
Specimen Validity Testing Market Report 2026
Global Outlook – By Product And Service (Products, Service), By Testing Type (Laboratory Testing, Rapid Or Point-Of-Care Testing), By End-User (Drug Rehabilitation Centers, Drug Screening Laboratories, Pain Management Centers, Criminal Justice And Law Enforcement Agencies, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Specimen Validity Testing Market Overview
• Specimen Validity Testing market size has reached to $4.03 billion in 2025 • Expected to grow to $5.95 billion in 2030 at a compound annual growth rate (CAGR) of 7.8% • Growth Driver: Impact Of Increased Drug Abuse On The Specimen Validity Testing Market • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Specimen Validity Testing Market?
Specimen Validity Testing (SVT) is a process used to assess the integrity and validity of biological specimens, such as urine, blood, saliva, or hair samples, collected for drug testing or clinical analysis. SVT examines various parameters such as pH, creatinine levels, specific gravity, and the presence of common masking agents such as oxidizing adulterants, nitrites, glutaraldehyde, and pyridinium chlorochromate. The main products and services in the specimen validity testing market are products and services. In the context of the specimen validity testing market, products refer to items such as assay kits, reagents, calibrators, and disposables used in the testing process to detect any adulteration, dilution, or substitution in urine samples. The various testing types are laboratory testing, and rapid or point-of-care testing used by various end-users including drug rehabilitation centers, drug screening laboratories, pain management centers, criminal justice and law enforcement agencies, and others.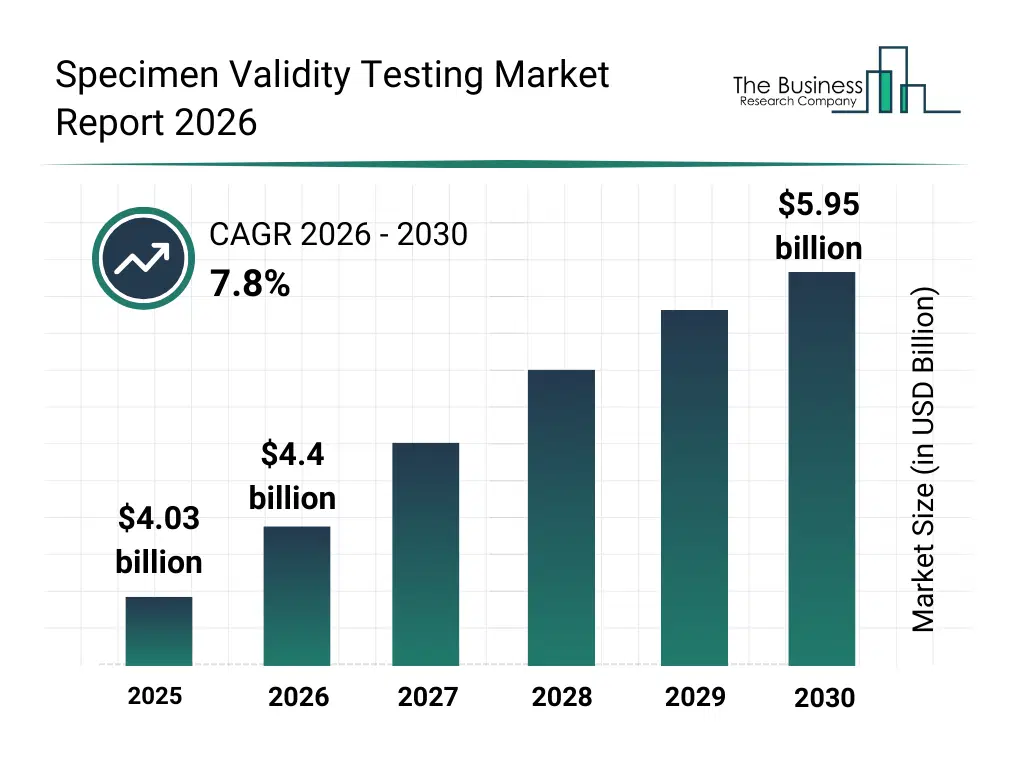
What Is The Specimen Validity Testing Market Size and Share 2026?
The specimen validity testing market size has grown strongly in recent years. It will grow from $4.03 billion in 2025 to $4.4 billion in 2026 at a compound annual growth rate (CAGR) of 9.3%. The growth in the historic period can be attributed to growth in workplace drug testing, regulatory mandates for urine testing, rise in substance abuse cases, forensic testing demand, expansion of clinical laboratories.What Is The Specimen Validity Testing Market Growth Forecast?
The specimen validity testing market size is expected to see strong growth in the next few years. It will grow to $5.95 billion in 2030 at a compound annual growth rate (CAGR) of 7.8%. The growth in the forecast period can be attributed to oral fluid testing adoption, technological advancements in assay sensitivity, increasing probation and legal testing, expansion of point-of-care solutions, emerging market penetration. Major trends in the forecast period include increased adulterant detection panels, automation of validity testing workflows, integration with lab information systems, rapid testing kits, workplace drug screening expansion.Global Specimen Validity Testing Market Segmentation
1) By Product And Service: Products, Service 2) By Testing Type: Laboratory Testing, Rapid Or Point-Of-Care Testing 3) By End-User: Drug Rehabilitation Centers, Drug Screening Laboratories, Pain Management Centers, Criminal Justice And Law Enforcement Agencies, Other End-Users Subsegments: 1) By Products: Reagents And Assay Kits, Controls And Calibrators, Specimen Collection Devices, Analyzers And Instruments 2) By Service: Laboratory Testing Services, Point-Of-Care Testing ServicesWhat Are The Drivers Of The Specimen Validity Testing Market?
The increased drug abuse is expected to propel the growth of the specimen validity testing market going forward. Drug abuse refers to the customary, excessive, or inappropriate use of legal or illegal substances for non-medical purposes. Drug abuse is increasing due to several interconnected factors, such as the opioid habit, developing mental health issues, barriers to treatment and support resources, and cultural depictions. SVT methods are developed to identify the presence of impurities and compounds by examining several parameters such as pH, specific gravity, creatinine content, and temperature. For instance, in October 2024, according to the Office for National Statistics, a UK-based government agency, in 2023, England and Wales recorded 5,448 drug poisoning-related deaths, marking an 11.0% increase from the 4,907 deaths registered in 2022. Therefore, the increased drug abuse is expected to propel the growth of the specimen validity testing industry. The rising healthcare expenditure is expected to propel the growth of the specimen validity testing market going forward. Healthcare expenditure refers to the total financial resources spent by individuals, organizations, or governments on preventing, treating, and managing health conditions. Healthcare expenditure is increasing due to advancements in medical technology, as new diagnostic tools, treatments, and innovative therapies often come at higher costs, leading to greater overall spending in healthcare. Rising healthcare expenditure drives specimen validity testing by funding advanced technologies, skilled personnel, and improved laboratory infrastructure, enhancing test accuracy and reliability. For instance, in December 2024, according to the Centers for Medicare & Medicaid Services, a US-based agency responsible for administering the Medicare and Medicaid programs, in 2023, U.S. health care expenditures increased by 7.5%, climbing to $4.9 trillion, or roughly $14,570 for each individual. Therefore, the rising healthcare expenditure is driving the growth of the specimen validity testing industry.Key Players In The Global Specimen Validity Testing Market
Major companies operating in the specimen validity testing market are Thermo Fisher Scientific Inc., Abbott Laboratories, Danaher Corporation, Siemens Healthineers , Medline Industries Inc., Becton, Dickinson and Company, Laboratory Corporation, Quest Diagnostics Incorporated, Alere Inc., Clinical Reference Laboratory Inc., Precision Diagnostics, ACM Global Laboratories, Millennium Health, Omega Laboratories Inc., Dominion Diagnostics, CannAmm, Sciteck Inc., Starplex Scientific Inc., Vision Diagnostics Inc., Premier Biotech Inc., Alfa Scientific Designs, Express Diagnostics, American Bio Medica Corporation, Titan Biotech Ltd., SureHire, Miraclean Technology Co. LtdWhat Are Latest Mergers And Acquisitions In The Specimen Validity Testing Market?
In November 2024, SPT Labtech, a UK-based manufacturer and designer of automated laboratory instruments partnered with ICE Bioscience. The partnership aims to accelerate drug discovery through advanced drug screening and automation, utilizing cutting-edge technologies. By enhancing assay development and broadening research capabilities, the collaboration strives to foster innovation within the pharmaceutical industry. ICE Bioscience is a China-based provider of drug screening platform.Regional Outlook
North America was the largest region in the specimen validity testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Specimen Validity Testing Market?
The specimen validity testing market includes revenues earned by entities by creatinine testing, pH testing, adulterant testing, and specific gravity measurement and related specimen collection containers, urine test strips, adulterant detection kits, and temperature strips. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Specimen Validity Testing Market Report 2026?
The specimen validity testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the specimen validity testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Specimen Validity Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $4.4 billion |
| Revenue Forecast In 2035 | $5.95 billion |
| Growth Rate | CAGR of 9.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product And Service, Testing Type, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Abbott Laboratories, Danaher Corporation, Siemens Healthineers , Medline Industries Inc., Becton, Dickinson and Company, Laboratory Corporation, Quest Diagnostics Incorporated, Alere Inc., Clinical Reference Laboratory Inc., Precision Diagnostics, ACM Global Laboratories, Millennium Health, Omega Laboratories Inc., Dominion Diagnostics, CannAmm, Sciteck Inc., Starplex Scientific Inc., Vision Diagnostics Inc., Premier Biotech Inc., Alfa Scientific Designs, Express Diagnostics, American Bio Medica Corporation, Titan Biotech Ltd., SureHire, Miraclean Technology Co. Ltd |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
