioperfectus Technologies Co. Ltd.Global Sexually Transmitted Infection (STI) And Vaginitis Polymerase Chain Reaction (PCR) Testing Market Trends and Insights
Major companies in the sexually transmitted infection (STI) and vaginitis PCR testing market are advancing technologies such as multiplex real-time PCR assays to address the growing need for rapid, accurate, and comprehensive pathogen detection. Multiplex PCR technology enables the simultaneous amplification and detection of DNA or RNA from multiple pathogens in a single test, reducing both turnaround time and cost compared to traditional single-pathogen assays. This approach also improves sensitivity and specificity, allowing detection of low levels of pathogen genetic material—even in co-infections—that conventional culture or microscopy methods might miss or identify less reliably. For example, in February 2023, Thermo Fisher Scientific, a US-based biotechnology firm, launched the Applied Biosystems TrueMark STI Select Panel, a multiplex real-time PCR assay designed to simultaneously detect four common STIs: Chlamydia trachomatis, Neisseria gonorrhoeae, Trichomonas vaginalis, and Mycoplasma genitalium. Intended for research use, this test enhances clinical outcomes by providing faster, highly sensitive diagnostics while streamlining laboratory workflows, representing a significant advancement in molecular STI diagnostics.What Are Latest Mergers And Acquisitions In The Sexually Transmitted Infection (STI) And Vaginitis Polymerase Chain Reaction (PCR) Testing Market?
In December 2024, OraSure Technologies, a US-based medical device and diagnostics company, acquired Sherlock Biosciences for an undisclosed amount. With this acquisition, OraSure aims to expand its molecular diagnostics portfolio in sexual health by integrating Sherlock’s rapid, self collected STI testing capabilities and accelerating its move into the over-the-counter diagnostics space. Sherlock Biosciences, a US-based molecular diagnostics company, provides PCR-based testing for sexually transmitted infections (STIs) and vaginitis.Regional Outlook
North America was the largest region in the sexually transmitted infection (STI) and vaginitis polymerase chain reaction (PCR) testing market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Sexually Transmitted Infection (STI) And Vaginitis Polymerase Chain Reaction (PCR) Testing Market?
The sexually transmitted infection (STI) and vaginitis polymerase chain reaction (PCR) testing market consists of revenues earned by entities by providing services such as clinical consultations, comprehensive screening tests, multiplex testing, treatment monitoring, and sample collection guidance. The market value includes the value of related goods sold by the service provider or included within the service offering. The sexually transmitted infection (STI) and vaginitis polymerase chain reaction (PCR) testing market also includes sales of nucleic acid extraction kits, positive and negative control materials, automated PCR analyzers, and single-pathogen PCR test kits. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Sexually Transmitted Infection (STI) And Vaginitis Polymerase Chain Reaction (PCR) Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $0.75 billion |
| Revenue Forecast In 2035 | $1.08 billion |
| Growth Rate | CAGR of 10.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Test Type, Condition, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Abbott Laboratories, Danaher Corporation, Becton Dickinson and Company, Hologic Inc., QIAGEN N.V., ARUP Laboratories Inc., Seegene Inc., LetsGetChecked Ltd., Sansure Biotech Inc., Visby Medical Inc., R-Biopharm AG, Savyon Diagnostics Ltd., altona Diagnostics GmbH, SpeeDx Pty Ltd., CERTEST BIOTEC S.L., ALPCO Diagnostics, Atila Biosystems Inc., Ridacom Diagnostics Ltd., Sinochips Diagnostics Co Ltd., Vitassay S.L., Bioperfectus Technologies Co. Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
